Molluscum Contagiosum
All content on Eyewiki is protected by copyright law and the Terms of Service. This content may not be reproduced, copied, or put into any artificial intelligence program, including large language and generative AI models, without permission from the Academy.
Disease Entity
Disease
Molluscum contagiosum (MC) is a DNA virus that infects skin and mucous membranes, resulting in characteristic dome-shaped umbilicated nodules. Infection commonly produces nodules on the skin of the face, abdomen, legs, arms, neck, and genital area. [1] Ocular MC typically results in eyelid nodules, but can affect the conjunctiva through a hypersensitivity reaction leading to a chronic follicular conjunctivitis. Other ocular manifestations include corneal changes such as a superior micropannus and fine epithelial keratitis. [2]
Etiology
The Molluscum Contagiosum virus (MCV) is of the Poxviridae family, which is a family of enveloped, double stranded DNA. The virus is spread by direct person-to-person physical contact, which may occur through contact with an infected person or fomite. Patients with MC may also exhibit autoinoculation, in which the virus may spread to other parts of the body by touching a lesion, then touching the body elsewhere. [1] Additionally, there is a sexually transmitted variant.
Risk Factors
Patients of any age may become infected, though MC most commonly affects children ages 10 and younger. Individuals with increased risk include immunocompromised individuals (HIV-infected persons, patients receiving immunosuppression), patients with atopic dermatitis (believed to be a risk factor due to frequent breaks in the skin), and individuals who live in warm, humid climates and crowded living conditions. [1][3]
Pathophysiology
MCV has 4 different genotypes: MCV 1, MCV 2, MCV 3, and MCV 4, with MCV 1 being the most common genotype (75-96%). [3] MCV is transmitted by direct contact with infected skin or fomites. MCV infects the epidermis and replicates in the cytoplasm of cells, with a variable incubation period between 2-6 weeks. [4] [5] Lesions on the eyelid skin may shed viral particles into the tear film, resulting in ocular surface involvement. [2]
Diagnosis
History
The result of the infection is usually mild skin involvement characterized by lesions that may appear anywhere on the body. MC lesions are typically self-limiting and resolve within 6-12 months, however, the course is variable and may persist for more than 3-4 years.[1] [2] [3]
Physical examination
- Visual acuity with the best correction
- External examination of skin of the face, including eyelids, periocular skin, and scalp. Additional skin examination may be recommended if patients endorse lesions, specifically looking for elevated nodular lesions with central umbilication.
- Slit-lamp biomicroscopy of the conjunctiva, including eversion of the eyelids to examine superior and inferior palpebral conjunctiva, and examination of the anterior segment with special attention to any corneal staining, stromal opacities, keratic precipitates, and anterior chamber cell and flare.
Signs
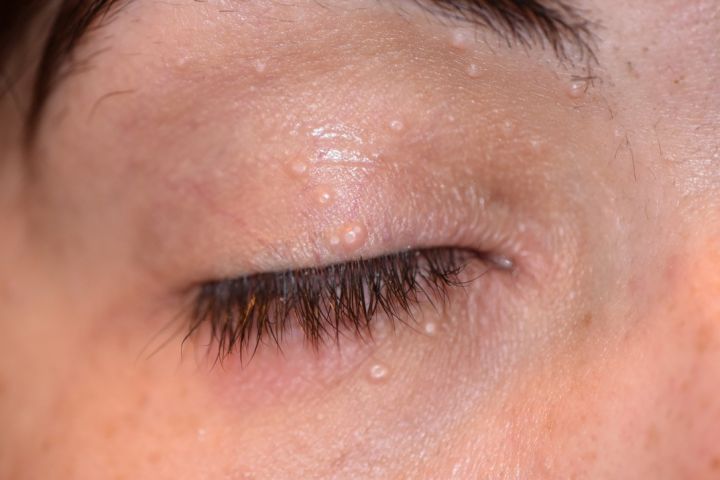
Skin lesions are characterized by firm rounded papules from 2 to 5 mm, pink or skin-colored, with a shiny umbilicated surface (Figures 1a & 1b). Lesions may be single, multiple or clustered, and occasionally they may have an erythematous halo or pedicle. [3]
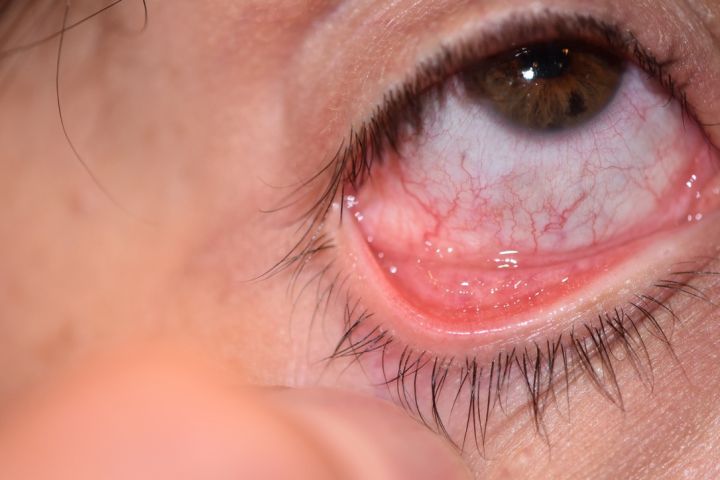
The eyelid is the most common site of ocular lesions although conjunctival lesions have been reported. Patients often present to the ophthalmologist with clusters of upper and lower eyelid lesions, or when a periocular lesion results in follicular conjunctivitis, as chronic conjunctivitis or keratoconjunctivitis can be associated with eyelid lesions. Keratoconjunctivitis associated with MC is generally chronic and features marked follicular conjunctivitis (Figure 2) along with punctate keratopathy and epithelial or subepithelial infiltrates [6] [7]
In children, the main affected areas are sites of exposed skin, such as the trunk, extremities, intertriginous regions, genitals, and face. Involvement of the palms and soles, and oral mucosa is atypical. In adults, lesions are most frequently located in the lower abdomen, thighs, genitals, and perianal area, with most cases being transmitted by sexual contact. In children, genital lesions are mainly due to autoinoculation and are not pathognomonic of sexual abuse. In immunosuppressed patients, such as those infected with HIV, lesions may be extensive and located in atypical sites. They may be greater than 1 cm in diameter (giant MC) and are often refractory to treatment. [2] [3]
Symptoms
MC skin lesions may be associated with pruritis and irritation. Keratoconjuctival involvement may present with symptoms of conjunctivitis, including redness, tearing, and irritation.
Clinical diagnosis
The diagnosis of MC can be made by history and clinical examination. Clinical diagnosis of ocular MC is based on the presence of characteristic lid lesions and follicular conjunctivitis. Diagnosis is usually straightforward when typical MC findings are seen; however, some irritated or excoriated cases of MC may be indistinguishable from other lesions, making definitive diagnosis difficult. [3][6]
Diagnostic procedures
Histopathological study may be indicated when the diagnosis is not clear. An excisional biopsy can be done to confirm the diagnosis. Histologically, MC is characterized by epidermal cells with large eosinophilic intracytoplasmic inclusion bodies known as Henderson-Paterson bodies or molluscum bodies. [2] [3]
Laboratory test
Laboratory testing is not routinely indicated, though evaluation for immunocompromised status, i.e. HIV-AIDS, should be considered in atypical cases. Clinical signs that may warrant further investigation include extensive lesions, larger or atypical appearing lesions in uncommon sites, bilateral extensive eyelid involvement, or lesions that are refractory to therapy.
Differential diagnosis
The MC differential diagnosis may include other infectious etiologies (verruca vulgaris, chickenpox, folliculitis, condyloma acuminatum), tumors (syringomas, steatocystoma multiplex, basal cell carcinoma), or other miscellaneous etiologies including acne vulgaris, Gianotti-Crosti Syndrome, milia, sebaceous hyperplasia, or papular uriticaria. [3] It is important to consider molluscum contagiosum on the differential of recurrent follicular conjunctivitis in addition to chlamydia and topical medication toxicity.
Management
General treatment
There is currently no Food and Drug Administration (FDA) approved treatment for MC, therefore, a standard of care has yet to be established. In healthy, immunocompetent patients, the disease is often self-limited, with treatment merely accelerating clearance. Treatment for cutaneous MC is generally indicated in patients with extensive disease, secondary complications (bacterial superinfection, molluscum dermatitis, conjunctivitis), or aesthetic complaints. [1] [3]
Medical and Surgical Therapy
Non-surgical therapies that are widely practiced for cutaneous MC include topical chemical agents, such as imiquimod, silver nitrate, and trichloroacetic acid, as well as oral cimetidine. [8] [9] [10][11] Mechanical methods may include cryotherapy, curettage, and pulse dye laser therapy which is suggested for refractory cases [1] [3] [12] In immunosuppressed patients with extensive or refractory disease, providers may consider the use of topical or intravenous cidofovir. [3] A case series of giant MC in HIV-positive patients demonstrated success using topical 20% potassium hydroxide solution. [13]
The recommended management of ocular MC and chronic anterior segment involvement is lesion excision, by unroofing and curettage, which usually leads to rapid and dramatic resolution of findings. .[7] Additionally, efficacy has been shown with cryotherapy, cauterization, and photodynamic therapy.[7][14] [15] The use of topical therapy, including steroids, antibiotics, or antivirals, is not uncommon, however these modalities are typically unnecessary and may cause injury to ocular surfaces. Clinical trials have not demonstrated efficacy for these practices. [6] [7] [8]
Prevention
For all patients, general measures are recommended to prevent the spread of MCV. It should be advised to follow good hygiene habits, including hand washing, and avoiding scratching or rubbing the lesions. Patients should not share towels, tubs, or bath utensils. It is advised to keep the lesions clean and covered with clothing or a bandage to prevent spread. [1]
Complications
Skin lesions caused by MC are usually benign and resolve without scarring, though scratching and scraping of lesions can induce scarring. The most common complication is secondary bacterial infection, which may be significant in immunocompromised patients. Thus, it is advised in these cases that treatment be pursued to prevent further spread. [1]
Prognosis
Recovery from MCV infection does not prevent future infection. Although MCV does not behave like herpes viruses, which may remain dormant and reactivate, reinfection with MCV may occur if contact with infected persons or objects is made. [1]
References
- ↑ 1.0 1.1 1.2 1.3 1.4 1.5 1.6 1.7 1.8 Molluscum Contagiosum. Center for Disease Control. www.cdc.gov.
- ↑ 2.0 2.1 2.2 2.3 2.4 Charteris DG, Bonshek RE, Tullo AB. Ophthalmic molluscum contagiosum: clinical and immunopathological features. Br J Ophthalmol. 1995;79:476–481.
- ↑ 3.00 3.01 3.02 3.03 3.04 3.05 3.06 3.07 3.08 3.09 3.10 Meza-Romero R, Navarrete-Dechent C, Downey C. “Molluscum Contagiosum: an update and review of new perspectives in etiology, diagnosis, and treatment” Clinical, Cosmetic and Investigational Dermatology 2019:12 373–381
- ↑ Braue A, Ross G, Varigos G, Kelly H. Epidemiology and impact of childhood molluscum contagiosum: a case series and critical review of the literature. Pediatr Dermatol. 2005;22(4):287–294. doi:10.1111/ j.1525-1470.2005.22401.x
- ↑ Leung AKC, Barankin B, Hon KLE. Molluscum contagiosum: an update. Recent Pat Inflamm Allergy Drug Discov. 2017;11(1):22–31. doi:10.2174/1872213X11666170518114456
- ↑ 6.0 6.1 6.2 Schornack MM, et al. Ocular manifestations of molluscum contagiosum. Clin Exp Optom 2006; 89: 390–393.
- ↑ 7.0 7.1 7.2 7.3 Ritterband DC, Friedberg DN. Virus infections of the eye. Rev Med Virol 1998; 8: 187–201.
- ↑ 8.0 8.1 Serin, Ş., Bozkurt Oflaz, A., Karabağlı, P., Gedik, Ş., & Bozkurt, B. (2017). Eyelid Molluscum Contagiosum Lesions in Two Patients with Unilateral Chronic Conjunctivitis. Turkish journal of ophthalmology, 47(4), 226–230. doi:10.4274/tjo.52138.
- ↑ A. Sadowska-Przytocka, M. Czarnecka-Operacz, D. Jenerowicz, A. Grzybowski. Ocular manifestations of infectious skin diseases. Clin Dermatol, 34 (2015), pp. 124-128.
- ↑ Nguyen HP, Franz E, Stiegel KR, Hsu S, Tyring SK. Treatment of molluscum contagiosum in adult, pediatric, and immunodeficient populations. J Cutan Med Surg (2014) 18(5):299–306.10.2310/7750.2013.13133.
- ↑ Skinner RB Jr. Treatment of molluscum contagiosum with imiquimod 5% cream. J Am Acad Dermatol. 2002;47(4 Suppl):S221–S224.
- ↑ Vakharia PP, Chopra R, Silverberg NB, Silverberg JI. Efficacy and safety of topical cantharidin treatment for molluscum contagiosum and warts: a systematic review. Am J Clin D
- ↑ Achdiat PA, Adriyani KM, Pospos AG, Rowawi R, Hindritiani R, Suwarsa O, Gunawan H. Two Cases of Giant Molluscum Contagiosum on the Eyelids of HIV Patients Successfully Treated with Adjuvant Self-Applied Topical 20% Potassium Hydroxide Solution. HIV AIDS (Auckl). 2021 Nov 20;13:993-997. doi: 10.2147/HIV.S341856. PMID: 34849035; PMCID: PMC8612666.
- ↑ Basdag H, Rainer BM, Cohen BA. Molluscum contagiosum: to treat or not to treat? Experience with 170 children in an outpatient clinic setting in the northeastern United States. Pediatr Dermatol. 2015;32 (3):353–357. doi:10.1111/pde.12504
- ↑ Leung AKC, Barankin B, Hon KLE. Molluscum contagiosum: an update. Recent Pat Inflamm Allergy Drug Discov. 2017;11(1):22–31. doi:10.2174/1872213X11666170518114456
- Horner ME, Abramson AK, Warren RB, Swanson S, Menter MA.The spectrum of oculocutaneous disease: Part I. Infectious, inflammatory, and genetic causes of oculocutaneous disease. J Am Acad Dermatol. 2014 May;70(5):795.e1-25.
- Moye V, Cathcart S, Burkhart CN, Morrell DS. Beetle juice: a guide for the use of cantharidin in the treatment of molluscum contagiosum. Dermatol Ther. 2013;26(6):445–451. doi:10.1111/dth.12105
- Ting PT, Dytoc MT. Therapy of external anogenital warts and molluscum contagiosum: a literature review. Dermatol Ther. 2004;17(1):68–101.