Secondary Acquired Nasolacrimal Duct Obstruction
All content on Eyewiki is protected by copyright law and the Terms of Service. This content may not be reproduced, copied, or put into any artificial intelligence program, including large language and generative AI models, without permission from the Academy.
Disease Entity
Secondary acquired nasolacrimal duct obstruction (SANDO) is an obstruction of the nasolacrimal duct caused by a secondary etiology, as opposed to the idiopathic primary acquired nasolacrimal duct obstruction (PANDO). To be classified as SANDO, a specific cause must be identified.
Disease
Epiphora is a very common complaint, and nasolacrimal duct obstruction (NLDO) is the most common cause of persistent epiphora. The incidence of NLDO is reported as 20.24 per 100,000. In this incidence study, NDLO accounted for 31.8% of all chronic epiphora and 67.6% of all tear drainage pathway obstructions.
Etiology
There are many causes of SANDO; however, five main categories of etiologies are common: infectious, inflammatory, neoplastic, traumatic, and mechanical. Infectious: These may be bacterial, viral, fungal, or even parasitic infections. There are many bacterial causes, but the most common pathogens are Staphylococcus, Streptococcus, and Actinomyces species. The most common infectious cause of SANDO is dacryocystitis.
Inflammatory: Causes of inflammation may be endogenous or exogenous. Examples of endogenous inflammation are sarcoidosis and granulomatosis with polyangiitis (GPA, formerly known as Wegener’s granulomatosis). Examples of exogenous inflammation are burns, allergies, eye drops (such as antiviral, chemotherapeutic, or radiotherapy agents), and radiation.
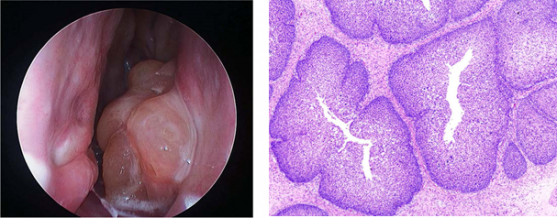
Neoplastic: Primary tumors that arise in the lacrimal system (papillomas, lymphomas, squamous cell carcinomas, and melanomas) can cause SANDO. Obstruction can also be caused from tumors in adjacent tissues, such as basal cell carcinomas, squamous cell carcinomas, adenoid cystic carcinomas, leukemia, and lymphomas. SANDO is rarely caused by metastasis, but breast carcinoma, malignant melanoma, and prostate carcinoma have been reported to cause obstruction.
Traumatic: Trauma includes both accidental and iatrogenic insults. Accidental trauma most commonly includes bone injury (such as naso-orbito-ethmoid fractures). Trauma is often mechanical, but can also be chemical, drug induced, or radiation induced.
Mechanical: Mechanical SANDO may occur with obstruction anywhere along the nasolacrimal duct, and examples include sinus mucoceles or dacryoliths. Mechanical obstruction of the canaliculi can cause dacryocystitis and indirectly lead to NLDO.
Pathophysiology
The specific pathophysiology of SANDO varies because each cause of obstruction can have a different pathophysiology. However, in most cases there is some type of inflammation and often infection.
NLDO secondary to infection: Infection can cause dysfunction of goblet cells, loss of blood vessels, and damage to epithelial cells. These changes impede the outflow of tears and eventually cause fibrosis. This resultant obstruction and fibrosis can exacerbate a cycle of recurrent infection, inflammation, and fibrosis. NLDO secondary to inflammation: The general pathophysiology of inflammation is similar to that of infection. Active inflammation causes hyperemia and mucous membrane edema, causing a temporary duct obstruction. This obstruction may then lead to the same cycle of recurrent infection, inflammation, and fibrosis described above. The specific pathophysiology will vary based on the cause of inflammation.
In NLDO secondary to radioactive iodine, the iodine enters epithelial cells of the nasolacrimal duct through the sodium/iodide symporter, and it then causes cell damage (inflammation and fibrosis). In NLDO secondary to sarcoidosis and GPA, granulomatous inflammation can cause distortion of the architecture of the lacrimal sac. GPA affects the eyes in two forms: contiguous and focal inflammation. In the contiguous form, inflammation originates in the paranasal sinuses or nasopharynx and then spreads to the orbit and adnexa. This is the most common route to NLDO secondary to GPA. However, recent case studies have shown that GPA can also cause a focal vasculitis of the nasolacrimal duct. This will also result in NLDO. Both forms of GPA can cause mucoceles and dacryocystitis, which almost always require surgery. NLDO secondary to neoplasm or mechanical obstruction: In these cases, the obstruction is caused by a mass in or outside the duct that causes obstruction directly or by compression.
NLDO secondary to trauma: The pathophysiology can vary significantly depending on the etiology. In iatrogenic cases, procedures or surgeries can cause infection, scarring, or direct obstruction. In accidental trauma, mid-face fractures commonly cause NLDO through edematous compression. Naso-orbito-ethmoidal fractures can directly injure the nasolacrimal duct, causing an inflammatory and cicatrizing reaction. These injuries can cause chronic fibrosis, scarring, and chronic NLDO. These obstructions can present shortly after injury but sometimes do not until years later.
Primary prevention
The best prevention is identifying these secondary causes before they cause obstruction. This is particularly true for the infectious, inflammatory, neoplastic, and traumatic etiologies. Naso-orbital fractures are often neglected or not recognized and may involve the nasolacrimal duct. Intubation may be performed at the time of fracture if there is concern the injury may involve the tear drainage pathway. Intubation should also be considered in all medial canthal tumors requiring radiation.
Diagnosis
History
As nearly all patients with SANDO complain of epiphora, the history should first attempt to determine if the epiphora is nonobstructive (hypersecretion or pump failure) or obstructive. Chronic, unrelenting epiphora is more common in obstruction, whereas transient, reproducible, or minor epiphora is suggestive of functional NLDO or a nonobstructive etiology. The history should also attempt to differentiate SANDO from PANDO by eliciting time of onset. Past ocular or systemic disease should be elicited, specifically a history of the following: sinus disease, previous trauma, previous ocular or nasal surgery, chemotherapy, eyedrop use, radiation, radioactive iodine, GPA, sarcoidosis, Kawasaki disease, scleroderma, and parasitic infections.
The history should also elicit all the patient’s symptoms and the associated duration and onset of symptoms.
Physical examination
Again, the specifics of the physical exam will depend largely on the presenting symptoms and clinical suspicion, but generally should include the following (many aspects rule out hypersecretion or pump failure):
External exam: The position of the eyelids should be evaluated, including the position of the eyelids, blinking ability, movement of the punctum, and closure strength. Other gross signs of infection, inflammation, or trauma should also be assessed.
Slit lamp: Key aspects of the slit lamp exam are observing the height of the tear lake (normal is 1 mm), the punctum patency and position, and the tear breakup time (normal is 15-30 seconds). The eyelid, conjunctiva, cornea, and anterior chamber should also be inspected (for indications of dry eye syndrome, inflammation, disease, or infection).
Canthus exam: This includes palpating the medial canthal region, feeling for a distended lacrimal sac. A tender lacrimal sac may be indicative of dacryocystitis. Reflux from the punctum suggests an obstruction in the lacrimal system. Firm canthal masses, bloody reflux from the punctum, and hypervascularity are suspicious of neoplasm.
Nasal exam: All patients with suspicion of SANDO should receive a nasal exam to rule out inflammatory, structural, or neoplastic abnormalities in the nasal passage. This is done with a nasal speculum or endoscope, if available.
Signs
Increased tear lake, epiphora, fluorescein pooling are the most common signs of NLDO. Often, patients with carry tissues with them at all times—periocular redness from excessive tissue use may be evident.
If the etiology is infectious, there may be a thickened canalicular portion of eyelid, an erythematous punctum, conjunctival injection, expressible punctal discharge, or even occasionally a lacrimal fistula. There may also be swelling of the lacrimal sac.
If the etiology is traumatic, a flattened nasal bridge, telecanthus, or a facial scar may be present. Facial bony abnormalities may also be apparent on external exam. Bloody punctal discharge is highly suggestive of neoplasm.
Symptoms
Epiphora is the common symptom of SANDO, regardless of etiology. Blurred vision may also be present. Besides epiphora, patients will usually complain of symptoms related to the secondary cause of the NDLO, and these symptoms vary significantly based on the etiology. For example, patients with obstruction secondary to infection may also complain of eyelid swelling, erythema, or pain.
Clinical diagnosis
SANDO is primarily a clinical diagnosis. Much of this diagnosis will be suggested by the physical exam described above. There are several additional clinical tests available to the clinician.
Dye disappearance test: examiner administers fluorescein drops and observes under cobalt blue filter. If the dye persists after 5 minutes, obstruction is suggested. Probing and irrigation test: a probe is used to dilate the punctum and a cannula is passed into the canaliculus and irrigated with saline. The system is patent if saline passes into the nose without reflux through either punctum. If there is reflux from the same punctum, the obstruction is likely canalicular. If there is reflux from the opposite punctum, there is likely obstruction in the common canaliculus. If the reflux from the opposite punctum has mucus or there is distention of the lacrimal sac, nasolacrimal duct obstruction is suggested.
The Jones 1 and 2 tests may also be employed; however, these tests appear to be falling out of favor. The Jones 1 test is performed by instilling fluorescein into the conjunctival fornices and passing a cotton-tip into the inferior meatus near the valve of Hasner to recover the dye. This is done at 2 and 5 minutes after instilling fluorescein. The Jones II test is performed after an unsuccessful Jones 1 test. The residual fluorescein is flushed from the conjunctival sac and the lacrimal drainage system is irrigated with clear saline. A cotton tip is used to test if fluorescein can be retrieved from the inferior meatus in the region of the nasolacrimal duct ostium. In the acute trauma setting, patients should receive a slit lamp exam when able. Tearing and fluorescein dye disappearance tests are unreliable in trauma patients as they have high false-positive results due to edema.
Diagnostic procedures
Diagnostic tests will depend on the patient presentation and clinical suspicion.
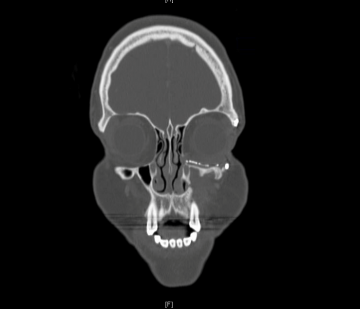
CT or MRI should be used when there is suspicion of traumatic, mechanical, or neoplastic obstruction. If there is suspicion for neoplasm, contrast is preferred, and scans should evaluate the lacrimal sac, orbit, and paranasal sinuses.
Dacryocystography is a radiographic procedure that involves dilating the lower punctum, inserting a small catheter, injecting contrast, and then imaging with x-ray or CT. Dacryocystography will visualize any blockage, trauma, inflammation, fistulas, diverticuli, tumors, or other anatomic abnormalities, but it does not differentiate well between the type of blockage. Due to modern imaging, dacryocystography is now rarely used.
Dacryoscintigraphy utilizes radionuclide tracers and gamma-gram imaging to examine the lacrimal drainage system. This method can detect incomplete obstructions and uses very little radiation, but it is less helpful in identifying complete obstructions or lacrimal neoplasms.
Laboratory test
For infectious etiologies, discharge should be sent for culture and susceptibilities. Cases associated with inflammatory ocular findings should include C-ANCA, angiotensin converting enzyme levels, and chest X-ray. Concerning masses should be biopsied. Closed biopsies are done with blunt needles passed through the punctum. Open biopsies are done transnasally through a skin crease or medial canthal incision. A positive biopsy and subsequent tumor resection will postpone surgical management of the NLDO (this is further discussed in the management and neoplastic sections below).
Differential diagnosis
The differential for excess lacrimation is extensive and includes: punctal malposition, lid laxity, eyelid malpositions, eyelid disease, ectropion, trichiasis, incomplete blink, blepharitis, irritation of trigeminal nerve, conjunctivitis, ocular surface tension, tear deficiency or instability, foreign body, and dry eye syndrome. There are also other lacrimal system obstructions or infections, such as canalicular obstruction or canaliculitis.
Management
General treatment
Specific treatment will vary based on the etiology of the obstruction, but in general treatment focuses on reestablishing the nasolacrimal duct or on creating new channels surgically. Therefore, identifying the cause of obstruction is the essential first step. Most complete obstructions will require surgery. Depending on the cause, available treatments have excellent success. Options for treating the obstruction are conservative (medical), procedural, and surgical.
Besides clearing the obstruction, it is also important to address the secondary cause, otherwise obstruction could recur. General treatment for each etiology is briefly discussed in the following sections. For a more detailed analysis, see individual pages on these topics.
Medical therapy
NLDO is primarily treated surgically. Infections should be treated with broad spectrum antibiotics (ciprofloxacin or Bactrim) and narrowed appropriately. Inflammatory etiologies may be treated with corticosteroids or immunomodulatory therapy. Treatment of neoplasm often involves chemotherapy and radiation. However, despite these medical therapies, most NLDO will require surgery.
Medical follow up
Appropriate treatment of infections and inflammation may prevent surgery, especially if diagnosis and treatment is done early. Patients should be followed regularly to insure the infection or inflammation is resolving. If antibiotic and anti-inflammatory medication does not improve the obstruction, the patient should undergo surgery after the offending etiology has been stabilized.
Surgery
Dacryocystorhinostomy (DCR) surgically bypasses the nasolacrimal duct and thus is the primary surgery for NLDO. It requires a patent punctum and canaliculus. A fistula is created from the lacrimal sac to the lateral nasal mucosa and a silicone tube is temporarily placed through this fistula to maintain patency. See the EyeWiki article a “Dacryocystorhinostomy” for more details about DCR.
DCR can be approached endoscopically (endonasal, or “endoDCR”) or via an external incision. External DCR is currently the gold standard of lacrimal bypass surgery, and multiple studies show a success rate over 95% for NLDO. Historically, external DCR has had greater success rates to endoscopic DCR. However, more recent studies suggest the success rate of endoDCR is approaching that of external DCR.[1] There is a high learning curve for endoscopic DCR, and this certainly influenced prior studies. Advantages of endoscopic DCR are that it is less invasive, has a shorter operative time, preserves lacrimal pump function, has faster recover, and avoids an external scar. However, the initial cost of endoscopic DCR can be high, including equipment price and the time spent learning the procedure. Endoscopic DCR assisted with laser ostium creation has been shown to have a lower success rate than endoDCR (71% and 83%, respectively in a study comparing the two), but has less bleeding and a quicker recovery time. Some authors suggest endoscopic DCR for the treatment of SANDO, as intranasal abnormalities can be directly visualized during surgery and additional nasal surgery can be accomplished if needed, generally with the assistance of an experienced rhinologist.
Conjunctivo-DCR is used for canalicular obstruction, bypassing the entire lacrimal system. It uses the Pyrex glass Jones tube to create a new channel extending from adjacent to caruncle into the nose. This is not used for NLDO alone as the tube must be irrigated periodically, has a propensity to migrate, and may allow reflux from the nose onto the eye. See the EyeWiki article “Conjunctivodacryocystorhinostomy with glass tube (endoscopic)” for further details on this surgery.
New research is being conducted on augmenting surgery with antimetabolites. These studies have mostly been done on cases of PANDO, but the treatments have also been useful in the treatment of SANDO. The studies have shown an increased efficacy and lower complication rates (decreased bleeding, mucosal necrosis, and infection post-operatively). Mitomycin has been more promising than 5-FU. Many revision surgeries are increasingly using mitomycin C.
Specific surgery considerations for each etiology of SANDO are explained below:
Infectious: It is ideal to defer surgery, if possible, until the infection has resolved with antibiotics. In infectious NLDO, abscess drainage may be necessary in addition to DCR.
Inflammatory: It is ideal to defer surgery, if possible, until the inflammation has resolved with corticosteroids. If delay is not possible, systemic corticosteroids should be administered prior to the operation. DCR is still the surgery of choice, but there is some discussion that dacryocystectomy may decrease the risk of recurrent obstruction. In systemic inflammatory cases, the inflammation may progress to involve the lacrimal sac mucosa and cause obstruction after the DCR. There may be a role for post-operative steroids as well. A growing number of surgeons are using budesonide rinses post-operatively in cases of acute inflammation (acute dacryocystitis, radiation induced, GPA, etc.) and have reported improved success rates.
Neoplastic: In general, the neoplasm is the priority and should be treated first before managing the obstruction. It is suggested to wait 5 years after tumor resection, confirming that there is no recurrence, prior to DCR (or other surgery) due to the risk of occult tumor spread.
Specific treatment of the tumor depends on the type of tissue and the extent of obstruction. Benign lesions can be managed by limited excision or dacryocystectomy. Papillomas, however, have a high recurrence rate and may become malignant, so they should be treated more aggressively. In localized malignant neoplasms, complete excision of the NLD should occur under frozen section control. Residual tumor should be treated with radiotherapy. More extensive lesions may require orbital exenteration and flap reconstruction. Lymphoproliferative disorders should have a systemic workup. Those that are localized can be treated with irradiation. Those that are systemic will require chemotherapy and additional treatment.
With lacrimal tumors, usually the entire lacrimal apparatus should be removed. The patient may also be treated with radiation therapy pre- or post-operatively. If the tumor has extended into the orbit, orbital exenteration and/or sinusectomy are required. If there is systemic metastasis, systemic chemotherapy and/or radiation therapy are required.
Traumatic: For nasolacrimal duct injuries, it is generally recommended to probe early and operate late. If probing fails, obstruction may occur years after the initial insult and will likely warrant DCR. When traumatic obstruction is suggested, bicanalicular intubation should be performed prophylactically and left in place for 3 to 6 months. After extubation, if epiphora remains and canalicular injury is ruled out, DCR is the surgery of choice. Delaying DCR has not been shown to decrease outcomes. In challenging surgical cases due to trauma, one study found that image-guided dacryolocalization was very helpful. This imaging was done with the Fusion ENT navigation system, which can localize precise lesions in a CT constructed 3-dimensional space. This technology is just beginning to be explored in DCR surgeries and may allow for more aggressive surgery with improved results.
Procedures: Probing with silicone tubing intubation and balloon dacryloplasty has been reported to successfully treat a subset of acquired NLDO, specifically in partial NLD obstruction, however the role in SANDO has not been established. Similarly, nasolacrimal stenting is mostly reported for PANDO, and it had a lower success rate than alternative treatments.
Surgical follow up
Patients should avoid exercise and strenuous activity for one week after DCR. Patients are advised to avoid blowing their noses during this week. Patients should return for follow-up one month and 2-3 months after the surgery. The silicone tubes placed during DCR are usually removed in the office 4-8 weeks after the surgery, depending on the degree of inflammation. In certain cases of SANDO with a propensity for robust postoperative inflammation, some authors advocate for endonasal debridement in the office 2-6 weeks after the surgery.
Complications
The most concerning complications of DCR include CSF leakage, recurrence, bleeding, infection, silicone tube extrusion, or scar. Fistula may rarely occur in the setting of GPA when external DCR is performed.
Prognosis
In general, external DCR has success rates from 90-95% for some entities of SANDO. EndoDCR rates may be approaching these rates as well. DCR success rates may be lower in cases with obstruction caused by radiation, PGA, sarcoidosis, radioactive iodine, chronic severe sinusitis, trauma, and revision surgeries. The most common cause of DCR failure is cicatricial closure of the ostium. Revision DCR success rates are not as high as primary DCR, but some authors report success rates as high as 86.9% with endoscopic revisions.
References
- ↑ Sobel RK, Aakalu VK, Wladis EJ, Bilyk JR, Yen MT, Mawn LA. A Comparison of Endonasal Dacryocystorhinostomy and External Dacryocystorhinostomy: A Report by the American Academy of Ophthalmology. Ophthalmology. 2019 Nov;126(11):1580-1585. doi: 10.1016/j.ophtha.2019.06.009. Epub 2019 Jul 26. PMID: 31358391.
- Akcay, E., Yuksel, N. & Ozen, U. Revision External Dacryocystorhinostomy Results After a Failed Dacryocystorhinostomy Surgery. Ophthalmol Ther 5, 75–80 (2016).
- Ali, M. J., Gupta, H., Honavar, S. G. & Naik, M. N. Acquired nasolacrimal duct obstructions secondary to naso-orbito-ethmoidal fractures: patterns and outcomes. Ophthal Plast Reconstr Surg 28, 242–245 (2012).
- Ali, M. J., Joshi, S. D., Naik, M. N. & Honavar, S. G. Clinical profile and management outcome of acute dacryocystitis: two decades of experience in a tertiary eye care center. Semin Ophthalmol 30, 118–123 (2015).
- Ali, M. J. & Naik, M. N. Image-Guided Dacryolocalization (IGDL) in Traumatic Secondary Acquired Lacrimal drainage Obstructions (SALDO). Ophthal Plast Reconstr Surg 31, 406–409 (2015).
- Ali, M. J., Psaltis, A. J. & Wormald, P. J. Long-term outcomes in revision powered endoscopic dacryocystorhinostomy. Int Forum Allergy Rhinol 4, 1016–1019 (2014).
- Al-Qahtani, K. H. et al. Nasolacrimal duct obstruction following radioactive iodine 131 therapy in differentiated thyroid cancers: review of 19 cases. Clin Ophthalmol 8, 2479–2484 (2014).
- Amadi, A. J. Endoscopic DCR vs External DCR: What’s Best in the Acute Setting? J Ophthalmic Vis Res 12, 251–253 (2017).
- Bartley, G. B. Acquired lacrimal drainage obstruction: an etiologic classification system, case reports, and a review of the literature. Part 1. Ophthal Plast Reconstr Surg 8, 237–242 (1992).
- Bartley, G. B. Acquired lacrimal drainage obstruction: an etiologic classification system, case reports, and a review of the literature. Part 2. Ophthal Plast Reconstr Surg 8, 243–249 (1992).
- Bartley, G. B. Acquired lacrimal drainage obstruction: an etiologic classification system, case reports, and a review of the literature. Part 3. Ophthal Plast Reconstr Surg 9, 11–26 (1993).
- Ben Simon, G. J. et al. External versus endoscopic dacryocystorhinostomy for acquired nasolacrimal duct obstruction in a tertiary referral center. Ophthalmology 112, 1463–1468 (2005).
- Ghanem, R. C., Chang, N., Aoki, L., Santo, R. M. & Matayoshi, S. Vasculitis of the lacrimal sac wall in Wegener granulomatosis. Ophthal Plast Reconstr Surg 20, 254–257 (2004).
- Kamal, S. & Ali, M. J. Primary Acquired Nasolacrimal Duct Obstruction (PANDO) and Secondary Acquired Lacrimal Duct Obstructions (SALDO). in Principles and Practice of Lacrimal Surgery (ed. Ali, M. J.) 163–171 (Springer Singapore, 2018).
- Mills, D. M. & Meyer, D. R. Acquired nasolacrimal duct obstruction. Otolaryngol. Clin. North Am. 39, 979–999, vii (2006).
- Pakrou, N., Selva, D. & Leibovitch, I. Wegener’s granulomatosis: ophthalmic manifestations and management. Semin. Arthritis Rheum. 35, 284–292 (2006).
- Paulsen, F. P., Thale, A. B., Hallmann, U. J., Schaudig, U. & Tillmann, B. N. The cavernous body of the human efferent tear ducts: function in tear outflow mechanism. Invest. Ophthalmol. Vis. Sci. 41, 965–970 (2000).
- Paulsen, F. P., Thale, A. B., Maune, S. & Tillmann, B. N. New insights into the pathophysiology of primary acquired dacryostenosis. Ophthalmology 108, 2329–2336 (2001).
- Pujari, A. & Ali, M. J. Infections of the Lacrimal Drainage System. in Principles and Practice of Lacrimal Surgery 179–188 (Springer, Singapore, 2018).
- Sibley, D., Norris, J. H. & Malhotra, R. Management and outcomes of patients with epiphora referred to a specialist ophthalmic plastic unit. Clin. Experiment. Ophthalmol. 41, 231–238 (2013).
- Sobel RK, Aakalu VK, Wladis EJ, Bilyk JR, Yen MT, Mawn LA. A Comparison of Endonasal Dacryocystorhinostomy and External Dacryocystorhinostomy: A Report by the American Academy of Ophthalmology. Ophthalmology. 2019 Nov;126(11):1580-1585. doi: 10.1016/j.ophtha.2019.06.009. Epub 2019 Jul 26. PMID: 31358391.
- Sundar, G. Lacrimal Trauma and Its Management. in Principles and Practice of Lacrimal Surgery 379–394 (Springer, Singapore, 2018).
- Sundar, G. Tumors of the Lacrimal Drainage System. in Principles and Practice of Lacrimal Surgery 417–428 (Springer, Singapore, 2018).
- Sweeney, A. R., Davis, G. E., Chang, S.-H. & Amadi, A. J. Outcomes of Endoscopic Dacryocystorhinostomy in Secondary Acquired Nasolacrimal Duct Obstruction: A Case-Control Study. Ophthal Plast Reconstr Surg 34, 20–25 (2018).
- Themes, U. F. O. Learning from a Difficult Case: Accessing the Anterolateral Maxillary Sinus. Ento Key (2016).
- Woo, T. L. et al. Australasian orbital and adnexal Wegener’s granulomatosis. Ophthalmology 108, 1535–1543 (2001).
- Woog, J. J. The incidence of symptomatic acquired lacrimal outflow obstruction among residents of Olmsted County, Minnesota, 1976-2000 (an American Ophthalmological Society thesis). Trans Am Ophthalmol Soc 105, 649–666 (2007).
- Yeatts, R. P. Current concepts in lacrimal drainage surgery. Curr Opin Ophthalmol 7, 43–47 (1996).
- Traumatic nasolacrimal duct obstruction: Clinical profile, management, and outcome. ResearchGate Available at: https://www.researchgate.net/publication/236068313_Traumatic_nasolacrimal_duct_ obstruction_Clinical_profile_management_and_outcome. (Accessed: 20th April 2018)