Choroidal Folds
All content on Eyewiki is protected by copyright law and the Terms of Service. This content may not be reproduced, copied, or put into any artificial intelligence program, including large language and generative AI models, without permission from the Academy.
Disease Entity
Choroidal Folds (or Chorioretinal Folds)
Disease
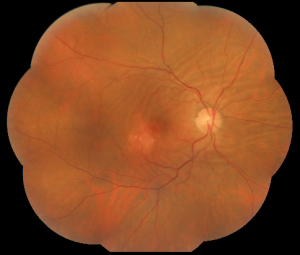
Choroidal folds are lines, grooves or striae predominantly involving the posterior pole of the eye which appear as alternating light and dark lines on fluorescein angiography. They are often arranged in a parallel and horizontal fashion but may be vertical, oblique, or irregular. These folds reflect the undulations of the choroid, retinal pigment epithelium (RPE) and the overlying neurosensory retina and tend to vary in length and width, rarely extending beyond the equator. [1] [2] [3] [4]
Etiology
They were first described clinically by Nettleship et al., as choroidal folds and then subsequently re-termed as "chorioretinal folds" by Gass to encompass the entire pathology seen clinically.[5] [6] These have been associated with a variety of ocular and orbital conditions such as acquired hyperopia, posterior scleritis, retrobulbar/orbital tumors, thyroid eye disease or Grave´s ophthalmopathy, choroidal naevi and tumors, optic nerve drusen, microphthalmos, following ophthalmic surgery (e.g. scleral buckling for retinal detachment or hypotony after trabeculectomy), hypotony, central serous chorioretinopathy, idiopathic intracranial hypertension or secondary pseudotumour cerebri syndrome and orbital cellulitis.[7] [8] [9]When no obvious cause is found, choroidal folds are classified as idiopathic.[10] [11]
Choroidal folds can occur unilaterally or bilaterally. Orbital tumors, intraocular tumors, hypotony, scleral buckling, trauma, choroidal detachment are associated with unilateral folds whereas thyroid disease, systemic medication, nanophthalmos, choroiditis are more commonly found in bilateral folds. Hyperopia and AMD can be seen in both unilateral and bilateral cases of folds.[12]
In the literature, the two largest case series of choroidal folds[3][12] as well as many other reports identified hyperopia and AMD as the most common associated conditions. A significant number of cases was considered idiopathic although with improved diagnostic testing, the patients formerly referred to as idiopathic actually represented a smaller portion of the total.
While the term “idiopathic” means the cause of the folds is unknown, previous authors proposed that such eyes may have had prior episodes of insidious or chronic inflammation of the orbit or paranasal sinuses or posterior scleritis, thus leading to permanent thickening of the posterior sclera and flattening of the posterior globe and resulting in relaxation and eventually compression of the posterior choroid, creating folds.[13] [14] [15] [16] In some cases of idiopathic choroidal folds, B-scan ultrasonography has been reported to show flattening of the posterior ocular wall, thought to be due to thickening of the ocular coats.[17]
An important topic to be mentioned is the possible (and sometimes inevitable) confusion between truly idiopathic choroidal folds and those associated with elevated intracranial pressure in which the optic nerve sheath becomes distended, which may in turn press on the globe causing distortion and hence choroidal folds[18] [19]. The findings may essentially be related to the timing of the evaluation. In some cases, the choroidal folds precede the development of papilloedema which suggests that the elevated intra-sheath pressure produces indentation of the globe before there is any reduction in retrolaminar perfusion and axoplasmic stasis.[20] In other cases choroidal folds may be present after the resolution of papilledema. It is possible that in the initial stages the globe is flattened together with distortion and folding of the choroid producing a hypermetropic shift[3] [7][21] [22] [18], and that later on when intracranial pressure is reduced and the papilloedema resolves, the mass effect of the distended nerve sheath on the globe is removed, allowing the refractive state of the eye to return to normal, but with the persistence of the choroidal folds.[23] Another explanation for this phenomenon is that retrolaminar optic nerve sheath enlargement may cause slight indentation of the posterior globe without causing obvious optic nerve head swelling. That is, the optic nerve sheath may distend enough to compress the retrolaminar globe, but the axons may not swell despite an increase in intracranial pressure.[23] Jacobson et. al have postulated that cases of acquired hyperopia with choroidal folds may also be a result of subclinical increases in intracranial pressure.[18] He suggested that the acquired hyperopia was a result of elevated pressure in the optic nerve sheath compressing the subarachnoid compartment and flattening the globe evident on neuroimaging and ultrasound evaluation. Unilateral cases of choroidal folds are associated with a higher frequency of orbital disease and with more significant ocular disease (e.g. scleritis, tumors, retinal vascular occlusion, and hypotony) than bilateral presentations, which are more associated with unknown causes and acquired hyperopia.[21][24]
Pathophysiology
The exact pathogenesis of choroidal folds is unknown, but it is believed that conditions causing thickening or shortening of the sclera, choroidal thickening and/or enlargement of various retrobulbar orbital structures, such as the optic nerve, the extraocular muscles, or Tenon's space produce wrinkling of Bruch’s membrane and the adjacent layers of the choroid and retinal pigment epithelium.[3][21] [22] [13][25]
Friberg et. al. were the first to explain the pathophysiology of choroidal folds based on biomechanical principles.[26] They postulated that choroidal folds are produced locally when the net compressive forces within the choroid, retinal pigment epithelium and Bruch’s membrane complex are of a sufficient magnitude to buckle or fold Bruch’s membrane and the retinal pigment epithelium. They emphasized that the magnitude of this force must exceed the counteracting tensile force within the choroid secondary to the intraocular pressure.[17] In a study with indocyanine green angiography, Haruyama et al. showed delayed filling in choroidal vessels during the early transit phase followed by choroidal venous dilation, suggesting choroidal vascular congestion in patients with choroidal folds.[14] This vascular congestion may result from resistance of choroidal outflow secondary to extravascular interstitial edema that accompanies many inflammatory conditions as well as from structural alterations within the scleral wall impairing choroidal filling or venous outflow.[27]
Diagnosis
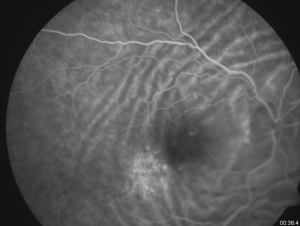
The diagnosis is made mainly by fluorescein angiography, wherein the folds appear as a series of alternating hyperfluorescent and hypofluorescent streaks starting early in the arteriovenous phase, persisting through the late venous phase, and not leaking in the late films. The hyperfluorescent areas correspond to the peaks of the choroidal folds, and the hypofluorescent areas to the valleys.[3]
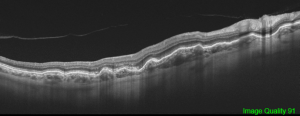
Fluorescein angiography cannot differentiate chorioretinal from choroidal folds, but the optical coherence tomography (OCT) patterns of the two are strikingly different although these are not completely singular entities. In cases of chorioretinal folds, OCT shows that the retina, RPE and choroid are all folded in a similar manner and maintain their normal relationships and apparently normal thickness, although a certain flattening of the inner retina can be seen along the crests, whereas choroidal folds correspond to undulations of the hyperreflective line corresponding to the RPE as well as the underlying choroid with a flat or nearly flat retinal surface with a variable retinal thickness variable, increasing in correspondence to the valleys and reducing in the crests. The retina, which has a softer structure, adapts itself to the wrinkling to preserve a flat inner surface.[19] The severity of choroidal folds have been staged on the basis of FA imaging. [28]
Ultrasound, computed tomography (CT) or magnetic resonance (MR) scanning of the orbits or brain may be indicated. The characteristic CT and ultrasonography findings of acquired hyperopia with choroidal folds are variable enlargement of the optic nerve image, sometimes discernible space between the nerve and its sheath, distension of the peri-optic subarachnoid space, scleral shortening and congestive choroidal thickening.[4] [21] [26] [24]
Linear signals of a decrease in choriocapillaris perfusion associated with choroidal folds can be seen on OCT angiography. [29]
Symptoms
Depending upon their location and onset, choroidal folds can be asymptomatic or lead to metamorphopsia and decreased vision. When the folds involve the macula, they may or may not affect visual acuity, normally through acute distortion of the overlying retinal photoreceptors, chorioretinal folds-related maculopathy (see below) or when associated with retinal disease. The syndrome of acquired hyperopia with choroidal folds is characterized by a painless loss in vision resulting from a hyperopic shift in their refractive error but where visual acuity typically returns to baseline levels with an increase in hyperopic correction.[4][26]
Differential diagnosis
Retinal folds.
The characteristic fluorescein pattern of hyperfluorescent bands alternating with hypofluorescent bands allows a clear distinction between choroidal and retinal folds.[15]
Management
There is sufficient evidence that as soon as choroidal folds are recognized, a prompt systemic investigation for a specific diagnosis or cause is mandatory in order to diagnose treatable causes or to exclude potentially serious conditions.
Complications
The presence of macular pathologic features secondary to chronic folds was reported first by Newell, who noted cases of RPE pigment clumping and atrophy as well as linear streaks of fluorescein staining along the folds. Olsen et al.[22] also reported that chronic choroidal folds involving the macula may result in a unique maculopathy that they called chorioretinal folds-related maculopathy (CRF-related maculopathy). In this condition there is usually an insidious progression and gradual decline in visual acuity over time associated with either a yellow, luteal macular appearance or atrophy of the retinal pigment epithelium as well as areas of retinal pigment epithelium hyperplasia. The choroidal vascular abnormalities reported by Haruyama et al.[23] may also result in an ischemic choroidal environment which in conjunction with tensile stress created by long-standing choroidal folds on the Bruch membrane–choroid complex create an ideal situation for Bruch membrane fracture and formation of choroidal neovascularization[22][20], and monitoring for the development of choroidal neovascularization is advised.[30] Nonetheless, in the series reported by Olsen et al. visual acuity loss seems to result mostly from degenerative changes to the retinal pigment epithelium–Bruch's complex. CRF-related maculopathy has a slow and indolent course with gradual macular dysfunction that evolves over many years[22] and careful monitoring of the retina (especially the macula) should therefore be undertaken at regular intervals.[4]
Prognosis
If the folds are longstanding, vision may be preserved[19] [20] unless a secondary maculopathy develops. The overall prognosis is otherwise excellent if no serious associated condition is diagnosed.
References
- ↑ Bowling B. Kanskiʼs Clinical Ophthalmology: A Systematic Approach. Eighth Edition. Cornea. 2016.
- ↑ Bullock JD, Egbert PR. The origin of choroidal folds a clinical, histopathological, and experimental study. Doc Ophthalmol. 1974. doi:10.1007/BF00147262
- ↑ 3.0 3.1 3.2 3.3 3.4 Cangemi FE, Trempe CL, Walsh JB. Choroidal folds. Am J Ophthalmol. 1978. doi:10.1016/0002-9394(78)90243-X
- ↑ 4.0 4.1 4.2 4.3 Dailey RA, Mills RP, Stimac GK, Shults WT, Kalina RE. The Natural History and CT Appearance of Acquired Hyperopia with Choroidal Folds. Ophthalmology. 1986. doi:10.1016/S0161-6420(86)33577-2
- ↑ Nettleship E. Peculiar lines in the choroid in a case of postpapillitic atrophy. Trans Ophthalmol Soc UK. 1884;4:167–168.
- ↑ Gass JD. Radial chorioretinal folds. A sign of choroidal neovascularization. Arch Ophthalmol. 1981 Jun; 99(6):1016-8.
- ↑ 7.0 7.1 Jaworski A, Wolffsohn JS, Napper GA. Aetiology and management of choroidal folds. Clin Exp Optom. 1999. doi:10.1111/j.1444-0938.1999.tb06638.x
- ↑ Wu W, Wen F, Huang S, Luo G, Wu D. Choroidal Folds in Vogt-Koyanagi-Harada Disease. Am J Ophthalmol. 2007. doi:10.1016/j.ajo.2006.11.050
- ↑ Corvi F, Capuano V, Benatti L, et al. Atypical presentation of chorioretinal folds-related maculopathy. Optom Vis Sci. 2016;93:1304–1314. doi:10.1097/OPX.0000000000000953
- ↑ Lavinsky J, Lavinsky D, Lavinsky F, Frutuoso A. Acquired choroidal folds: A sign of idiopathic intracranial hypertension. Graefe’s Arch Clin Exp Ophthalmol. 2007. doi:10.1007/s00417-006-0455-7
- ↑ Corvi F, Capuano V, Benatti L, Bandello F, Souied E, Querques G. Atypical presentation of chorioretinal folds-related maculopathy. Optom Vis Sci. 2016. doi:10.1097/OPX.0000000000000953
- ↑ 12.0 12.1 Leahey AB, Brucker AJ, Wyszynski RE, Shaman P. Chorioretinal Folds: A Comparison of Unilateral and Bilateral Cases. Arch Ophthalmol. 1993. doi:10.1001/archopht.1993.01090030075042
- ↑ 13.0 13.1 Friberg TR. The etiology of choroidal folds - A biomechanical explanation. Graefe’s Arch Clin Exp Ophthalmol. 1989. doi:10.1007/BF02172899
- ↑ 14.0 14.1 Jacobson DM. Intracranial Hypertension and the Syndrome of Acquired Hyperopia with Choroidal Folds. J Neuro-Ophthalmology. 1995. doi:10.1097/00041327-199509000-00011
- ↑ 15.0 15.1 Norton EWD. A Characteristic Fluorescein Angiographic Pattern in Choriodal Folds. J R Soc Med. 1969. doi:10.1177/003591576906200204
- ↑ Bagnis A, Cutolo CA, Corallo G, Musetti D, Nicolò M, Traverso CE. Chorioretinal folds: a proposed diagnostic algorithm. Int Ophthalmol. 2019. doi:10.1007/s10792-019-01083-y
- ↑ 17.0 17.1 Bird AC, Sanders MD. Choroidal folds in association with papilloedema. Br J Ophthalmol. 1973. doi:10.1136/bjo.57.2.89
- ↑ 18.0 18.1 18.2 Griebel SR, Kosmorsky GS. Choroidal folds associated with increased intracranial pressure. Am J Ophthalmol. 2000. doi:10.1016/S0002-9394(99)00412-2
- ↑ 19.0 19.1 19.2 Giuffrè G, Distefano MG. Optical coherence tomography of chorioretinal and choroidal folds. Acta Ophthalmol Scand. 2007. doi:10.1111/j.1600-0420.2006.00799.x
- ↑ 20.0 20.1 20.2 Harrington SP. Unilateral choroidal folds. Clin Eye Vis Care. 1995. doi:10.1016/0953-4431(95)00138-7
- ↑ 21.0 21.1 21.2 21.3 Cappaert WE, Purnell EW, Frank KE. Use of B-sector scan ultrasound in the diagnosis of benign choroidal folds. Am J Ophthalmol. 1977. doi:10.1016/0002-9394(77)90682-1
- ↑ 22.0 22.1 22.2 22.3 22.4 Olsen TW, Palejwala N V., Lee LB, Bergstrom CS, Yeh S. Chorioretinal folds: Associated disorders and a related maculopathy. Am J Ophthalmol. 2014. doi:10.1016/j.ajo.2014.02.021
- ↑ 23.0 23.1 23.2 Haruyama M, Yuzawa M, Kawamura A, Yamazaki C, Matsumoto Y. Indocyanine green angiographic findings of chorioretinal folds. Jpn J Ophthalmol. 2001. doi:10.1016/S0021-5155(01)00323-9
- ↑ 24.0 24.1 Atta HR, Byrne SF. The Findings of Standardized Echography for Choroidal Folds. Arch Ophthalmol. 1988. doi:10.1001/archopht.1988.01060140394040
- ↑ Hyvärinen L, Walsh FB. Benign chorioretinal folds. Am J Ophthalmol. 1970. doi:10.1016/0002-9394(70)90660-4
- ↑ 26.0 26.1 26.2 Kalina RE, Mills RP. Acquired Hyperopia with Choroidal Folds. Ophthalmology. 1980. doi:10.1016/S0161-6420(80)35279-2
- ↑ Cassidy LM, Sanders MD. Choroidal folds and papilloedema. Br J Ophthalmol. 1999. doi:10.1136/bjo.83.10.1139
- ↑ The Olsen TW, Palejwala NV, Lee LB, Bergstrom CS, Yeh S. Chorioretinal folds: associated disorders and a related maculopathy. Am J Ophthalmol. 2014 May;157(5):1038-47.
- ↑ Del Turco C, Rabiolo A, Carnevali A, et al. Optical coherence tomography angiography features of chorioretinal folds: a case series. Eur J Ophthalmol. 2017;27:e35–e38. doi:10.5301/ejo.5000872
- ↑ Goel N, Kumar V, Raina UK, Ghosh B. An unusual pattern of idiopathic choroidal folds. Oman J Ophthalmol. 2012. doi:10.4103/0974-620X.99381