Atopic Keratoconjunctivitis
All content on Eyewiki is protected by copyright law and the Terms of Service. This content may not be reproduced, copied, or put into any artificial intelligence program, including large language and generative AI models, without permission from the Academy.
Atopic Keratoconjunctivitis
Disease Entity
Atopic keratoconjunctivitis (AKC) is a chronic inflammatory disease of eye that affects patients with atopic dermatitis (AD). The severity of the patient's AD does not affect the presentation or development of AKC.[1] A recent study found that AKC patients, especially children, showed broader polysensitization—often to food and pet allergens—linking AKC more closely to systemic atopic conditions like atopic dermatitis and asthma.[2]
Disease
AKC was first described in 1952 by Hogan [3], who described five cases of atopic eczema associated with bilateral keratoconjunctivitis. Peak incidence is between 30-50 years old.[4] It is characterized by bilateral, non-infectious inflammation of the conjunctiva and can present with ocular complications such as cataracts, blepharitis, keratoconus, cornea scarring, and even cornea perforation.
Epidemiology
Atopic dermatitis is a common chronic inflammatory skin disease that affects up to 15-20% of children and 1-3% of adults worldwide that presents as acute exacerbations of eczematous pruritic lesions of the skin[4]. It is estimated that 25-40% of patients with atopic dermatitis suffer from AKC [1].
Etiology
AKC is the chronic ocular surface inflammation suffered by patients with atopic dermatitis.
Pathophysiology
The pathophysiology is incompletely understood, but likely represents a combination genetic predisposition and immune dysregulation involving type I (IgE mediated) and type IV delayed hypersensitivity reactions. The pathophysiology of AKC involves the release of cytokines by mast cells, eosinophils, T cells and conjunctival epithelial cells.[5] The release of allergic mediators leads to activation of T helper cells and chronic mast cell degranulation.[6] AKC patients have also been shown to have decreased corneal sensitivity and conjunctival goblet cell density compared to controls.[7] The tears of patients who suffer from AKC have been shown to have elevated inflammatory cytokines and chemokines.[8]
Risk Factors
AKC is a multifactorial disease with genetic and environmental risk factors. The strongest associations are genetic predisposition for poor skin barrier function and dysregulation of the immune system, asthma, allergic rhinitis, and environmental allergens. The presence of food allergies and sensitivities in infancy and childhood is associated with a more severe form of atopic dermatitis.[9] Allergy and sensitivity to pollen, pets and dust mites have also been associated with AKC.
Environmental factors such as climate, diet, urban living (including pollution exposure), tobacco smoke, duration of breastfeeding during infancy, and obesity have been suggested as risk factors for AKC.[9] It has also been proposed that antibiotic use may increase the risk of developing atopic disease via alterations to the gut microbiome.[10]
Dupilumab, an IL-4 receptor alpha (IL-4Rα) antibody used to treat atopic dermatitis (AD), has been linked to worsening eye symptoms resembling AKC in some patients—including conjunctival hyperemia and superficial punctate keratitis[11]. Although AKC is associated with AD, a 2020 meta-analysis of 3,303 patients found that 26.1% of those treated with dupilumab developed conjunctivitis, despite its effectiveness in managing skin symptoms[11]. In contrast, upadacitinib, a novel alternative treatment, may reduce ocular surface inflammation by selectively inhibiting Janus kinases, bypassing IL-4/13 feedback and potentially offering better control of both skin and eye symptoms in AD patients.[11]
General Pathology
Histology of AKC typically shows proliferation of goblet cells, invasion of eosinophils and mast cells into the epithelium as well as mononuclear cell infiltrate in the substantia propria.[8] Conjunctival epithelium of AKC patients shows increased levels of T cells, T-helper cells, macrophages and dendritic cells.[12]
Primary Prevention
Primary prevention involves reducing the probability of disease in patients that do not have clinically evident disease. Breastfeeding has shown to be a protective factor, particular before 3 months of age.[13] Additionally, prenatal and postnatal probiotics could reduce the incidence of atopic disease by as much as 30%.[14] Dietary supplementation with vitamins and minerals has been studied, but has not shown any benefit.
Diagnosis
The diagnosis of atopic keratoconjunctivitis is largely clinical, relying on history and exam findings.
History
A family history of allergies, asthma, urticaria, and/or hay fever is often present. Typically, patients have atopic dermatitis or eczema since childhood, with ocular symptoms developing later in life. Mean onset of ocular symptoms is 38 years.[15] AKC can present with seasonal exacerbations, but symptoms are often chronic and persistent throughout the year.[4]
Symptoms
The chief symptom of AKC is intense, bilateral itching of the conjunctiva, eyelids, and periorbital skin. Tearing, burning, photophobia and blurry vision are commonly encountered symptoms. Patients with AKC can have copious mucoid discharge, often described as rope-like.[15]
Physical Examination
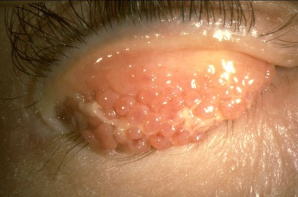
- Eyelids and periorbita: The eyelids and periorbital skin will almost always show evidence of eczematous dermatitis (erythematous, thickened dry skin with blistered patches). Eyelid findings include tylosis (thickening of the tarsal border of the eyelid) with crusting and scaling.[16] Meibomian gland disease is very common in patients with AKC. Other lid findings often encountered are ectropion, trichiasis and madarosis.
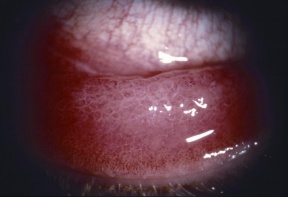
- Conjunctiva: The conjunctiva is hyperemic and edematous, with prominent tarsal papillae (papillary conjunctivitis). Mucoid discharge is often present. Severe conjunctival disease can result in scarring and symblepharon. Horner-Trantas dots may or may not be present.
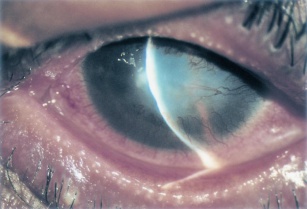
- Cornea: Corneal involvement is frequent and can range from punctate epithelial erosions to ulcers and even perforation. Peripheral vascularization and pannus are common.[1]
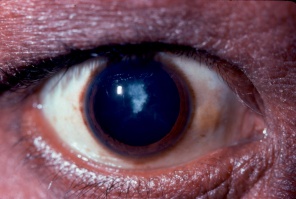
- Anterior Segment: Anterior and posterior subcapsular cataracts are common.
Ancillary Testing
Clinical diagnosis is based mainly on history and exam findings, though adjunctive diagnostic procedures and lab tests can aid in diagnosis and assessing severity of disease. Many of these tests require equipment not readily available to most ophthalmologists.
Serum IgE and skin prick testing is non-specific, but can be helpful in establishing that a patient has atopic disease.[17] IgE levels in tears have been shown to correlate with clinical severity of allergic eye disease.[18]
Brush cytology, which involves taking scrapings from the tarsal conjunctiva, can quantify levels of inflammatory cells such as eosinophils and neutrophils. It has been show to correlate with the amount of corneal damage.[19] Confocal scanning laser microscopy has been described as a less invasive means of evaluating the ocular surface of AKC patients by identification of inflammatory cell densities.[20] A significant correlation was found between the extent of inflammation as determined via brush cytology and that found by in vivo confocal microscopy.[21]
Conjunctival biopsy is rarely used to assist in diagnosing AKC. As above, on pathology, mast cell infiltration of the conjunctival epithelium can be seen.
A recent review article by Wang et. al. emphasizes that combining corneal morphological and biomechanical assessments improves the detection of early or subclinical keratoconus, which is especially important in patients with AKC[22]. These patients are more prone to corneal ectasia due to chronic inflammation and eye rubbing, which can compromise corneal integrity. The study supports the use of tools like the Pentacam and Corvis ST to better screen for biomechanical instability preoperatively in individuals with allergic predispositions[22]. For AKC patients, this dual approach helps ensure safer outcomes in refractive surgery by identifying subtle corneal abnormalities that may be missed by morphology alone[22].
Differential Diagnosis
Clinically, AKC is very similar to vernal keratoconjunctivitis (VKC), and there is controversy regarding classification of the two diseases. It has been proposed that VKC is a childhood form of AKC that evolves into AKC in adulthood. However, VKC can be present without signs of atopic disease. VKC is generally regarded to end at puberty, and disease severity can fluctuate with seasons. Giant papillary reaction is more common in VKC. Atopic disease is typically a year-round condition. Though AKC peaks in adults, it can be present in childhood.
Differential Diagnosis of AKC
- Vernal keratoconjunctivitis
- Seasonal allergic conjunctivitis
- Giant papillary conjunctivitis
- Phlyctenular keratoconjunctivitis
- Toxic conjunctivitis
- Infections conjunctivitis (esp. trachoma)
- Ocular rosacea
Management
The purpose of treatment in AKC is to improve symptoms, limit exacerbations, and prevent complications/sequelae that lead to vision loss. It is also important to balance this with limiting side effects of treatment.
General Treatment
Involvement of other specialists such as dermatologists and allergists is essential for successful management. Attention to limiting environmental factors that exacerbate disease. This can include use of hypoallergenic bedding, elimination of pets, when necessary, use of air filtering devices, and dietary changes in cases of food sensitivity.
Medical Therapy
Topical Therapy: Mild disease can be treated similarly to other allergic eye disease, with emphasis placed on hand hygiene, cold compresses, and topical mast cells stabilizers (olopatadine 0.1% or lodoxamide 0.1%) and antihistamines (azelastine 0.05%). While almost all patients will require more intensive treatment, these are vital to efficacious treatment. Corticosteroid drops and ointments are commonly employed in the treatment of AKC, especially to achieve control of exacerbations and breakthrough inflammation. Steroid therapy should be tapered as quickly as possible while avoiding rebound inflammation. Calcineurin inhibitors (cyclosporine and tacrolimus) are an effective steroid-sparing therapy. Calcineurin is an enzyme involved in the activation of T cells, a key player in AKC. Tacrolimus is available as a 0.03% and 0.1% topical ointment for dermatologic use (Protopic). It has been used off- label for treatment of ophthalmic diseases.[23] Topical cyclosporine is available commercially in a 0.05% eye drop preparation (Restasis). Topical calcineurin inhibitors are safe and generally well tolerated, with stinging and eyelid skin maceration being the most common side effects.[24] A recent meta- analysis demonstrated the efficacy of topical immunomodulators in treating AKC.[25]
Systemic Medications: Systemic antihistamines are routinely used in atopic disease. Oral steroids and cyclosporine are generally reserved for severe/recalcitrant disease or for treatment of dermatologic disease and are often employed in conjunction with the patient’s dermatologist. Prolonged treatment with oral steroids is avoided due to unacceptable side effects. Systemic cyclosporine, given at dose of 5mg/kg per day has been shown helpful in inducing remission of severe atopic disease.[26][27] Once remission is obtained, frequency can be decreased to as little as every 5th day. Patients on systemic cyclosporine need regular monitoring of renal function, liver function, blood counts, and blood pressure.
Additional Therapy: Zaouali et al reported improvement of symptoms and clinical features of AKC with injection of supratarsal triamcinolone acetonide.[28] Punctal occlusion can useful for concomitant dry eye.
Medical follow up
Patients with AKC require close, long- term follow up for development of vision threatening complications and medication side effects such as cataract formation and intraocular pressure elevation associated with steroid usage.
Surgery
Amniotic membrane transplantation has been shown to be very effective for persistent corneal epithelial defects.[29]
Tectonic keratoplasty may be necessary for patients with severe corneal thinning or perforations. Penetrating keratoplasty (PK) may be indicated for corneal opacities or severe ulceration and thinning.
Patients with AKC are at increased risk of cataract formation (especially anterior and posterior subcapsular), independent of steroid exposure. Postoperative care and final visual outcomes are impacted by ongoing ocular surface disease.
Eyelid surgery may be necessary for correction of trichiasis, ectropion or entropion.[15]
Surgical Follow Up
PK in AKC patients is associated with higher rate of postoperative complications such as graft rejection, cataracts and glaucoma.[29] Additionally, PK is not curative for AKC; these patients continue to have severe ocular surface disease that can affect viability of the corneal graft. Despite these complications, PK can provide significant improvements in visual acuity. One study looking at long term success of PK in AKC found a 50% failure rate in primary PK, with the primary causes of graft failure being rejection and stromal ulceration.[15]
Complications
The clinical presentation of AKC can vary greatly, but in recalcitrant cases, it can cause severe and site threatening corneal complications. Corneal findings range from punctate epithelial erosions to ulcers (infectious or sterile), neovascularization, and lipid keratopathy. Herpes simplex keratitis is more common in patients with AKC and often more severe.[15] Patients with AKC are also more likely to get bilateral herpetic disease, which is relatively unusual in the absence of atopy.[30][31] As with other ocular allergic diseases, patients with AKC are at increased risk of keratoconus due to chronic eye rubbing. Therefore, patients with AKC should be counseled to limit eye rubbing as much as possible and should be provided with treatment to reduce or eliminate itch. Due to common use of topical and oral corticosteroids, glaucoma is a potential complication of treatment. There are various case reports of conjunctival squamous cell carcinoma in patients with AKC.[32][33]
References
- ↑ Jump up to: 1.0 1.1 1.2 Guglielmetti S, Dart JK, Calder V. Atopic keratoconjunctivitis and atopic dermatitis. Curr Opin Allergy Clin Immunol. 2010;10(5):478-485.
- ↑ Component Sensitization Profile in Children and Adolescents with Ocular Allergy in Brazil Marino, Luiza et al. Journal of Allergy and Clinical Immunology, Volume 155, Issue 2, AB155
- ↑ Hogan MJ. Atopic keratoconjunctivitis. Am J Ophthalmol 1953; 36:937-47
- ↑ Jump up to: 4.0 4.1 4.2 Bielory B, Bielory L. Atopic dermatitis and keratoconjunctivitis. Immunol Allergy Clin North Am. 2010;30(3):323-336.
- ↑ Enríquez-de-Salamanca A, Calder V, Gao J, et al. Cytokine responses by conjunctival epithelial cells: an in vitro model of ocular inflammation. Cytokine. 2008;44(1):160-167.
- ↑ Metz DP, Hingorani M, Calder VL, Buckley RJ, Lightman SL. T-cell cytokines in chronic allergic eye disease. J Allergy Clin Immunol. 1997;100(6 Pt 1):817-824.
- ↑ Hu Y, Adan ES, Matsumoto Y, et al. Conjunctiva in vivo confocal scanning laser microscopy in patients with atopic keratoconjunctivitis. Mol Vis. 2007;13:1379-1389.
- ↑ Jump up to: 8.0 8.1 Chen, J.J., Applebaum, D.S., Sun, G.S. & Pflugfelder, S.C. Atopic keratoconjunctivitis: A review. J Am Acad Dermatol 70, 569-575 (2014).
- ↑ Jump up to: 9.0 9.1 Nutten S. Atopic dermatitis: global epidemiology and risk factors. Ann Nutr Metab. 2015;66 Suppl 1:8-16.
- ↑ Dom S, Droste JHJ, Sariachvili MA, et al. Pre and post-natal exposure to antibiotics and the development of eczema, recurrent wheezing and atopic sensitization in children up to the age of 4 years. Clin Exp Allergy. 2010;40(9):1378-1387.
- ↑ Jump up to: 11.0 11.1 11.2 Galluzzo M, Tofani L, Spelta S, Talamonti M, Micera A, Bianchi L, Coassin M, Bonini S, Di Zazzo A. Dupilumab-associated ocular surface disease or atopic keratoconjunctivitis not improved by dupilumab? Upadacitinib may clarify the dilemma: A case report. Skin Health Dis. 2024 Mar 15;4(3):e354. doi: 10.1002/ski2.354. PMID: 38846697; PMCID: PMC11150752.
- ↑ Foster CS, Rice BA, Dutt JE. Immunopathology of atopic keratoconjunctivitis. Ophthalmology. 1991;98(8):1190-1196.
- ↑ Flohr C, Nagel G, Weinmayr G, et al. Lack of evidence for a protective effect of prolonged breastfeeding on childhood eczema: lessons from the International Study of Asthma and Allergies in Childhood (ISAAC) Phase Two. Br J Dermatol. 2011;165(6):1280-1289.
- ↑ Fiocchi A, Burks W, Bahna SL, et al. Clinical Use of Probiotics in Pediatric Allergy (CUPPA): A World Allergy Organization Position Paper. World Allergy Organ J. 2012;5(11):148-167.
- ↑ Jump up to: 15.0 15.1 15.2 15.3 15.4 Power WJ, Tugal-Tutkun I, Foster CS. Long-term follow-up of patients with atopic keratoconjunctivitis. Ophthalmology. 1998;105(4):637-642.
- ↑ . Brémond-Gignac D, Nischal KK, Mortemousque B, Gajdosova E, Granet DB, Chiambaretta F. Atopic Keratoconjunctivitis in Children: Clinical Features and Diagnosis. Ophthalmology. 2016;123(2):435-437.
- ↑ Correale CE, Walker C, Murphy L, Craig TJ. Atopic Dermatitis: A Review of Diagnosis and Treatment. AFP. 1999;60(4):1191.
- ↑ Inada N, Shoji J, Kato H, Kiely S, Mulyanto null, Sawa M. Clinical evaluation of total IgE in tears of patients with allergic conjunctivitis disease using a novel application of the immunochromatography method. Allergol Int. 2009;58(4):585-589.
- ↑ Takano Y, Fukagawa K, Dogru M, Asano-Kato N, Tsubota K, Fujishima H. Inflammatory cells in brush cytology samples correlate with the severity of corneal lesions in atopic keratoconjunctivitis. British Journal of Ophthalmology. 2004;88(12):1504-1505.
- ↑ Hu Y, Adan ES, Matsumoto Y, et al. Conjunctival in vivo confocal scanning laser microscopy in patients with atopic keratoconjunctivitis. Mol Vis. 2007;13:1379-1389.
- ↑ Wakamatsu TH, Tanaka M, Satake Y, et al. Eosinophil cationic protein as a marker for assessing the efficacy of tacrolimus ophthalmic solution in the treatment of atopic keratoconjunctivitis. Mol Vis. 2011;17:932-938.
- ↑ Jump up to: 22.0 22.1 22.2 Wang, Q., Deng, Y., Li, S. et al. Corneal biomechanical changes in allergic conjunctivitis. Eye and Vis 8, 17 (2021). https://doi.org/10.1186/s40662-021-00241-7
- ↑ Al-Amri AM. Long-term follow-up of tacrolimus ointment for treatment of atopic keratoconjunctivitis. Am J Ophthalmol. 2014;157(2):280-286.
- ↑ González-López JJ, López-Alcalde J, Morcillo Laiz R, Fernández Buenaga R, Rebolleda Fernández G. Topical cyclosporine for atopic keratoconjunctivitis. Cochrane Database Syst Rev. 2012;(9):CD009078.
- ↑ Erdinest N, Noyman DBE, London N, et al. Applications of topical immunomodulators enhance clinical signs of vernal keratoconjunctivitis (VKC) and atopic keratoconjunctivitis (AKC): a meta-analysis. Int Ophthalmol. Mar 24 2024;44(1):157. doi:10.1007/s10792-024-03097-7
- ↑ Munro CS, Levell NJ, Shuster S, Friedmann PS. Maintenance treatment with cyclosporin in atopic eczema. Br J Dermatol. 1994;130(3):376-380.
- ↑ Salek MS, Finlay AY, Luscombe DK, et al. Cyclosporin greatly improves the quality of life of adults with severe atopic dermatitis. A randomized, double-blind, placebo-controlled trial. Br J Dermatol. 1993;129(4):422-430.
- ↑ Zaouali S, Kahloun R, Attia S, et al. Supratarsal injection of triamcinolone acetonide and childhood allergic keratoconjunctivitis. Int Ophthalmol. 2012;32(2):99-106.
- ↑ Jump up to: 29.0 29.1 Koçluk Y, Yalniz-Akkaya Z, Burcu A, Örnek F. Atopic keratoconjunctivitis: long-term results of medical treatment and penetrating keratoplasty. Arq Bras Oftalmol. 2016;79(6):376-379.
- ↑ Garrity JA, Liesegang TJ. Ocular complications of atopic dermatitis. Can J Ophthalmol. 1984;19(1):21-24.
- ↑ Easty D, Entwistle C, Funk A, Witcher J. Herpes simplex keratitis and keratoconus in the atopic patient. A clinical and immunological study. Trans Ophthalmol Soc U K. 1975;95(2):267-276.
- ↑ Kallen C, Reinhard T, Schilgen G, et al. [Atopic keratoconjunctivitis: probably a risk factor for the development of conjuntival carcinoma]. Ophthalmologe.
- ↑ Heinz C, Fanihagh F, Steuhl K-P. Squamous cell carcinoma of the conjunctiva in patients with atopic eczema. Cornea. 2003;22(2):135-137.