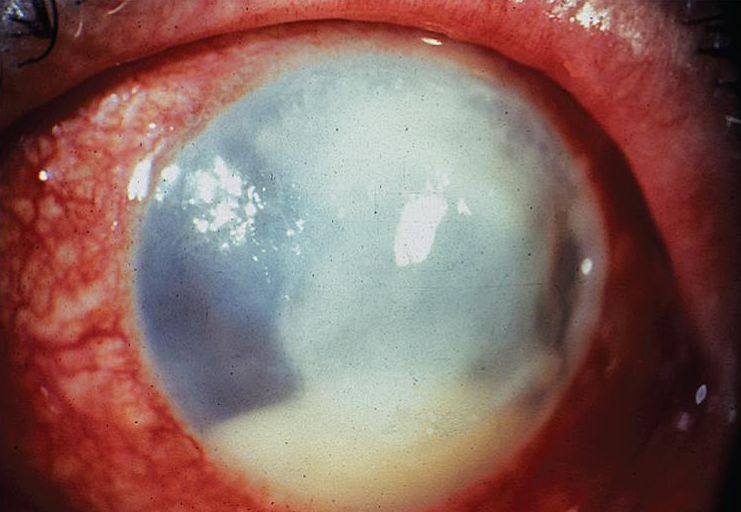
Bacterial Keratitis
All content on Eyewiki is protected by copyright law and the Terms of Service. This content may not be reproduced, copied, or put into any artificial intelligence program, including large language and generative AI models, without permission from the Academy.
Disease Entity
2022 ICD-10
- H16.013 Central corneal ulcer, bilateral
- H16.011 Central corneal ulcer, right eye
- H16.012 Central corneal ulcer, left eye
Disease
Bacterial keratitis is also often referred to as a 'corneal ulcer'. In practice, these terms are not directly interchangeable because a cornea may harbor a bacterial infection (i.e bacterial keratitis) without having a loss of tissue (an ulcer) and a cornea may have an ulcer without a bacterial infection.
Bacterial keratitis is a serious bacterial infection of the cornea which can, in severe cases, cause loss of vision.
Etiology
Although infectious ulcers may also be due to fungi, viruses, mycobacteria and protozoa, bacteria are the most common cause of infectious keratitis. Both gram positive and gram negative organisms are implicated as causative agents. About 80 % of bacterial keratitis is caused by Staphylococcus, Streptococcus and Pseudomonas species, though prevalence can depend on geographical regions. Certain bacteria are known to penetrate the intact epithelium which include Neisseria, Corynebacterium, Shigella and Listeria. Polymicrobial infections can occur, up to 43% in one study[2]. [3][4][5]
Risk Factors
Risk factors for bacterial keratitis are those that cause disruption of the integrity of the corneal epithelium. The most common risk factor for bacterial keratitis is contact lens wear. Contact lens wear has been associated with 19%-42% of cases of culture proven corneal infections. Overnight wear and inadequate lens disinfection have been associated with increased risk of infection. Other predisposing factors include: trauma (including foreign bodies and chemical and thermal injuries), contaminated ocular solutions, changes in the corneal surface (from dry eye, eyelid misdirection, and exposure), altered ocular defense mechanisms (from topical and systemic immune suppression), loose sutures with adjacent infections (blepharitis and viral keratitis), and corneal edema. In younger patients, trauma and contact lens wear are the most common predisposing factors while in older patients, chronic corneal disease such as dry eyes, surgical trauma, and bullous keratopathy are also important risk factors.
General Pathology
Bacterial keratitis can advance through four stages: progressive infiltration, active ulceration, regression, and healing.
Pathophysiology
Corneal infections rarely occur in the normal eye. They are a result of an alteration in the cornea’s defense mechanisms that allow bacteria to invade when an epithelial defect is present. The organisms may come from the tear film or as a contaminant from foreign bodies, contact lenses or irrigating solutions. The severity of the disease depends on the strain of the organism, the size of the inoculum, the susceptibility of the host and immune response, the antecedent therapy, and the duration of the infection. The process of corneal destruction can take place rapidly (within 24hrs with virulent organisms) so that rapid recognition and initiation of treatment is imperative to prevent visual loss.
Primary Prevention
Avoidance of predisposing factors may reduce the risk of corneal infection. Proper education of contact lens use and care can help decrease the risk. The use of protective eyewear for sports and outdoor activities can help prevent trauma and the subsequent development of infection.
Diagnosis
A diagnosis of bacterial keratitis should include a detailed history and a complete ophthalmic examination.
History
A detailed history is as important as the examination. Physicians should take a detailed history and ask about the characteristics and onset of symptoms, whether there was recent trauma to the eye, and whether the patient engaged in activities such as swimming in contact lenses. Patients should be asked about contact lens wear (including type of lens used, time since last change to a new pair of lenses, hours of continuous wear, and cleaning regimen). A past ocular history should include whether there was a history of eye trauma, previous eye diseases (such as viral keratitis), or eye surgeries. A past medical history (history of diabetes), a list of medications and eye drops, a documentation of allergies, a pertinent family history, substance abuse history, and a review of systems should be obtained.
Physical Examination
A complete examination including vision, intraocular pressure, pupil assessment, and slit-lamp examination should be initiated. Flourescein can be used to highlight areas of epithelial cell loss. The ophthalmologist should document the location, size and depth of the corneal infiltrate. Any anterior chamber reaction (cells, flare, fibrin, or hypopyon), should be recorded. Patients should be dilated and possible posterior pole involvement should be ruled out. If the posterior pole is unable to be visualized, an ultrasound should be performed. The ophthalmologist should also test corneal sensation, proper eyelid closure, eyelids and lashes, and nasolacrimal apparatus to look for risk factors for infection. Both eyes should always be examined.
Signs
Signs of bacterial keratitis might include conjunctival injection and focal white infiltrates (with epithelial demarcation and underlying stromal inflammation). Other signs can include: corneal thinning, stromal edema, endothelial inflammatory plaque, Descemet’s folds, mucopurulent discharge, anterior chamber reaction and hypopyon. Eyelid edema may be present in some cases. In severe cases, posterior synechiae, hyphema, and glaucoma may occur.
Symptoms
Symptoms include rapid onset of ocular pain, redness, photophobia, discharge, and decreased vision. The rate of progression of the symptoms is related to the virulence of the infecting organism.
Clinical Diagnosis
Diagnosis is based on clinical history and slit lamp examination showing the presence of a corneal infiltrate.
Diagnostic procedures
Corneal scrapings for Gram and Giemsa staining and cultures are performed to determine the causative organism in all ulcers that are either large (>2mm), involve the middle to deep stroma, are sight threatening, are chronic in nature, are atypical, or are unresponsive to treatment. Small infiltrates that do not stain may sometimes be treated with broad spectrum antibiotics without scraping.
Corneal scrapings can be performed using 57 blade, beaver blade, or sterile Kimura spatula after administration of topical anesthetic. An eyelid speculum can be used for exposure if patient tolerates. Samples should be directly inoculated onto culture media, to increase yield. Typical culture media should include chocolate, blood, sabouraud agar, non-nutrient agar (with E Coli overlay for acanthamoeba), and thioglycollate broth.
Corneal biopsies should be considered if patient is unresponsive to treatment or cultures are negative despite high clinical suspicion for infectious etiology. This can be performed at the slit lamp or the operating room using a small 2-3 mm dermatologic punch and crescent blade at the edge of the ulcer. Biopsing the central cornea should be avoided.
Suture pass cultures can be obtained for deep stromal ulcers especially if the overlying epithelium appears intact or uninvolved. A 7-0 or 8-0 vicryl or silk suture can be passed through the deep abscess, cut into smaller pieces using sterile scissors and plated onto culture media[6].
Differential diagnosis
The differential diagnosis of bacterial keratitis is large. Other infectious etiologies must be considered. Non-infectious (or sterile) ulcers may be related to dry eye syndrome, exposure or neurotrophic keratopathy, autoimmune diseases (such as rheumatoid arthritis), vernal keratoconjunctivitis, vitamin A deficiency, and staphylococcal hypersensitivity. Round, white scars from old foreign bodies may be confused with small infiltrates.
Management
General treatment
Contact lenses should be discontinued. Topical antibiotic drops should be prescribed. Oral antibiotics may be considered for patients with deep ulcers or scleral involvement. Oral medication can be used, as needed, for pain. Oral antivirals should be started if viral keratitis is suspected as the underlying etiology.
Medical therapy
Topical broad spectrum antibiotic therapy should be used until culture results are available. Treatment may be selected according to the risk of potential visual loss. Small non-staining peripheral ulcers may be started on fluoroquinolone drops every 2 to 6 hours. For ulcers with epithelial defects and an anterior chamber reaction, a fluoroquinolone drop every hour around the clock is recommended. Large or vision threatening ulcers (with moderate to severe anterior chamber reaction and/or involving the visual axis) should be cultured then treated with fortified tobramycin or gentamicin (15mg/ml) every hour around the clock alternating with fortified vancomycin (25-50 mg/ml) every hour around the clock. Sensitivity testing should be used to guide de-escalation of broad spectrum fortified antibiotics to more targeted therapy.
Cycloplegia is recommended in patients with significant anterior chamber reaction for pain control and prevention of posterior synechia formation. IOP lowering therapy should be initiated if IOP is elevated.
Topical corticosteroids can be considered 24-48 hours after topical antibiotics if the causative organism is identified or there is demonstrated response to topical antibiotics. A subgroup analysis of the Steroids for Corneal Ulcers Trial (SCUT) found that the addition of topical corticosteroids within 2 to 3 days of antibiotic therapy vs 4 or more days resulted in a 1-line better visual acuity at 3 months compared with placebo. [7][8] However, Corticosteroids should be avoided in cases of Acanthamoeba, Nocardia, and fungus.
Oral doxycycline and vitamin C supplementation can be considered in severe cases to prevent keratolysis.
Corneal crosslinking is an emerging treatment option for refractory cases of bacterial keratitis. Photoexcitation of riboflavin or rose-bengal[9] are being studied in its germicidal properties and ability to strengthen collagen fibrils to prevent corneal melt[10].
Medical follow up
Daily follow up is needed until a response to antibiotic regimen is noted. On follow up, assess the size of the epithelial defect, the size and depth of the infiltrate, the degree of pain and the anterior chamber reaction. Taper the antibiotics when ulcer improves. If the ulcer worsens or does not improve, consider culturing again, adding fortified antibiotics (e.g., vancomycin and tobramycin), subconjunctival antibiotics. Consider a corneal biopsy if the ulcer does not improve. In severe non-responding ulcers, hospitalization for frequent monitoring and drop administration may be required. Admission may be needed if there is scleral extension or corneal perforation and systemic antibiotics and/or surgery are needed; the patient is unable to instill the medication as prescribed, he is unable to return for follow up or is noncompliant.
Surgery
Lamellar or penetrating keratoplasties can be performed in cases that are unresponsive to medical therapy or in cases of large perforations. Peripheral iridectomies should be performed at the time of penetrating keratoplasty. Smaller perforations (<2mm) can be treated with corneal gluing and bandage contact lenses.
For patients with poor eyelid closure or neurotrophic keratitis, temporary or permanent tarsorrhaphy should be considered to aid healing.
Surgical follow up
When a corneal transplant is indicated, close postoperative follow-up is recommended. The cornea surgeon should evaluate for recurrence of the infection in the graft as well as graft failure and other possible postoperative complications. The host corneal button should be sent for pathologic analysis and culture.
Complications
Potential complications include scleral extension of the infection, residual corneal scarring, irregular astigmatism, loss of vision, corneal perforation, and endophthalmitis .
Prognosis
The prognosis depends on the size, location, depth, and etiology of the corneal ulcer as well as any pre-existing ocular conditions.
Additional Resources
- Lin A, Rhee MK, Akpek EK, Amescua G, Farid M, Garcia-Ferrer FJ, Varu DM, Musch DC, Dunn SP, Mah FS; American Academy of Ophthalmology Preferred Practice Pattern Cornea and External Disease Panel. Bacterial Keratitis Preferred Practice Pattern®. Ophthalmology. 2019 Jan;126(1):P1-P55. doi: 10.1016/j.ophtha.2018.10.018. Epub 2018 Oct 23. PMID: 30366799.
- Turbert D, Huffman JM. Bacterial Keratitis. American Academy of Ophthalmology. EyeSmart/Eye health. https://www.aao.org/eye-health/diseases/bacterial-keratitis-27. Accessed November 17, 2022.
- Turbert D, Huffman JM. Corneal Ulcer. American Academy of Ophthalmology. EyeSmart/Eye health. https://www.aao.org/eye-health/diseases/corneal-ulcer-list. Accessed November 17, 2022.
References
- ↑ American Academy of Ophthalmology. Bacterial keratitis. https://www.aao.org/education/image/bacterial-keratitis-42 Accessed February 26, 2025.
- ↑ Pakzad-Vaezi K, Levasseur SD, Schendel S et al. The corneal ulcer one-touch study: a simplified microbiological specimen collection method. Am J Ophthalmol. 2015; 159 (e31.): 37-43
- ↑ Synder RW, Hyndiuk RA. Mechanisms of bacterial invasion of the cornea. In: Tasman W, Jaeger EA, editors. Duane‘s Foundations of Clinical Ophthalmology. Philadelphia: J B Lippincott & Co.; 1990. pp. 11–44. [Google Scholar]
- ↑ Tuberville AW, Wood TO. Corneal ulcers in corneal transplantation. Curr Eye Res. 1981;8:479. [PubMed] [Google Scholar]
- ↑ Badenoch PR, Aggarwal RK, Coster DJ. Clostridium perfringens keratitis after penetrating keratoplasty. Aust NZ J Ophthalmol. 1995;23:245. [PubMed]
- ↑ Lin A, Rhee MK, Akpek EK, Amescua G, Farid M, Garcia-Ferrer FJ, Varu DM, Musch DC, Dunn SP, Mah FS; American Academy of Ophthalmology Preferred Practice Pattern Cornea and External Disease Panel. Bacterial Keratitis Preferred Practice Pattern®. Ophthalmology. 2019 Jan;126(1):P1-P55. doi: 10.1016/j.ophtha.2018.10.018. Epub 2018 Oct 23. PMID: 30366799
- ↑ Ray KJ, Srinivasan M, Mascarenhas J et al. Early addition of topical corticosteroids in the treatment of bacterial keratitis. JAMA Ophthalmol. 2014; 132: 737-741
- ↑ Srinivasan M, Mascarenhas J, Rajaraman R, Steroids for Corneal Ulcers Trial Group et al. Corticosteroids for bacterial keratitis: the Steroids for Corneal Ulcers Trial (SCUT). Arch Ophthalmol. 2012; 130: 143-150
- ↑ Altamirano D, Martinez J, Leviste KD, Parel JM, Amescua G. Photodynamic Therapy for Infectious Keratitis. Curr Ophthalmol Rep. 2020 Dec;8:245-251. doi: 10.1007/s40135-020-00252-y. Epub 2020 Sep 12. PMID: 34540359; PMCID: PMC8445507.
- ↑ Singh RB, Das S, Chodosh J, Sharma N, Zegans ME, Kowalski RP, Jhanji V. Paradox of complex diversity: Challenges in the diagnosis and management of bacterial keratitis. Prog Retin Eye Res. 2021 Nov 20:101028. doi: 10.1016/j.preteyeres.2021.101028. Epub ahead of print. PMID: 34813978.
- H.E., Barron, L., McDonald, M.B., eds., The Cornea, 2d ed., 1996
- External Disease and Cornea, Chapter 12: Infectious Diseases of the Cornea and External Eye: Bacterial, Fungal, and Parasitic Infections. Basic and Clinical Science Course, AAO, 2022.
- The Wills Eye Manual, 4th Edition. Kunimoto, Kanitkar, & Makar. Lippincott, 2004.