Indocyanine Green Angiography: Difference between revisions
Evan.Wotipka (talk | contribs) (Added more images) |
Evan.Wotipka (talk | contribs) No edit summary |
||
| Line 1: | Line 1: | ||
{{Article | {{Article | ||
|Authors=Evan.Wotipka, Talhah.Zubair | |Authors=Evan.Wotipka, Talhah.Zubair, Hossein Nazari | ||
|Additional contributors=Talhah.Zubair | |Additional contributors=Hossein Nazari, Talhah.Zubair | ||
|Category=Retina/Vitreous | |Category=Retina/Vitreous | ||
|Assigned editor=Vaidehi.Dedania | |Assigned editor=Vaidehi.Dedania | ||
Revision as of 14:17, November 26, 2023
All content on Eyewiki is protected by copyright law and the Terms of Service. This content may not be reproduced, copied, or put into any artificial intelligence program, including large language and generative AI models, without permission from the Academy.
Indocyanine green angiography (ICGA) provides improved imaging of choroidal vasculature compared to fluorescein angiography. Because of the excitation and emission properties of indocyanine green (excitation at 790 nm and emission at 835 nm) pathologies involving retinal and choroidal vascular system can be imaged even in the presence of sub-retinal or sub-retinal pigment epithelium (RPE) fluid, hemorrhage, or pigment that may obscure the view to the retina or choroid.
History
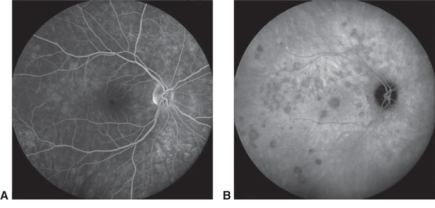
Indocyanine green (ICG) was developed in Kodack laboratories, and its physical and physiological properties were first described by Fox and Wood in 1960.[1] Kogure and others first used ICG to visualize the fundus of an owl monkey in the 1970s.[2] However, it was not routinely used in humans until 1990s because of technological limitations of the fundus cameras. There was an uptick in ICGA administration in the 1990s due to improvements in digital video angiography, scanning laser ophthalmoscopy, and optical systems of fundus cameras.[3][4] In addition, the advent of ICGA allowed for better detection of occult choroidal neovascularization (CNV), which in turn led to an increase in the number of eyes that were eligible for photocoagulation which, at the time, was the only treatment option for CNV.[5] Since 2000s, after anti-vascular endothelial growth factor (anti-VEGF) was shown to improve visual acuity in patients with CNVs, laser photocoagulation of the CNV lesions was mostly abandoned and ICGA administration was diminished mostly to the indications summarized below.[6]
Properties
Two properties make ICG an effective dye for visualizing choroidal vasculature. First, indocyanine green affinity to circulating proteins is high (95%) which limits ICG leakage from vessel walls. Comparatively, more heavily leaking fluorescein (only 80% bound) from the choriocapillaris can obscure the details of the underlying choroidal vasculature. Another drawback of fluorescein dye in visualizing the choroid is that fluorescein molecule absorbs and emits shorter wavelength photons. Since the retinal pigment epithelium (RPE) also absorbs and emits photons around this wavelength, the resulting scatter from RPE can obscure the choroid. Indocyanine, however, absorbs and emits photons in the infrared spectrum, allowing the viewer to see the choroid through the retinal pigment epithelium or through disease processes such as overlying hemorrhage.[7]
See “Dyes in Ophthalmology” for more information.
Uses
For a thorough review of the uses of indocyanine green see Cohen et al.’s “Is indocyanine green still relevant?” editorial in Retina 2011.[8]
Age-Related Macular Degeneration (ARMD)
Although the localization of CNV is no longer crucial for managing exudative ARMD as it was in the past, ICGA is still utilized when there is doubt in the diagnosis.[8] If examination, FA, and OCT cannot exclude conditions such as central serous chorioretinopathy or idiopathic polypoidal choroidal vasculopathy, ICGA can provide additional objective information to differentiate these entities from CNV.[8]
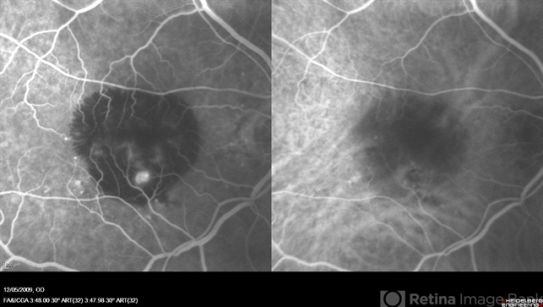
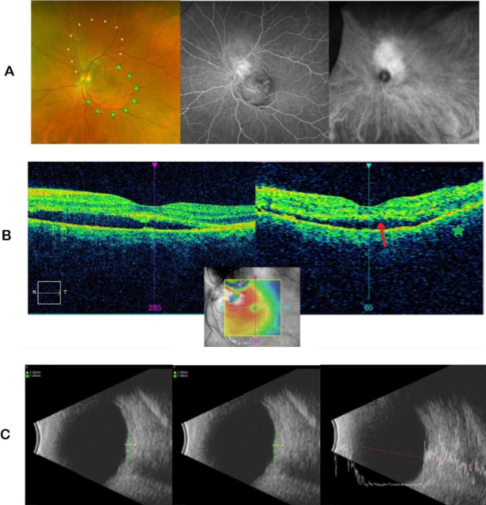
Type 1 macular neovascularization (MNV) with CNV located under the RPE is the most common form of exudative AMD. Often, clinical examination, OCT, and FA are sufficient for the diagnosis and follow up, however, ICGA may be required when the lesion is covered with hemorrhage or if there is a question about the presence of accompanying type 3 MNV (retinal angiomatous proliferation – RAP) that is shown to respond well to early anti-VEGF treatment and has a higher risk of second eye involvement.
Polypoidal Choroidal Vasculopathy (PCV)
Aneurysmal type 1 MNV or polypoid choroidal vasculopathy (PCV) is characterized by a growth of cavernous thin-walled vessels between the RPE and the Bruch’s membrane and is often associated with recurrent, serosanguineous detachments of the RPE and neurosensory retina secondary to leakage and bleeding from the polypoid neovascular membranes. ICGA, that may delineate the polypoid or “bulging” capillaries better, will be helpful with accurate diagnosis and giving a better insight to the possible clinical course and prognosis. In this setting, ICGA can guide management primarily in three ways:
1. ICG can help reveal the polypoidal lesions and the branching vascular networks.
2. It can help identify active PCV for selective treatment with photodynamic therapy (PDT).
3. It can help identify any recurrences after PDT (which is seen as late geographic hypercyanescence).[9][10][11]
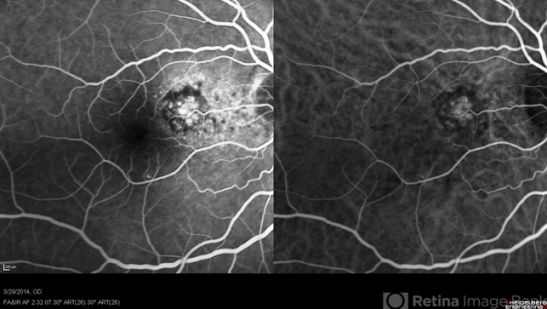
Choroidal tumors
ICGA and ultrasonography help identify choroidal hemangiomas and differentiate them from simulating tumors such as choroidal melanoma or metastasis. Choroidal hemangioma has a very distinctive filling pattern on ICGA: there is a progressive filling of abnormal choroidal vessels in the early phase, followed by very intense hypercyanescence at 2-4 minutes, with decreased cyanescence of the tumor at later frames compared to the rest of the choroid, known as the washout phenomenon.[12][13][14][15]
[16] Although other tumors of the choroid do not have a characteristic ICGA feature, ICGA may still be used to differentiate them from simulating pathologies such as peripheral exudative hemorrhagic chorioretinopathy (PEHCR). [8]
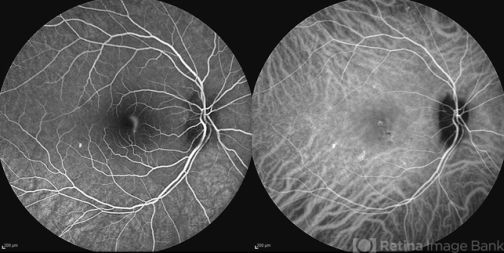
Central Serous Chorioretinopathy (CSCR)
Choroidal hypervascularity in the pachychoroid spectrum of diseases such as central serous chorioretinopathy (CSCR) may be better delineated with ICGA. ICGA is especially helpful in guiding photodynamic therapy to include hypervascular areas of the choroid.[17][18]
Pathologic Myopia
ICGA may provide a clear view of the complications associated with pathologic myopia. CNV inside areas with lacquer cracks or underneath subretinal hemorrhages related to newly formed lacquer cracks are better visualized with ICGA.[19]
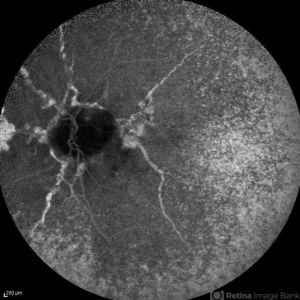
Angioid Streaks
Angioid streaks are typically visualized more clearly, extensively, and prominently with ICGA as compared to FA or fundus exam.[20]
Inflammatory
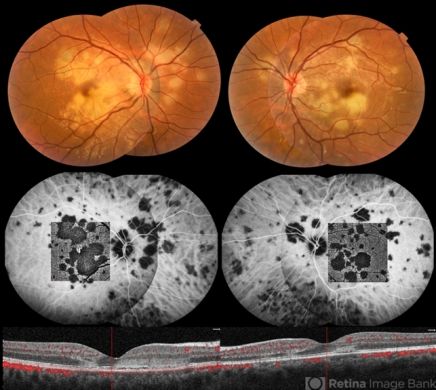
Multiple evanescent white dot syndrome (MEWDS)
ICGA is helpful in the diagnosis of white dot syndromes, particularly if the white retinal dots are of an atypical appearance or have already faded away.[21] In MEWDS, ICGA shows numerous hypercyanascent dots scattered throughout the posterior pole and hypocyanascent area surrounding the optic nerve. The hypocyanascent area on ICGA disappears over time as the inflammation resolves. [22][23][24]
Acute Posterior Multifocal Placoid Pigment Epitheliopathy
APMPPE is a multifocal inflammation involving the choriocapillaris, and the resulting delayed and defective choroidal filling may manifest as a unique pattern of multifocal hypocyanascence in areas seen in the retinal examination as white spots.[25]
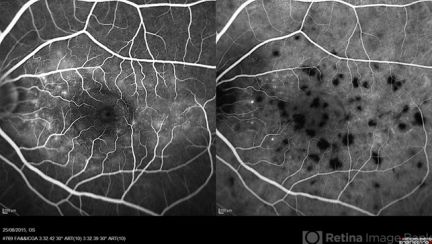
Vogt-Koyanagi-Harada disease
Although ICGA is not crucial for diagnosing VKH, active disease can be differentiated from completely treated and inactive VKH by the ICGA pattern. ICGA signs of acute VKH include, early choroidal vessel hypercyanescence, intermediate to late phase fuzziness of choroidal stromal vessels, disc hypercyanescence, and hypocyanescent dark dots (HDDs). ICGA is an excellent modality to detect subtle choroidal inflammation in subclinical VKH reactivation, thus helping to monitor adequate response to corticosteroid treatment.[26][27]
Birdshot Chorioretinopathy
Deep, cream-colored lesions diffusely scattered throughout the fundus in Birdshot chorioretinopathy (BS) appear in ICGA as round or oval hypocyanascent spots with a similar size spread across the fundus, particularly affecting the nasal quadrant. FA does not show these lesions well and ICGA often shows a more spots compared to clinical examination. Other ICGA features include an alteration of the vascular pattern of the choroid, with choroidal vessels appearing fuzzy and indistinct in the intermediate phases of the angiogram and a late diffuse hypercyanascence resulting from the ICG dye leaking through the inflamed choroidal vessels. In the chronic phase of the disease the hypocyanescent dots persist in the late phases of the angiogram and correspond to chorioretinal scars. [28]
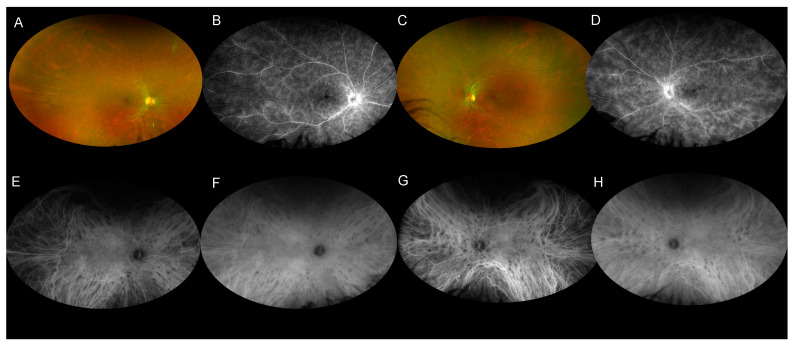
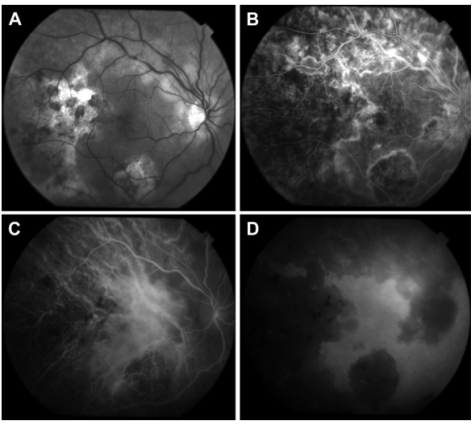
Choroidal Granulomas
ICGA can reveal choroidal involvement and help with disease classification for monitoring sarcoidosis.[29][30]
Serpiginous Choroidopathy
ICGA can help to better differentiate between active and healed lesions.[31][32][33]In new active lesions ICGA will typically show two patterns: 1) early and late hypoautocyanescence with ill-defined margins, or 2) early hypoautocyanescence with increased cyanescence toward late-phase ICGA. Choroiditis reactivation will present similarly with lesions usually at the margin of healed lesions. ICGA in healed serpiginous choroidopathy will show early and late-phase hypocyanescence with well-defined margins.
Ocular Infections
While posterior inflammation involving the choroid could be seen with ICGA in many infectious processes, ICGA often does not offer additional information to help with the diagnosis or management of ocular infectious conditions.[8]
Trauma
ICGA can reveal choroidal involvement as with ocular traumatic choroidopathy, choroidal rupture, and traumatic ocular hypotony, although it is rarely ordered in ocular trauma settings.[34][35] [36][37]
Procedure
ICG is injected at a loading dose of 25 mg , usually in a mixture of fluorescein and ICG. There are three temporal phases for ICG imaging, similar to fluorescein angiography.[7][38] The three phases are:
Early phase - 1 minute after injecting the eye, this is when larger choroidal arteries and veins are highlighted, as the dye is still making its way through the choriocapillaris.[38]
Middle phase - 5 to 15 minutes after injection, at this time the choroidal vasculature is now more diffuse and less distinct.[38]
Late phase - >15 minutes after dye injection, during this phase a hypercyanescent lesion is more visible against the slowly fading background.[38]
Safety
Toxicity and side effects from ICGA are extremely rare. An editorial described four reports of patients who have experienced an adverse reaction out of 240,000 indocyanine intravenous injections.[7][39] One patient had urticaria and three patients had anaphylactic reactions, with one resulting in death. Another study looked at 1923 patients with ICG angiograms; only one had an adverse reaction in the form of a decrease in blood pressure without anaphylaxis.[7][40]
Absolute contraindications
1. Prior allergic reaction to ICG
2. Iodine allergy (ICG is dissolved in an aqueous sodium iodide solvent).[38]
Relative contraindications
1. End stage renal disease
2. Liver disease
3. Pregnancy (Category C)[7]
Summary
ICGA is a safe and important imaging modality that offers a better view of the choroidal vasculature and related pathologies. Although no longer used as often as before in managing exudative AMD, ICGA is still helpful in guiding the treatment of PCV and CSC and diagnosing choroidal hemangiomas and ocular inflammations primarily affecting the choroid.
References
- ↑ Fox IJ, Wood EH. Indocyanine green: physical and physiologic properties. Proc Mayo Clin 1960; 35: 732-44.
- ↑ Kogure K, David NJ, Yamanouchi U, Chromokos E. Infrared absorption angiography of the fundus circulation. Arch Ophthalmol 1970; 83: 209-14.
- ↑ Hayashi K, Hasegawa Y, Tokoro T. Indocyanine green angiography of central serous chorioretinopathy. Int Ophthalmol 1986; 9: 37-41.
- ↑ Scheider A, Schroedel C. High resolution indocyanine green angiography with a scanning laser ophthalmoscope. Am J Ophthalmol 1989; 108: 458-9.
- ↑ Yannuzzi LA, Slakter JS, Sorenson JA, Guyer DR, Orlock DA. Digital indocyanine green videoangiography and choroidal neovascularization. Retina 1992; 12: 191-223.
- ↑ Rosenfeld PJ, Brown DM, Heier JS, et al. Ranibizumab for neovascular age-related macular degeneration. N Engl J Med 2006;355:1419–1431.
- ↑ Jump up to: 7.0 7.1 7.2 7.3 7.4 Owens SL. Indocyanine green angiography. Br J Ophthalmol. 1996;80(3):263-266. doi:10.1136/bjo.80.3.263
- ↑ Jump up to: 8.0 8.1 8.2 8.3 8.4 Cohen SY, Dubois L, Quentel G, Gaudric A. Is indocyanine green angiography still relevant?. Retina. 2011;31(2):209-221. doi:10.1097/IAE.0b013e31820a69db
- ↑ Rosen RB, Hathaway M, Rogers J, et al. Simultaneous OCT/SLO/ICG imaging. Invest Ophthalmol Vis Sci 2009;50: 851–860.
- ↑ Eandi CM, Ober MD, Freund KB, Slakter JS, Yannuzzi LA. Selective photodynamic therapy for neovascular age-related macular degeneration with polypoidal choroidal neovascularization. Retina 2007;27:825–831.
- ↑ Yamashiro K, Tsujikawa A, Nishida A, Mandai M, Kurimoto Y. Recurrence of polypoidal choroidal vasculopathy after photodynamic therapy. Jpn J Ophthalmol 2008;52:457–462.
- ↑ Shields CL, Shields JA, De Potter P. Patterns of indocyanine green videoangiography of choroidal tumours. Br J Ophthalmol 1995;79:237–245.
- ↑ Arevalo JF, Shields CL, Shields JA, Hykin PG, De Potter P. Circumscribed choroidal hemangioma: characteristic features with indocyanine green videoangiography. Ophthalmology 2000;107:344–350.
- ↑ Schalenbourg A, Piguet B, Zografos L. Indocyanine green angiographic findings in choroidal hemangiomas: a study of 75 cases. Ophthalmologica 2000;214:246–252.
- ↑ Piccolino FC, Borgia L, Zinicola E. Indocyanine green angiography of circumscribed choroidal hemangiomas. Retina 1996;16:19–28.
- ↑ Wen F, Wu D. Indocyanine green angiographic findings in diffuse choroidal hemangioma associated with Sturge-Weber syndrome. Graefes Arch Clin Exp Ophthalmol 2000;238: 625–627.
- ↑ Yannuzzi LA, Slakter JS, Gross NE, et al. Indocyanine green angiography-guided photodynamic therapy for treatment of chronic central serous chorioretinopathy: a pilot study. Retina 2003;23:288–298.
- ↑ Piccolino FC, Borgia L. Central serous chorioretinopathy and indocyanine green angiography. Retina 1994;14:231–242.
- ↑ Axer-Siegel R, Cotlear D, Priel E, Rosenblatt I, Snir M, Weinberger D. Indocyanine green angiography in high myopia. Ophthalmic Surg Lasers Imaging 2004;35:139–145.
- ↑ Quaranta M, Cohen SY, Krott R, Sterkers M, Soubrane G, Coscas GJ. Indocyanine green videoangiography of angioid streaks. Am J Ophthalmol 1995;119:136–142.
- ↑ Obana A, Kusumi M, Miki T. Indocyanine green angiographic aspects of multiple evanescent white dot syndrome. Retina 1996;16:97–104.
- ↑ Ie D, Glaser BM, Murphy RP, Gordon LW, Sjaarda RN, Thompson JT. Indocyanine green angiography in multiple evanescent white-dot syndrome. Am J Ophthalmol 1994;117:7–12.
- ↑ Pece A, Sadun F, Trabucchi G, Brancato R. Indocyanine green angiography in enlarged blind spot syndrome. Am J Ophthalmol 1998;126:604–607.
- ↑ Martinet V, Ducos de Lahitte G, Terrada C, Simon C, LeHoang P, Bodaghi B. Multiple evanescent white dot syndrome and acute idiopathic blind spot enlargement: angiographic and electrophysiologic findings [French]. J Fr Ophtalmol 2008;31:265–272.
- ↑ Howe LJ, Woon H, Graham EM, Fitzke F, Bhandari A, Marshall J. Choroidal hypoperfusion in acute posterior multifocal placoid pigment epitheliopathy. An indocyanine green angiography study. Ophthalmology 1995;102:790–798.
- ↑ Bouchenaki N, Herbort CP. The contribution of indocyanine green angiography to the appraisal and management of Vogt-Koyanagi-Harada disease. Ophthalmology 2001;108:54–64.
- ↑ Kawaguchi T, Horie S, Bouchenaki N, Ohno-Matsui K, Mochizuki M, Herbort CP. Suboptimal therapy controls clinically apparent disease but not subclinical progression of Vogt-Koyanagi-Harada disease. Int Ophthalmol 2010;30:41–50.
- ↑ Fardeau C, Herbort CP, Kullmann N, Quentel G, LeHoang P. Indocyanine green angiography in birdshot chorioretinopathy. Ophthalmology. 1999;106(10):1928-1934. doi:10.1016/S0161-6420(99)90403-7
- ↑ Akova YA, Kadayifcilar S, Aydin P. Assessment of choroidal involvement in sarcoidosis with indocyanine green angiography. Eye 1999;13:601–603.
- ↑ Wolfensberger TJ, Herbort CP. Indocyanine green angiographic features in ocular sarcoidosis. Ophthalmology 1999; 106:285–289
- ↑ Giovannini A, Mariotti C, Ripa E, Scassellati-Sforzolini B. Indocyanine green angiographic findings in serpiginous choroidopathy. Br J Ophthalmol 1996;80:536–540.
- ↑ Giovannini A, Ripa E, Scassellati-Sforzolini B, Ciardella A, Tom D, Yannuzzi L. Indocyanine green angiography in serpiginous choroidopathy. Eur J Ophthalmol 1996;6: 299–306.
- ↑ Nazari Khanamiri H, Rao NA. Serpiginous choroiditis and infectious multifocal serpiginoid choroiditis. Surv Ophthalmol. 2013;58(3):203-232. doi:10.1016/j.survophthal.2012.08.008
- ↑ Kohno T, Miki T, Hayashi K. Choroidopathy after blunt trauma to the eye: a fluorescein and indocyanine green angiographic study. Am J Ophthalmol 1998;126:248–260.
- ↑ Masaoka N, Sawada K, Komatsu T, Fukushima A, Ueno H. Indocyanine green angiographic findings in 3 patients with traumatic hypotony maculopathy. Jpn J Ophthalmol 2000;44:283–289.
- ↑ Kohno T, Miki T, Shiraki K, Kano K, Hirabayashi-Matsushita M. Indocyanine green angiographic features of choroidal rupture and choroidal vascular injury after contusion ocular injury. Am J Ophthalmol 2000;129:38–46.
- ↑ Baltatzis S, Ladas ID, Panagiotidis D, Theodossiadis GP. Multiple post-traumatic choroidal ruptures obscured by hemorrhage: imaging with indocyanine green angiography. Retina 1997;17:352–354.
- ↑ Jump up to: 38.0 38.1 38.2 38.3 38.4 Gologorsky D Rosen RB. Principles of Ocular Imaging : A Comprehensive Guide for the Eye Specialist. Thorofare NJ: SLACK Incorporated; 2021. http://public.eblib.com/choice/PublicFullRecord.aspx?p=6647261. Accessed July 21 2023.
- ↑ Carski TR, Staller BJ, Hepner G, Banka V, Finney RA. Adverse reactions after administration of indocyanine green. JAMA 1978; 240: 635.
- ↑ Hope-Ross M, Yannuzzi L, Gragoudas ES, Guyer DR, Slakter FS, Sorenson JA, et al. Adverse reactions due to indocyanine green. Ophthalmology 1994; 101: 529-33.