Demyelinating Optic Neuritis
All content on Eyewiki is protected by copyright law and the Terms of Service. This content may not be reproduced, copied, or put into any artificial intelligence program, including large language and generative AI models, without permission from the Academy.
Introduction
Demyelinating optic neuritis (ON) in an adult is one of the most common reasons for a young patient to seek consultation with a neuro-ophthalmologist. This brief guide will help the general ophthalmologist to understand the following:
- ON is a clinical diagnosis, thus magnetic resonance imaging (MRI) of the brain and orbit with gadolinium is helpful to document optic nerve enhancement as well as demyelinating white matter lesions in the brain. Although in typical cases, serologic testing for inflammatory, infiltrative, or infectious etiologies are not necessary, in atypical cases these tests may help to differentiate demyelinating or idiopathic ON from alternative etiologies. Likewise, cerebrospinal (CSF) analysis is not required for the diagnosis, but can be considered for cases which are atypical (e.g., marked visual loss at onset, bilateral ON, cases which are progressive or do not recover).
- Typical (i.e., acute, unilateral vision loss, relative afferent pupillary defect (RAPD), pain with eye movement, and normal fundus exam in a young patient) ON will usually recover with good visual prognosis by a few weeks and normal or near normal visual function by 6-12 months. Lack of visual recovery or progressive visual loss is atypical for ON.
- The final amount of vision recovery is independent of treatment with or without corticosteroids, however in the ON Treatment Trial (ONTT), intravenous (IV) steroids hastened the speed of recovery but not the final visual outcome compared with either oral prednisone (1 mg/kg/day) or placebo. Thus, although the natural history and prognosis of typical ON is very favorable with or without steroid treatment, steroids can be considered for speeding the rate of recovery.
- Oral prednisone alone in standard doses (1 mg/kg/day) however is not recommended for ON because in the ONTT there was a higher rate of recurrence compared to either placebo or intravenous (IV) steroids followed by oral prednisone. Higher dose (1250 mg/day) oral prednisone however could be considered on a case by case basis.
- MRI of the brain is helpful after an initial episode of ON because the number of lesions seen can stratify the patient’s risk for developing clinically definite (CD) multiple sclerosis (MS) after a clinically isolated syndrome (CIS). MRI of brain can detect radiographic dissemination in time and space as described by the McDonald criteria.
- The most common neurologic disorder associated with ON is MS and ophthalmologist can play a vital role to decrease morbidity from MS by understanding the role of early diagnosis and referral to neurology for consideration for MS treatment with disease modifying drugs (DMD).
This article will focus on adults, 18 years and older who present with ON.
Disease Entity
- Optic neuritis ICD 377.3
- Retrobulbar neuritis (acute) ICD 377.32
- Optic neuritis unspecified, ICD 377.30
Disease
ON is characterized by acute, unilateral decreased vision, optic nerve dysfunction (decreased central and/or peripheral vision, decreased color vision, decreased contrast/brightness sense, RAPD and tends to be associated with periorbital pain worse with eye movement. Periocular pain (62 to 92%) may precede the visual loss by days and is characteristically precipitated or aggravated by eye movement (87%). When the clinical history and examination suggest ON and the optic disc appears normal, the term retrobulbar ON is used. It may be the initial presentation of MS with obvious implications for future diagnosis, prognosis and treatment. Prevalence roughly estimated to be 1-5/100,000 depending upon geography and ethnicity.
Etiology
Believed to be an autoimmune, inflammatory, demyelination process, many different pathophysiologic mechanisms have been proposed and remain under investigation.
Risk Factors
- 3:2-female:male ratio
- Young age (20-45 years old)
- A prodromal flu like illness commonly accompanies the event but does not always occur
- Patients with MS often have ON and up to 75% of patients with CDMS will have at least one episode of ON in their lifetime. In addition, autopsies of MS patients show up to 90% had some optic nerve involvement.
General Pathology
Immune-mediated inflammatory demyelination of the optic nerve. The myelin undergoes destruction causing axons to poorly conduct impulses and the axons themselves become damaged, depending on the degree of inflammation within the lesion. Thus, retinal ganglion cell axons often become damaged in ON.
Pathophysiology
After myelin destruction occurs, the retinal ganglion cell axons start to degenerate. Monocytes localize along blood vessels and macrophages follow to remove myelin. Astrocytes then proliferate with deposition of glial tissue where axons may have been present before. These gliotic (sclerotic) areas can be multiple in number throughout the brain and spinal cord, hence the term multiple sclerosis.
Many different theories have been proposed for MS but none are proven. One theory is that a viral trigger may be responsible for the autoimmune inflammatory response. One recent study found viral DNA in the CSF of some ON patients indicating that there might be an associated role for varicella zoster virus and herpes simplex virus in the development of this disease.[1]
Diagnosis
ON is a clinical diagnosis that is made when a patient presents with:
- Unilateral (but may be bilateral) decreased visual acuity
- Pain with eye movement and/or periorbital discomfort
- RAPD if unilateral or bilateral and asymmetric
- Visual field defect (nerve fiber layer or central loss)
- Normal-appearing fundus (retrobulbar ON) although optic nerve swelling can be seen in up to 35% of cases.
History
The importance of the optic nerve has been known since the time of the Greeks. Galen recognized it as a conduit from the brain to the eye, but not until the Renaissance period was the optic nerve and the retina recognized to be the actual organ of vision instead of the lens. Despite misunderstandings about the exact function of the optic nerve, early medieval physicians including Hunayn ibn Ishaq in the 800s recognized that a poorly reactive dilated pupil could indicate damage or obstruction at the level of the optic nerve.
After the introduction of the ophthalmoscope in the late 1800s, a clearer understanding of ocular diseases with respect to etiology was worked out. Thomas Allbutt first suggested that ON might be caused by toxins and infectious agents and made the point that it was most often associated with other neurological abnormalities.
Interestingly, nearly 2000 years of medical history recognized some association between menstruation cessation and optic nerve disease, but not until Harvey Cushing in the early 1900s was the etiology shown to be pituitary tumors compressing the optic chiasm.[2]
Symptoms
- Prodromal viral illness, may or may not be present
- Acute vision loss that tends to worsen over days and nadirs around 2 weeks prior to improvement. Eye discomfort or pain particularly with eye movement (mild to severe)
- Phosphenes may occur (flashes of lights)
- Vision may become blurry when body temperature rises during exercise or bathing in warm to hot water (Uhthoff phenomenon)
- "Washed out" color vision
- Altered perception of motion (Pulfrich phenomenon)
Signs
- Decreased visual acuity
- Decreased color vision (red desaturation) and/or decreased contrast/brightness sense
- RAPD unless both eyes are affected or RAPD was present prior in contralateral eye
- Other efferent lesions may be present in MS (e.g., ocular dysmetria or internuclear ophthalmoplegia)
- Any nerve fiber layer or central scotoma on Amsler grid and/or confrontation visual fields or formal visual field testing
- Typically normal fundus but optic disc edema can be seen in up to 35%. Thus lack of optic disc edema is the rule rather than the exception for ON.
- Retinal vascular sheathing, pars planitis (periphlebitis occurs in 5-10% of MS patients)
Physical examination
- Snellen visual acuity with best correction for distance and/or near vision
- Color vision and/or Contrast measurement (e.g., Pelli-Robson or VisTech)
- Swinging flashlight test for RAPD
- Evaluation of extraocular movement
- Confrontation and automated visual fields (although automated perimetry has proven extremely useful in establishing the progression and recovery of visual field loss in ON, the patterns of visual field defects are not particularly helpful in distinguishing ON from any other form of optic nerve disease (eg, ischemic, compressive)).
- Amsler grid with best correction central visual field defects (be aware that this test may also be abnormal in macular disease)
- Biomicroscopy/direct ophthalmoscopy of the optic nerve and retina
Clinical Diagnosis
ON is diagnosed clinically by symptoms of acute unilateral decrease in vision, eye pain (especially with movement), RAPD (in unilateral or bilateral asymmetric cases), decreased color vision/contrast/brightness sense and a visual field defect.
Less commonly, patients may present with absence of eye pain, absence of orbital discomfort, bilateral, marked vision loss, and severe optic disc swelling with hemorrhages or retinal exudates. These cases should be considered for further work up to determine if an underlying inflammatory or infectious process can be found.
An MRI depicting enhancement of the optic nerve after administration of gadolinium is helpful but not required to make the clinical diagnosis. The main indication of MRI is derived from its prognostic value for the presence of or future development of MS. MRI is far superior to computed tomography (CT) in the diagnosis of ON. In a retrospective observational series of patients with acute ON who had gadolinium-enhanced fat-suppressed cranial MRI scans within 20 days of the visual loss, enhancement of the orbital optic nerve occurred in (94%) of patients. Optic nerve enhancement is not specific for ON and a similar appearance on MRI can be seen in many other inflammatory optic neuropathies.
Diagnostic procedures
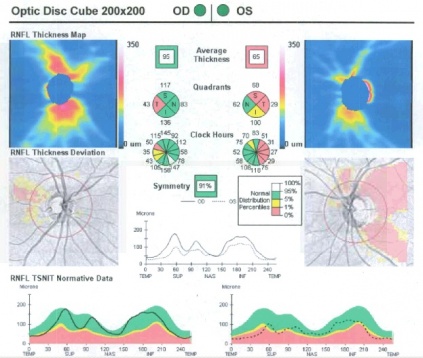
Optical coherence tomography (OCT) of the peripapillary retinal nerve fiber layer (pRNFL) is helpful as an adjunctive measurement of nerve function in patients with ON. OCT can quantify the onset of optic disc pallor that ensues 6-8 weeks post onset of ON. Optic atrophy can be subtle on biomicroscopy, thus OCT is useful for detection and quantification of the optic atrophy. OCT of the pRNFL also serves in patient education and documentation. Figure 1 below shows an example of an OCT image from a patient’s right eye 2.5 months after and left eye 3.5 months after presentation. In retrobulbar ON however, the OCT may initially be normal and only show pRNFL or macular ganglion cell loss downstream after recovery from acute ON.
It is known that ON demonstrates a delayed response on pattern visual evoked potentials (P-VEPs), and there has been recent interest in using trans-orbital sonography to enhance diagnostic accuracy for ON by measuring differences in optic nerve sheath diameter. One study showed a 68% sensitivity and 88% specificity for trans-orbital sonography but it was not found to be superior to P-VEP.[3][4]
In order to determine whether ON represents a CIS or related to MS, the McDonald criteria are applied. These criteria aim to diagnose MS when a patient presents with CIS likely to be associated with MS. Recent recommendations are to obtain an MRI in all patients for whom the clinician has a high suspicion of MS. Spinal cord MRI in these cases is not mandatory but should be considered in patients with spinal involvement, patients with progressive symptoms, and patients who are from atypical populations (e.g. non-white or elderly individuals). Figure 2 below summarizes the 2017 McDonald criteria.[5] Changes from the 2010 criteria include a stipulation that in patients with a clinically isolated syndrome and clinical or imaging evidence of dissemination in space who don’t fulfill the 2010 criteria for dissemination in time according to MRI, CSF analysis showing oligoclonal bands provides sufficient support for a confident diagnosis of MS. Importantly, lesions of the optic nerve seen on imaging should not be considered in determination of dissemination in space when applying the McDonald criteria due to insufficient evidence.[5]
The criteria also state that on MRI, one or more T2 hyperintense lesions in the periventricular, cortical or juxtacortical, infratentorial, or spinal cord regions is demonstrative of dissemination in space. There is no need to differentiate between symptomatic and asymptomatic lesions for diagnostic purposes in the most updated guidelines. Dissemination in time is demonstrated by gadolinium-enhanced and non-enhanced lesions appearing simultaneously, or new lesions appearing on recent imaging compared to older imaging.[5]
The clinician should keep in mind that periventricular lesions, which can be classic for MS, are also nonspecific, and can be seen as non-specific findings in the elderly.
When approaching a patient with ON, an MRI of brain and orbits should be obtained.[6] Imaging can show some broad differences between etiologies of ON whether myelin oligodendrocyte glycoprotein (MOG)-ON, neuromyelitis optica spectrum disorder (NMOSD) -ON, or MS-ON. Bilateral involvement of the optic nerves is more common in NMOSD-ON and MOG-ON. Retrobulbar optic nerve involvement is seen more often in MOG-ON, and intracranial involvement is seen more often in NMOSD-ON. MOG-ON may be associated with optic nerve sheath and surrounding orbital fat enhancement. Figure 3 summarizes the imaging characteristics of ON.[7]
Figure 1
Figure 2: 2017 McDonald criteria for diagnosing multiple sclerosis in patients with an attack at onset[5]
|
Number of lesions with objective clinical evidence |
Additional data needed for diagnosing multiple sclerosis | |
|---|---|---|
|
≥2 clinical attacks |
≥2 |
None |
|
≥2 clinical attacks |
1 (plus clear historical evidence of a previous attack with a lesion in a distinct anatomical location) |
None |
|
≥2 clinical attacks |
1 |
Dissemination in space demonstrated by an additional clinical attack implicating a different CNS site or by MRI |
|
1 clinical attack |
≥2 |
Dissemination in time demonstrated by an additional clinical attack or by MRI OR demonstration of CSF-specific oligoclonal bands |
|
1 clinical attack |
1 |
Dissemination in space demonstrated by an additional clinical attack implicating a different CNS site or by MRI AND Dissemination in time demonstrated by an additional clinical attack or by MRI OR demonstration of CSF-specific oligoclonal bands |
Figure 3: Diagnostic Imaging Clues in ON[7]
|
NMOSD-ON |
MOG-ON |
MS-ON | |
|---|---|---|---|
|
Distribution of ON lesions |
Bilateral |
Bilateral |
Unilateral |
|
Segment involvement |
Intracranial, chiasmal, optic tract |
Retrobulbar |
Retrobulbar and canalicular |
|
Length of lesions |
Longitudinally extensive |
Longitudinally extensive |
Short segment/focal |
|
Degree of ON swelling |
Mild |
Severe |
Mild |
|
Location of postcontrast enhancement |
Optic nerve |
Optic nerve and perineural |
Optic nerve |
|
Presence of brain MRI lesions |
Commonly observed |
Infrequently observed |
Frequently observed |
|
Location/characteristics of brain lesions |
Hypothalamic lesions more common than MOG-ON and MS-ON; posterior fossa and periaqueductal gray |
Large, tumefactive lesions; cortical and subcortical lesions |
Periventricular, ovoid lesions; subcortical and juxtacortical lesions |
A lumbar puncture is not required for the diagnosis of ON. A lumbar puncture can also help to rule out elevated intracranial pressure, and in cases of atypical bilateral ON with bilateral anterior disc edema. The CSF can also be examined for infectious and inflammatory conditions as well in atypical cases of optic neuritis.
Laboratory Tests
Bilateral ON with poor visual recovery is atypical and further testing with blood tests for NMOSD, MOG and other infectious and inflammatory diseases and neuroimaging of the brain and spine may be considered.
If vision loss has occurred in a young male with a significant family history of maternally-related males with bilateral vision loss, genetic consulting and testing should be entertained for Leber hereditary optic neuropathy (LHON). Patients with LHON are much less likely to recover vision, and the contralateral eye is often affected within weeks to months of the first eye.
Differential diagnosis
Differential Diagnosis for Retrobulbar Optic Neuritis (normal appearance of optic nerve and vision loss)
- Of note, the conditions noted below tend to be painless in nature, whereas 92% of patients with demyelinating ON present with some form of eye pain and/or eye pain with movement.
With a Relative Afferent Pupillary Defect
- Compressive lesions like meningioma or sphenoid sinus mucocele
- Central serous chorioretinopathy (OCT of the macula can help to rule out macular etiology)
- Incipient central retinal artery occlusion
- Posterior ischemic optic neuropathy
- Toxic/metabolic or drug-induced (ethambutol,[8] linezolid,[9] imatinib[10] – these present early without optic atrophy and tend to be more symmetric)
- Paracentral acute middle maculopathy
No Relative Afferent Pupillary Defect
- Visual field defects from lesions beyond the lateral geniculate body.
- Retinal degeneration such as retinitis pigmentosa
- Macular disease (OCT of the macula helpful)
- Age-related macular degeneration
- Macular edema (post-cataract surgery, diabetic)
- Central serous chorioretinopathy
- Macular hole (traumatic, idiopathic)
- Epiretinal membrane/surface wrinkling maculopathy
- Acute macular neuroretinopathy (AMNR)
Differential Diagnosis of Optic Neuritis (unilateral optic disc edema, vision loss)
- NMOSD (Devic disease)
- MOG immunoglobulin G-associated disorder (MOGAD)[11][12]
- Glial fibrillary acidic protein (GFAP) meningoencephalomyelitis[12] (often visually asymptomatic with bilateral disc edema and imaging showing linear periventricular radial enhancement)
- Anterior ischemic optic neuropathy (tends to be painless in nature)
- Neuroretinitis (early before the macular star formation)
- Chronic relapsing inflammatory optic neuropathy (CRION) (painful)
- Pending central retinal vein occlusion (painless)
- Optic papillitis in patient with uveitis (e.g. Vogt-Koyanagi-Harada (VKH) syndrome, sarcoidosis)
- Diabetic papillopathy (painless)
- Compressive lesion along the anterior pathway of the optic nerve (up to and including optic chiasm)
- Optic disc drusen (pseudodisc edema, visual field defects common due to crowding of the disc, painless)
- Infectious ON (e.g. syphilis, Lyme, Herpes virus (HSV, VZV); Bartonella—often with macular star in neuroretinitis)
- Vasculitis (e.g. systemic lupus erythematosus, granulomatosis with polyangiitis)
- Malignant hypertension (tends to be bilateral)
- Neoplastic (e.g. CNS leukemia, CNS lymphoma, metastatic lesion)
- Posterior scleritis (pain with eye movement, decreased vision, B-scan with thickened choroid and fluid-"T-sign")
- LHON (painless, telangiectatic disc vessels)
- Vascular lesion (e.g. juxtapapillary hemangioblastoma, combined hamartoma of the retina and retinal pigment epithelium)
- Radiation induced optic neuropathy
- Multiple evanescent white dot syndrome (MEWDS) (painless, "wreath sign" on fluorescein angiography)
Management
The ONTT was a landmark multi-center, randomized, prospective, controlled clinical trial designed to evaluate the efficacy and safety of oral prednisone vs. intravenous methylprednisolone followed by oral prednisone as compared with oral placebo for the treatment of acute ON. Patients were recruited from July 1988 to June 1991. 457 patients (85% Caucasian, range of 18-60 years old) at 15 different medical centers with presentation of symptoms of decreased vision at 8 days or less without any prior episodes of ON, not currently on any prednisone, and without any significant systemic diseases were randomized to either: 1) oral placebo, 2) 250mg IV solumedrol q6hrs for 3 days then 1mg/kg of prednisone for 11 days, or 3) 1mg/kg of PO prednisone alone for 14 days. Primary outcome measures were contrast sensitivity and visual field and secondary measures were vision and color. The patients were followed for a minimum of 6 months.
ONTT Outcomes
- IV steroid treated patients recovered faster within the first 4-6 weeks post onset. However at 6 months to 10 years out, there was no statistical difference in final visual outcome between IV-steroid treated patients and placebo.
- Oral steroids in conventional doses alone are contraindicated for acute ON because of higher rate of recurrences of ON
Practically speaking, physicians should consider the various causes of ON and have a high suspicion for ON as a clinically isolated syndrome associated with MS.
Treatment involves 1000 mg IV methylprednisolone daily for 3 days followed by an 11 day oral prednisone taper. This treatment has been shown to accelerate visual recovery, but not improve long-term outcomes. Oral formulations which approximate the bioavailability of the IV formulation also exist.[7]Adrenocorticotropic hormone has also been approved in subcutaneous or intramuscular injections for treatment. Depending on the etiology (NMOSD, MOG, MS, etc.) and response to therapy of ON there may be a role for plasma exchange (PLEX) and IV immunoglobulin therapy depending on clinician judgement. Further study determining timing of treatment and benefits of these modalities is needed.[7]
Medical therapy
- Side effects of IV steroids should be fully discussed, especially in diabetics.
- Low dose (60 mg/day) oral steroids are contraindicated in patients who have acute ON especially in patients that carry a diagnosis of MS
- Some MS specialists and neuro-ophthalmologists are using a methylprednisolone (Solumedrol) smoothie. Instead of administering the 1 gram of Solumedrol IV, they dilute the powder in a smoothie drink and the patient drinks one dose daily for 3 days. The smoothie drink camouflages the bitter taste. It is generally not available at local pharmacies in this form. Also, repository corticotropin (intramuscular/subcutaneous) injection at 80 units/1 ml has been used to treat acute ON.
Medical follow-up
Visual acuity testing (color vision and/or contrast can elucidate subtle changes not seen on Snellen acuity testing).
If neuroretinitis is unable to be ruled out as the cause, a dilated exam 4 weeks later should show the macular star if neuroretinitis was indeed the cause. A macular star figure would not be expected in MS however and suggests an atypical ON.
A fluorescein angiogram may be performed to rule out true optic nerve edema if LHON or other non-MS etiologies are suspected.
Visual field testing upon onset if possible (Humphrey or Goldmann) and repeat at follow up visual fields at 3, 6, and 12 months.
OCT of the pRNFL at onset, at 6-8 week visit, and at 6 month follow up for quantitation of optic atrophy.
All patients with suspected ON should have a fat-suppressed contrast-enhanced MRI at the time of presentation for diagnostic and prognostic purposes.
Complications
Visual prognosis is excellent with normal to near normal recovery. Patients may still complain of decreased brightness sense, contrast deficit, and loss of stereopsis. It is important to forewarn them at the beginning that even though visual recovery is the norm, they still have the possibility of:
- Permanent visual loss either mild (20/30) to severe (20/200 or worse)
- Permanent scotomas that may limit driving
- Recurrences
IV steroids at high dosages for the treatment of ON according to the ONTT can cause insomnia, mood changes, dyspepsia, weight gain, flushing, nausea, vomiting, and elevated blood pressure. Patients should be advised to take a proton pump inhibitor such as omeprazole and may need to seek medical attention for anxiety and insomnia. Diabetics may be at risk for episodes of hyperglycemia and also for diabetic ketoacidosis. Co-management with the patient's primary physician, internist or endocrinologist is a must. Patients tend to have more side effects from the oral prednisone taper. Some clinicians abbreviate the taper or prescribe no taper at all, others prescribe a prolonged taper over one month. It is advisable to prescribe calcium and vitamin D while taking the corticosteroids.
Prognosis
The vast majority (94%) of patients recover vision to 20/40 or better at 5 years. Only 3% of patients had 20/200 or worse visual outcomes at 5 years (based on the ONTT).
Visual recovery tends to occur by 1 month after onset and the majority recover within 1-3 months. At 6 months, patients tend to have similar visual outcomes no matter if they were treated with IV steroids or placebo. Vision improvement can take up to one year. Prolonged pain with eye movement, lack of recovery, recurrence within 2 months would alert the physician to re-evaluate for atypical causes of ON such as sarcoidosis, syphilis or an idiopathic autoimmune steroid-responsive ON .
As stated above, up to 28% of patients developed recurrent ON associated with oral prednisone. As a result, oral steroids at a dose of 1 mg/kg/day are contraindicated in the management of acute ON.
Since ON is common among patients who have MS, (up to 75% have at least one episode of ON in their lifetime), these patients are at risk for developing CDMS. The ONTT showed that even without any brain lesions present on MRI, there still was a 16% chance of developing MS in 5 years, 22% in 10 years and 25% in 15 years. One recent study in South Korea showed that the 7 year conversion rate to MS was 10.6%.[13]
At 10 years, the overall risk of MS was 38% after an initial episode of ON. That risk is 56% if the MRI had one or more brain lesions. The risk is lower if the patient is male, disc swelling is present, pain is absent, the patient has no visual light perception, severe optic disc swelling with hemorrhages are seen, or macular exudates such as a retinal star pattern occur. In patients with CDMS, the median time to diagnosis was 3 years and 34% of the diagnoses were made in the first 2 years whereas 72% were made within 5 years.
At 15 years, the overall risk of MS was 50% after an initial episode of ON and strongly related to presence of lesions on a baseline non-contrast-enhanced MRI of the brain. Twenty-five percent of patients with no lesions on baseline brain MRI developed MS during follow-up compared with 72% of patients with 1 or more lesions. Baseline factors associated with a lower risk for MS included male sex, sever optic disc swelling, and atypical features of ON.
Patients with signs and symptoms of a CIS such as ON require prompt MRI brain imaging for lesions to determine if patients are at high risk for the development of CDMS. The role of the initiation of DMDs for MS at the time of the first ON episode should be considered depending on the MRI brain findings as well as the neurological examination.
Additional Resources
- Patient Information Brochure. North American Neuro Ophthalmology Society (NANOS). https://www.nanosweb.org/Optic_Neuritis_information_brochure. Accessed March 3, 2025
- Boyd K, DeAngelis KD. What Is Optic Neuritis? American Academy of Ophthalmology. EyeSmart/Eye health. https://www.aao.org/eye-health/diseases/what-is-optic-neuritis. Accessed March 23, 2023.
References
- ↑ Ordoñez G, Rivas V, Santos M, et al. Herpes viruses in optic neuritis: Similar to Bell’s palsy. Clinical Neurology and Neurosurgery. 2020;188:105588. doi: https://doi.org/10.1016/j.clineuro.2019.105588.
- ↑ Reeves C, Taylor D. A history of the optic nerve and its diseases. Eye. 2004;18(11):1096-1109.
- ↑ Elkholy SH, El-Jaafary SI, Kotb MS, El Gohary AM, Elbhy BA. Trans-orbital sonography versus visual evoked potentials in acute demyelinating optic neuritis. Multiple Sclerosis and Related Disorders. 2020;40:101934. doi: https://doi.org/10.1016/j.msard.2020.101934.
- ↑ Schroeder C, Katsanos AH, Ayzenberg I, et al. Atrophy of optic nerve detected by transorbital sonography in patients with demyelinating diseases of the central nervous system. Eur J Neurol. 2020;27(4):626-632.
- ↑ Jump up to: 5.0 5.1 5.2 5.3 Thompson AJ, Banwell BL, Barkhof F, et al. Diagnosis of multiple sclerosis: 2017 revisions of the McDonald criteria. The Lancet Neurology. Invalid date;17(2):162-173.
- ↑ Meltzer E, E, Frohman EM FAU - Costello, M, Costello FE FAU - Burton, C, Burton JM FAU - Frohman, Frohman TC. Should spinal MRI be routinely performed in patients with clinically isolated optic neuritis? Journal of neuro-ophthalmology : the official journal of the North American Neuro-Ophthalmology Society JID - 9431308.
- ↑ Jump up to: 7.0 7.1 7.2 7.3 Horton L, Bennett JL. Acute management of optic neuritis: An evolving paradigm. Journal of neuro-ophthalmology : the official journal of the North American Neuro-Ophthalmology Society. Invalid date; 38(3):358-367.
- ↑ Fang JT, Ming-Yang,Chen YC FAU - Chang, Chang MY. Ethambutol-induced optic neuritis in patients with end stage renal disease on hemodialysis: Two case reports and literature review. Renal failure JID - 8701128. (0886-022; 0886-022).
- ↑ Frippiat F, Bergiers C, Michel C, Dujardin J, Derue G. Severe bilateral optic neuritis associated with prolonged linezolid therapy. J Antimicrob Chemother. 2004;53(6):1114-1115.
- ↑ Rotstein DL, Sawicka K, Bharatha A, Montalban X, Lipton JH. CNS demyelination after initiating the tyrosine kinase inhibitor imatinib: A report of two cases. Mult Scler. 2019:1352458519892914.
- ↑ Chen JJ, Flanagan EP, Jitprapaikulsan J, et al. Myelin oligodendrocyte glycoprotein antibody-positive optic neuritis: Clinical characteristics, radiologic clues, and outcome. Am J Ophthalmol. 2018;195:8-15.
- ↑ Jump up to: 12.0 12.1 Levin MH. Demyelinating optic neuritis and its subtypes. Int Ophthalmol Clin. 2019;59(3).
- ↑ Lee J, Han J, Yang M, Oh SY. Population-based incidence of pediatric and adult optic neuritis and the risk of multiple sclerosis. Ophthalmology. 2020;127(3):417-425.
- Miller NR, Newman NJ, et al, eds. Walsh & Hoyt's Clinical Neuro-ophthalmology. 6th ed. Lippincott Williams & Wilkins; 2005:3460-3497.
- Neuro-Ophthalmology, Section 5. Basic and Clinical Science Course, AAO, 2003.
- Cleary PA, Beck RW, Anderson MM Jr, et al. Design, methods, and conduct of the Optic Neuritis Treatment Trial. Control Clin Trials. 1993;14(2):123-42.
- Optic Neuritis Study Group. The 5-year risk of MS after optic neuritis. Experience of the Optic Neuritis Treatment Trial. Neurology. 1997;49:1404-1413.
- Trobe JD, Sieving PC, Guire KE, et al. The impact of the optic neuritis treatment trial on the practices of ophthalmologists and neurologists. Ophthalmology. 1999;106(11):2047-53.
- Jacobs LD, Beck RW, Simon JH, et al. Intramuscular interferon beta-1a therapy initiated during a first demyelinating event in multiple sclerosis. CHAMPS Study Group. N Engl J Med.2000;343:898-904.
- McDonald WI, Compston A, Edan G, et al. Recommended diagnostic criteria for multiple sclerosis: guidelines from the International Panel on the diagnosis of multiple sclerosis. Ann Neurol.2001;50(1):121-7.
- Comi G, Filippi M, Barkhof F,et al.; Early Treatment of Multiple Sclerosis Study Group. Effect of early interferon treatment on conversion to definite multiple sclerosis: a randomised study. Lancet. 2001;357(9268):1576-82.
- Optic Neuritis Study Group. High- and low-risk profiles for the development of multiple sclerosis within 10 years after optic neuritis. Arch Ophthalmol. 2003;121:944-949.
- Fisher JB, Jacobs DA, Markowitz CE, et al. Relation of visual function to retinal nerve fiber layer thickness in multiple sclerosis. Ophthalmology. 2006;113(2):324-32. Epub 2006 Jan 10.
- Kappos L, Freedman MS, Polman CH, et al. Long-term effect of early treatment with interferon beta-1b after a first clinical event suggestive of multiple sclerosis: 5-year active treatment extension of the phase 3 BENEFIT trial. BENEFIT Study Group. Lancet Neurol. 2009;8(11):987-97. Epub 2009 Sep 10.
- Comi G. Shifting the paradigm toward earlier treatment of multiple sclerosis with interferon beta. Clin Ther. 2009;31(6):1142-57.
- El-Dairi M, Bhatti MT, Vaphiades MS. A shot of adrenaline. Surv Ophthalmol. 2009;54(5):618-24.
- Vaphiades MS1, Doyle J, Hudgins PA, Brat DJ. Acute monocular visual loss in an elderly woman: a neuro-ophthalmologic emergency. J Neuroophthalmol. 2013 ;33(4):390-3.
- Nikoskelainen E. Symptoms, signs and early course of optic neuritis. Acta Ophthalmol (Copenh). 1975;53:254-72.
- Trapp BD, Peterson J, Ransohoff RM, Rudick R, Mörk S, Bö L. Axonal transection in the lesions of multiple sclerosis. N Engl J Med. 1998 ;338(5):278-85.
- Sellebjerg F1, Nielsen HS, Frederiksen JL, Olesen J. A randomized, controlled trial of oral high-dose methylprednisolone in acute optic neuritis. Neurology. 1999 ;52(7):1479-84.
- Kupersmith MJ, Alban T, Zeiffer B, Lefton D: Contrast-enhanced MRI in acute optic neuritis: relationship to visual performance. Brain. 2002;125:812-22.
- Optic Neuritis Study Group. Multiple sclerosis risk after optic neuritis: final optic neuritis treatment trial follow-up. Arch Neurol. 2008;65(6):727-32.
- Chen JJ, Pittock SJ, Flanagan EP, Lennon VA, Bhatti MT. ON in the era of biomarkers. Surv Ophthalmol. 2020;65(1):12-17.
- Iorga RE, Moraru A, Ozturk MR, Costin D. The role of optical coherence tomography in optic neuropathies. Romanian journal of ophthalmology. 2018;62(1):3-14.