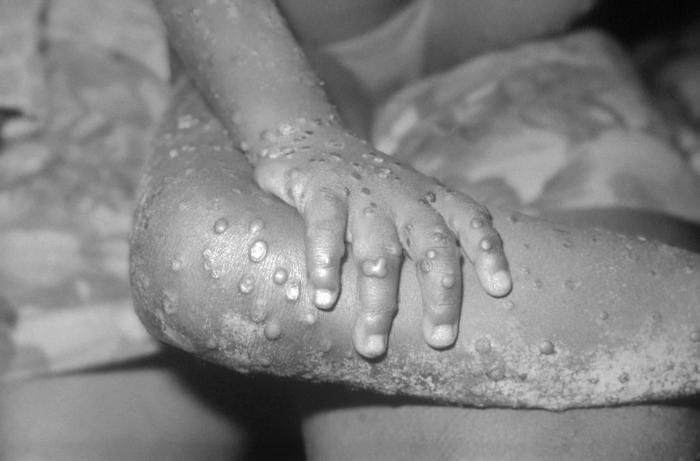
Monkeypox
All content on Eyewiki is protected by copyright law and the Terms of Service. This content may not be reproduced, copied, or put into any artificial intelligence program, including large language and generative AI models, without permission from the Academy.
Disease Entity
Disease
Human Monkeypox (MPX) is a zoonosis which presents with a smallpox-like disease.[2] MPX was named for its initial identification in 1958 in a group of monkeys transported to Copenhagen, Denmark, from Africa.[3]
Etiology
MPX is caused by the monkeypox virus (MPXV), an enveloped double-stranded DNA virus that is a member of the genus Orthopoxvirus. Other members of Orthopoxvirus include variola (smallpox), vaccinia, cowpox virus, camelpox virus, and ectromelia virus (mousepox).[2] MPXV is, in a sense, a misnomer: although initially identified in monkeys, the reservoir species for MPXV is thought to be an unidentified species of rodent (possibly Gambian pouched rat, rope squirrel, or other rodent).[4][5] Transmission is by broken skin, droplets, or bodily fluids.[6] Sexual activity is thought to be either a route of transmission or may amplify transmission.[7] The incubation period for the virus is 7–14 days most frequently but has been reported to be up to 3 weeks.[6]
Epidemiology
There are two clades of MPXV, a West African (WA) clade and a Congo Basin (CB) clade.[8] MPX disease caused by the CB clade may have greater mortality, morbidity, viremia, and transmission relative to disease caused by the WA strain.[8]
Countries which are endemic for MPX include: Cameroon, the Central African Republic, the Democratic Republic of the Congo, Gabon, Ghana (identified in animals only), Cote d’Ivoire, Liberia, Nigeria, the Republic of the Congo, and Sierra Leone.[9] MPX is rare outside of endemic countries, but in 2003, an outbreak occurred in the United States related to MPXV-infected prairie dogs.[10]
Since May 2022, there has been an ongoing international outbreak of MPX which includes nonendemic countries.[7][11][12] As of July 29, 2022, there have been 22,485 cases of which the greatest number of cases are in the United States (4906 cases), Spain (4298 cases), Germany (2595 cases), United Kingdom (2546 cases), and France (1955 cases).[13]
Risk Factors
Outbreaks in endemic countries have revealed the following risk factors for MPX infection: lack of smallpox vaccination, living in forested area, male sex, age less than 15 years and consumption of bushmeat.[3]
The 2022 global outbreak has shown a disproportionate occurrence in men who have sex with men (MSM), which has not been noted in previous outbreaks.[7] Although a majority of cases in the 2022 outbreak have been in adult men (especially MSM),[7] cases in women[14] and in children[15] have also been reported. Incident cases commonly had a history of travel (most frequently to Europe) in the month prior to onset symptoms.[7]
Pathology
Dermatopathology shows similar histologic findings to vaccinia and cowpox virus.[16] Guarnieri bodies, characteristic of poxviruses, are present in skin keratinocytes.[16] Other pathologic features may include proliferation of basal keratinocytes, epidermal necrosis and spongiosis, keratinocyte pallor, and dense inflammatory cell infiltrate with vasculitis.[11][16]
Primary Prevention
Data from previous outbreaks have shown that smallpox vaccination provides 85% protection against MPX.[17] In the United States, there are two currently available smallpox vaccines that may provide protection against MPX. ACAM2000 utilizes replication-competent vaccinia virus, and is administered by percutaneous skin scarification in a single dose.[18][19] JYNNEOS is a newer vaccine preparation which contains a replication-deficient vaccinia virus and is administered subcutaneously in two doses. These vaccines are available in the United States for pre-exposure prophylaxis in certain high-risk occupational groups,[18] and for postexposure prophylaxis.[11] The JYNNEOS vaccine is becoming increasingly available, and local/regional departments of public health have information on how vaccines are being distributed in any particular area.[20]
Clinical Manifestations
Ophthalmic
Ophthalmic manifestations of monkeypox have been termed monkeypox-related ophthalmic disease (MPXROD)[21] or ocular monkeypox[22]. It begins at the site of inoculation at the skin or mucosa and then subsequently spreads to the regional lymph nodes. MPXROD is mainly an external disease, involving the lids and periorbita, conjunctiva and cornea. However, anterior uveitis has been described in recent case reports. Unlike smallpox, other ophthalmic manifestations, such as retinitis, retinochoroiditis, optic neuritis, dacryoadenitis and ophthalmoplegia have yet to be reported. In persistent cases, MPXROD may be present even up to 6 weeks from monkeypox onset.[23]
The 2022 monkeypox outbreak reported a low incidence of ocular monkeypox of <1% (27 EU/EEA countries (> 20,000 cases as of 27 September 2022). However, previous outbreaks observed a significantly higher incidence - between 9-23% - in regions where monkeypox was endemic[24].
Lid and Adnexa
Skin lesions classically appear vesiculo-pustular and involve the eyelids in up to 25% of patients. They evolve in stages from macules, papules, vesicles, pustules to crusted lesions, not unlike the Varicella-Zoster virus. [25] Patients can also present with eyelid edema, blepharitis and preseptal cellulitis. [26][27] Refractory skin necrosis with eschar formation has also been reported in severe cases.[28] Long-term sequelae include scarring and deformation of the eyelids.
Conjunctiva
Conjunctivitis may be seen in 20% of patients and more commonly affects children younger than 10 years old[21]and unvaccinated (30%) than vaccinated individuals (7%)[29]. They may present with conjunctival follicular reaction, discrete vesicular or papular conjunctival lesions, conjunctival ulceration, pseudomembranes or subconjunctival nodules[21][24].
Cornea
Monkeypox can lead to ulcerative keratitis, immune stromal keratitis, neurotrophic keratitis[21], corneal edema, limbitis and corneal staphyloma[27]. Ulcerative keratitis was reported to occur in 3%–4% of MPX cases in a CB outbreak.[30] Severe cases can cause corneal scarring and bacterial superinfection, leading to permanent visual impairment[31] and necessitating corneal transplant[21].
A case report described bilateral corneal perforation in a patient with severe necrotizing oculocutaneous monkeypox. Despite attempts to seal the perforation with cyanoacrylate glue for one of his eyes, the corneal perforation recurred with extrusion of the crystalline lens[28].
Sclera
Scleral involvement has been described in a few case reports. One case report had sectoral hyperemia associated with a subconjunctival nodule. [32] Two others were associated with extensive conjunctivitis [33]and confluent corneal lesions[34].
Avascular scleral necrosis was reported in a man with male sexual partners who had direct inoculation of semen into his eye. The patient presented with an itchy eye and nasal scleral redness which progressed to purulent conjunctivitis, corneal epithelial sloughing , necrosis of his nasal sclera and anterior uveitis. Anterior chamber paracentesis confirmed the diagnosis of monkeypox[35].
Uveitis
Although MPXORD is understood to be mainly an external disease, there have been 7 reported cases of anterior uveitis. 6 of the 7 cases[36][37][38][39][35]were associated with corneal involvement suggesting the intraocular inflammation might be a secondary sequela. Anterior chamber activity ranged between 0.5-2+ cells and keratic precipitates varied in size from small to mutton-fat in appearance. Only 1 case had monkeypox-related anterior uveitis in the absence of corneal involvement. The patient presented with anterior uveitis causing painless blurring of vision 2 weeks after resolution of a conjunctival vesicle[40].
Systemic
Skin lesions progress through the following stages in order: macule, papule, vesicle, pustule, crusting, and scar.[41] Lesions on the same part of the body are typically in the same stage.[42] Classically, the distribution of skin lesions in MPX is centrifugal and may be most dense on the face, trunk, and extremities.[42] As the skin lesions may occur on the face, the eyelids may be involved[43]. In the 2022 outbreak, the anogenital area has been the most common site for skin lesions (73% of patients).[44]
Mucosal lesions may also occur and may affect oropharyngeal,[41] nasal,[44] genital,[41][44] and ocular mucosa.[41]
Systemic sequelae may include bronchopneumonia and respiratory distress, gastrointestinal involvement with vomiting and diarrhea, encephalitis, and secondary bacterial infections and sepsis.[45][43] Lymphadenopathy is a clinical feature that distinguishes MPX from smallpox.[43]
Diagnosis
History
Patients may provide a history of contact with a known MPX case. There may be a history of recent travel to MPX-endemic countries (West or Central Africa) or to nonendemic countries where the present outbreak is ongoing.[46] Given the recent outbreak’s possible sexual transmission, it is prudent to inquire about sexual practices, new sexual partners, or whether sexual partners are experiencing symptoms.[7]
Physical Examination
The classic ophthalmic presentation is the presence of vesicles and pustules on the eyelids and periorbita[27]. However, given the spectrum of ophthalmic involvement, it is prudent to thoroughly examine the lids, conjunctiva, cornea and sclera as well. Though rare, intraocular involvement in the form of anterior uveitis can also be present.
Investigations
Ancillary testing to confirm diagnosis is generally performed by swabbing skin lesions, and when MPXROD is present the ocular surface may also be sampled.[47] The swabs may be sent for nucleic acid amplification testing by real-time or conventional polymerase chain reaction.
Differential Diagnosis
- Chickenpox (varicella)
- Cowpox
- Herpes simplex
- Molluscum contagiosum
- Scabies
- Shingles (herpes zoster)
- Smallpox (variola)
- Syphilis
- Vaccinia
Management
Systemic Treatment
MPX course is usually self-limited, so treatment is frequently supportive only.[6] Patients with severe disease or at risk for developing severe disease may be treated with systemic antiviral agents, which include tecovirimat, cidofovir, brincidofovir, and intravenous vaccinia immune globulin may be utilized for systemic treatment.[6][11][48] Antiviral agents are available in the United States through state/regional health departments and the Centers for Disease Control and Prevention.[48] Hospitalization may be needed.[7]
Treatment of Ocular Disease
Aggressive lubrication has historically been used for treatment of ocular manifestations of orthopoxvirus infections including by MPXV[49]. Topical antibiotics may be necessary for prophylaxis of epithelial defects or for bacterial superinfection.[49] Trifluridine has been used for ocular manifestations of the closely related virus vaccinia,[50] and may be considered in MPXROD.[12] In severe cases, corneal transplantation may be needed for perforation or for visual rehabilitation.[51]
Prognosis
The typical duration of symptoms and signs of MPX is 2–5 weeks.[3] As noted above, the course of MPX is usually self-limited. Skin lesions may lead to scarring, especially when scratched.[49][52] The lesions may appear atrophic and may be either hypo- or hyper-pigmented.[19] The historical case fatality rate from prior outbreaks is 10.6% in the CB clade and 3.6% in the WA clade.[53] In the 2022 outbreak, the mortality rate appears to be lower: in the retrospective series by Thornhill et al, of 528 cases no deaths were reported.[7]
When the cornea is involved, MPX may cause severe corneal scarring which may result in vision loss.[49][52][26]
Transmission Prevention
Affected individuals are advised to isolate at home to minimize transmission. They are advised to remain in isolation until scabs have fallen off, the rash has fully healed, and new intact skin has formed. Contact with other persons and with pets or animals should be avoided. Commonly touched surfaces should be cleaned and disinfected, and potentially contaminated items (including linens and eating/drinking utensils) should not be shared.[54]
In the healthcare setting, a suspected or known case should be placed in a single-person room with the door closed, and movement beyond the room minimized. Healthcare providers should wear personal protective equipment including a gown, gloves, eye protection that covers the front and side of the face, and a particulate respiratory mask with an N95 filter or higher.[55] Surfaces should be cleaned, using an United States Environmental Protection Agency (EPA)-registered hospital-grade disinfectant with an emerging viral pathogen claim,[55] a claim that is regulated by the EPA.[56] For eyecare providers, it may be prudent to utilize slit lamp shields as a barrier. Detailed guidance for infection prevention both at home and in the healthcare setting are available on the CDC website.[54][55]
It is not known if monkeypox can be transmitted through cadaveric corneal transplantation, as no such cases have been reported. Studies in infected rodents suggest conjunctival shedding of MPXV,[5] so the possibility for transmission in humans via cadaveric corneal transplantation is theoretically plausible if viral shedding occurs similarly in humans. Because MPXV is an enveloped virus, it should be susceptible to povidone-iodine disinfection.[57] The Eye Bank Association of America (EBAA) recommended exclusion of potential donors who in the last 21 days developed a rash characteristic of MPX, had a close contact with a confirmed MPX infection, tested positive for MPXV, or tested positive for Orthopoxvirus.[58]
Additional Resources
- Centers for Disease Control and Prevention (CDC) webpage about monkeypox
- World Health Organization (WHO) webpage about monkeypox
- Eye Bank Association of American (EBAA) webpage with Monkeypox Informational Alert
References
- ↑ Centers for Disease Control and Prevention (CDC). Poxviridae Infections. Public Health Image Library (PHIL) https://phil.cdc.gov/Details.aspx?pid=2329 Accessed January 9, 2022.
- ↑ 2.0 2.1 Di Giulio DB, Eckburg PB. Human monkeypox: an emerging zoonosis. Lancet Infect Dis. Jan 2004;4(1):15-25. doi:10.1016/s1473-3099(03)00856-9
- ↑ 3.0 3.1 3.2 Petersen E, Kantele A, Koopmans M, et al. Human monkeypox: epidemiologic and clinical characteristics, diagnosis, and prevention. Infect Dis Clin North Am. 2019;33(4):1027-1043.
- ↑ Hutson CL, Nakazawa YJ, Self J, et al. Laboratory investigations of African pouched rats (Cricetomys gambianus) as a potential reservoir host species for monkeypox virus. PLoS Negl Trop Dis. 2015;9(10):e0004013.
- ↑ 5.0 5.1 Falendysz EA, Lopera JG, Lorenzsonn F, et al. further assessment of monkeypox virus infection in gambian pouched rats (Cricetomys gambianus) using in vivo bioluminescent imaging. PLoS Negl Trop Dis. 2015;9(10):e0004130.
- ↑ 6.0 6.1 6.2 6.3 Kumar N, Acharya A, Gendelman HE, Byrareddy SN. The 2022 outbreak and the pathobiology of the monkeypox virus. J Autoimmun. 2022;131:102855.
- ↑ 7.0 7.1 7.2 7.3 7.4 7.5 7.6 7.7 Thornhill JP, Barkati S, Walmsley S, et al. Monkeypox virus infection in humans across 16 countries — 2022. New Eng J Med. 2022. Published Online Ahead of Print.
- ↑ 8.0 8.1 Likos AM, Sammons SA, Olson VA, et al. A tale of two clades: monkeypox viruses. J Gen Virol. 2005;86(Pt 10):[[1]].
- ↑ World Health Organization. Multi-country monkeypox outbreak: situation update. Accessed July 22, 2022, https://www.who.int/emergencies/disease-outbreak-news/item/2022-DON393.
- ↑ Reynolds MG, Davidson WB, Curns AT, et al. Spectrum of infection and risk factors for human monkeypox, United States, 2003. Emerg Infect Dis. 2007;13(9):1332-1339.
- ↑ 11.0 11.1 11.2 11.3 Guarner J, Del Rio C, Malani PN. Monkeypox in 2022 – what clinicians need to know. JAMA. 2022;328(2):139-140.
- ↑ 12.0 12.1 Cash-Goldwasser S, Labuda SM, McCormick DW, et al. Ocular Monkeypox — United States, July–September 2022. MMWR Morb Mortal Wkly Rep. 2022;71(42):1343-1347.
- ↑ Centers for Disease Control and Prevention. 2022 Monkeypox Outbreak Global Map. Accessed August 1, 2022, https://www.cdc.gov/poxvirus/monkeypox/response/2022/world-map.html.
- ↑ Bruno G, Fabrizio C, Rodano L, Buccoliero GB. Monkeypox in a 71-year-old woman. J Med Virol. 2022. Published online ahead of print.
- ↑ Tutu van Furth AM, van der Kuip M, van Els AL, et al. Paediatric monkeypox patient with unknown source of infection, the Netherlands, June 2022. Euro Surveill. 2022;27(29):2200552.
- ↑ 16.0 16.1 16.2 Bayer-Garner IB. Monkeypox virus: histologic, immunohistochemical and electron-microscopic findings. J Cutan Pathol. 2005;32(1):28-34.
- ↑ McCollum AM, Damon IK. Human monkeypox. Clin Infect Dis. 2014;58(2):260-267.
- ↑ 18.0 18.1 Rao AK, Petersen BW, Whitehill F, et al. Use of JYNNEOS (Smallpox and Monkeypox Vaccine, Live, Nonreplicating) for Preexposure Vaccination of Persons at Risk for Occupational Exposure to Orthopoxviruses: Recommendations of the Advisory Committee on Immunization Practices - United States, 2022. MMWR Morb Mortal Wkly Rep. 2022;71(22):734-742.
- ↑ 19.0 19.1 Xiang Y, White A. Monkeypox virus emerges from the shadow of its more infamous cousin: family biology matters. Emerg Microbes Infect. 2022;11(1):1768-1777.
- ↑ Administration for Strategic Preparedness and Response, JYNNEOS Monkeypox Vaccine Distribution by Jurisdiction. Accessed August 1, 2022, https://aspr.hhs.gov/SNS/Pages/JYNNEOS-Distribution.aspx.
- ↑ 21.0 21.1 21.2 21.3 21.4 Kaufman AR, Chodosh J, Pineda R. Monkeypox Virus and Ophthalmology—A Primer on the 2022 Monkeypox Outbreak and Monkeypox-Related Ophthalmic Disease. JAMA Ophthalmol. 2023;141(1):78–83. doi:10.1001/jamaophthalmol.2022.4567
- ↑ Cash-Goldwasser S, Labuda SM, McCormick DW, et al. Ocular Monkeypox — United States, July–September 2022. MMWR Morb Mortal Wkly Rep. 2022;71(42):1343-1347.
- ↑ Kaufman AR, Chodosh J, Pineda R. Monkeypox Virus and Ophthalmology—A Primer on the 2022 Monkeypox Outbreak and Monkeypox-Related Ophthalmic Disease. JAMA Ophthalmol. 2023;141(1):78–83. doi:10.1001/jamaophthalmol.2022.4567
- ↑ 24.0 24.1 Rayati Damavandi A, Semnani F, Hassanpour K. A Review of Monkeypox Ocular Manifestations and Complications: Insights for the 2022 Outbreak. Ophthalmol Ther. 2023 Feb;12(1):55-69. doi: 10.1007/s40123-022-00626-4. Epub 2022 Dec 13. PMID: 36512187; PMCID: PMC9834445.
- ↑ Abdelaal A, Serhan HA, Mahmoud MA, Rodriguez-Morales AJ, Sah R. Ophthalmic manifestations of monkeypox virus. Eye (Lond). 2023 Feb;37(3):383-385. doi: 10.1038/s41433-022-02195-z. Epub 2022 Jul 27. PMID: 35896700; PMCID: PMC9905463.
- ↑ 26.0 26.1 Jezek Z, Szczeniowski M, Paluku KM, Mutombo M. Human monkeypox: clinical features of 282 patients. J Infect Dis. 1987;156(2):293-298.
- ↑ 27.0 27.1 27.2 Rojas-Carabali W, Cifuentes-González C, Agrawal R, de-la-Torre A. Spectrum of ophthalmic manifestations in monkeypox virus infection worldwide: Systematic review and meta-analysis. Heliyon. 2023 Jul 22;9(8):e18561. doi: 10.1016/j.heliyon.2023.e18561. PMID: 37576249; PMCID: PMC10413003.
- ↑ 28.0 28.1 Carrubba S, Geevarghese A, Solli E, Guttha S, Sims J, Sperber L, Meehan S, Ostrovsky A. Novel severe oculocutaneous manifestations of human monkeypox virus infection and their historical analogues. Lancet Infect Dis. 2023 May;23(5):e190-e197. doi: 10.1016/S1473-3099(22)00869-6. Epub 2023 Jan 23. PMID: 36702137; PMCID: PMC9870321.
- ↑ Damon IK. Status of human monkeypox: clinical disease, epidemiology and research. Vaccine. 2011;29 Suppl 4:D54-D59.
- ↑ Jezek Z, Szczeniowski M, Paluku KM, Mutombo M. Human monkeypox: clinical features of 282 patients. J Infect Dis. 1987;156(2):293-298.
- ↑ Zong Y, Kamoi K, Zhang J, Yang M, Ohno-Matsui K. Mpox (Monkeypox) and the Eye: Ocular Manifestation, Diagnosis, Treatment and Vaccination. Viruses. 2023 Feb 23;15(3):616. doi: 10.3390/v15030616. PMID: 36992325; PMCID: PMC10054449.
- ↑ Foos W., Wroblewski K., Ittoop S. Subconjunctival nodule in a patient with acute monkeypox. JAMA Ophthalmol. 2022 doi: 10.1001/jamaophthalmol.2022.3742.
- ↑ Learned L.A., Reynolds M.G., Wassa D.W., Li Y., Olson V.A., Karem K., Stempora L.L., Braden Z.H., Kline R., Likos A., Libama F., Moudzeo H., Bolanda J.D., Tarangonia P., Boumandoki P., Formenty P., Harvey J.M., Damon I.K. Extended interhuman transmission of monkeypox in a hospital community in the Republic of the Congo. Am. J. Trop. Med. Hyg. 2005;73:428–434. 2003.
- ↑ Mbala K.P., Huggins W.J., Riu R.T., Mulembakani M.P., Ahuka M.S., Kumakamba M.C., Martin W.J., Muyembe T.J.J. The clinical characterization of human monkeypox infections in the Democratic Republic of Congo. Trop. Med. Int. Health. 2017;22:31. doi: 10.1111/(ISSN)1365-3156.
- ↑ 35.0 35.1 Nguyen MT, Mentreddy A, Schallhorn J, Chan M, Aung S, Doernberg SB, Babik J, Miles K, Yang K, Lydon E, Minter DJ, Gonzales J, Shantha J, Doan T, Seitzman GD. Isolated Ocular Mpox without Skin Lesions, United States. Emerg Infect Dis. 2023 Jun;29(6):1285-1288. doi: 10.3201/eid2906.230032. Epub 2023 May 2. PMID: 37130504; PMCID: PMC10202873.
- ↑ Alsarhani W.K., Chan C.C., Boyd S.R., Hasso M., Almeer A., Tan D.H.S. Monkeypox-associated disciform keratitis. Cornea. 2022 doi: 10.1097/ICO.0000000000003171
- ↑ Finamor L.P.S., de Freitas D., Andrade G., Bergamasco V.D., Cunha L., Lázari C., Muccioli C. Tecovirimat treatment for monkeypox virus keratouveitis. JAMA Ophthalmol. 2023;141:210–212. doi: 10.1001/jamaophthalmol.2022.5486.
- ↑ Lamas-Francis D., Treviño M., Pérez-Freixo H., Martínez-Perez L., Touriño R., Rodríguez-Ares T. Corneal ulcer due to monkeypox infection. Ocul. Immunol. Inflamm. 2022:1–3. doi: 10.1080/09273948.2022.2153343
- ↑ Androudi, S., Kaufman, A. R., Kouvalakis, A., Mitsios, A., Sapounas, S., Al-Khatib, D., … Baglivo, E. (2023). Non-Healing Corneal Ulcer and Uveitis Following Monkeypox Disease: Diagnostic and Therapeutic Challenges. Ocular Immunology and Inflammation, 32(3), 253–258. https://doi.org/10.1080/09273948.2023.2202746
- ↑ Carvalho E.M., Medeiros M., Veloso V.G., Biancardi A.L., Curi A.L.L. Monkeypox infection causing conjunctival vesicles and anterior uveitis. Ocul. Immunol. Inflamm. 2023:1–2. doi: 10.1080/09273948.2023.2174884
- ↑ 41.0 41.1 41.2 41.3 Petersen E, Kantele A, Koopmans M, et al. Human monkeypox: epidemiologic and clinical characteristics, diagnosis, and prevention. Infect Dis Clin North Am. 2019;33(4):1027-1043.
- ↑ 42.0 42.1 Xiang Y, White A. Monkeypox virus emerges from the shadow of its more infamous cousin: family biology matters. Emerg Microbes Infect. 2022;11(1):1768-1777.
- ↑ 43.0 43.1 43.2 Reynolds MG, McCollum AM, Nguete B, Shongo Lushima R, Petersen BW. Improving the care and treatment of monkeypox patients in low-resource settings: applying evidence from contemporary biomedical and smallpox biodefense research. Viruses. 2017;9(12):380.
- ↑ 44.0 44.1 44.2 Thornhill JP, Barkati S, Walmsley S, et al. Monkeypox virus infection in humans across 16 countries — 2022. New Eng J Med. 2022. Published Online Ahead of Print.
- ↑ Petersen E, Kantele A, Koopmans M, et al. Human monkeypox: epidemiologic and clinical characteristics, diagnosis, and prevention. Infect Dis Clin North Am. 2019;33(4):1027-1043.
- ↑ Centers for Disease Control and Prevention. What Healthcare Professionals Should Know. Accessed July 22, 2022. https://www.cdc.gov/poxvirus/monkeypox/clinicians/what-hcps-should-know.html.
- ↑ Centers for Disease Control and Prevention. Interim Clinical Considerations for Management of Ocular Monkeypox Virus Infection. Accessed December 12, 2022. https://www.cdc.gov/poxvirus/monkeypox/clinicians/ocular-infection.html.
- ↑ 48.0 48.1 Centers for Disease Control and Prevention. Treatment Information for Healthcare Professionals. Accessed July 30, 2022, https://www.cdc.gov/poxvirus/monkeypox/clinicians/treatment.html.
- ↑ 49.0 49.1 49.2 49.3 Reynolds MG, McCollum AM, Nguete B, Shongo Lushima R, Petersen BW. Improving the care and treatment of monkeypox patients in low-resource settings: applying evidence from contemporary biomedical and smallpox biodefense research. Viruses. 2017;9(12):380.
- ↑ Yu J, Raj SM. Efficacy of three key antiviral drugs used to treat orthopoxvirus infections: a systematic review. Global Biosecur. 2019;1(1).
- ↑ Croasdale C, Wise J, Holland E. Human monkeypox ocular infection: first Western Hemisphere case report. presented at: 2003 Federated Scientific Session of the Cornea Sociey and Eye Bank Association of America; 2003; Anaheim, California, United States.
- ↑ 52.0 52.1 Damon IK. Status of human monkeypox: clinical disease, epidemiology and research. Vaccine. 2011;29 Suppl 4:D54-D59.
- ↑ Bunge EM, Hoet B, Chen L, et al. The changing epidemiology of human monkeypox-A potential threat? A systematic review. PLoS Negl Trop Dis. 2022;16(2):e0010141.
- ↑ 54.0 54.1 Centers for Disease Control and Prevention. Isolation and Infection Control: Home. Accessed July 30, 2022, https://www.cdc.gov/poxvirus/monkeypox/clinicians/infection-control-home.html.
- ↑ 55.0 55.1 55.2 Centers for Disease Control and Prevention. Infection Prevention and Control of Monkeypox in Healthcare Settings. Accessed July 30, 2022, https://www.cdc.gov/poxvirus/monkeypox/clinicians/infection-control-healthcare.html.
- ↑ United States Environmental Protection Agency, What is an emerging viral pathogen claim?, Accessed August 1, 2022https://www.epa.gov/coronavirus/what-emerging-viral-pathogen-claim.
- ↑ Kawana R, Kitamura T, Nakagomi O, et al. Inactivation of human viruses by povidone-iodine in comparison with other antiseptics. Dermatology. 1997;195 Suppl 2:29-35.
- ↑ Eye Bank Association of America. Monkeypox Informational Alert: Monkeypox and Eye Tissue Donation. Accessed July 30, 2022, 2022. https://restoresight.org/news/monkeypox-informational-alert/?utm_source=rss&utm_medium=rss&utm_campaign=monkeypox-informational-alert.