Interstitial Keratitis
All content on Eyewiki is protected by copyright law and the Terms of Service. This content may not be reproduced, copied, or put into any artificial intelligence program, including large language and generative AI models, without permission from the Academy.
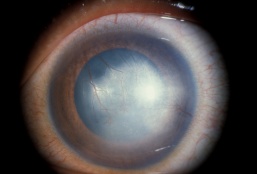
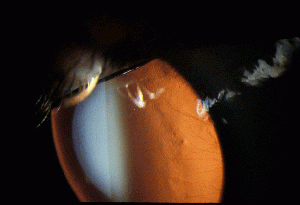
Defined narrowly, interstitial keratitis is any non-ulcerating inflammation of the corneal stroma without the involvement of either the epithelium or endothelium. Practically, however, the term refers to a common endpoint for a number of diseases which primarily manifest as inflammation and vascularization of the corneal stroma with minimal loss of tissue. Diseases that also involve other layers of the cornea secondarily are also included as long as the stroma is the predominant layer affected. The underlying causes of interstitial keratitis can broadly be either infectious or immune-mediated. The stroma constitutes the greatest thickness of all of the layers of the cornea and is made up of specially arranged collagen fibrils that allow clear transmission of light. The inflammation and blood vessel invasion characteristic of interstitial keratitis can result in scarring of this layer resulting in decreased vision.
Disease Entity
Interstitial Keratitis (ICD-9 #370.50)
Disease
Interstitial keratitis is a non-ulcerating inflammation of the corneal stroma.
Etiology
The most common etiologies of interstitial keratitis in the United States are Herpes Simplex Virus and Syphilis. Other associated bacterial diseases include Lyme disease, Tuberculosis, Leprosy, Brucellosis, and Leptospirosis. Viral etiologies include the other Herpes viruses including Herpes Zoster and Epstein-Barr, as well as HLTV-1, Mumps, and Measles. Onchocerciasis, Trypanosomiasis, and Microsporidiosis are important considerations in developing countries while Acanthamoeba may be seen more commonly in developed countries. Finally, diseases thought to be immune-mediated in nature including Cogan syndrome, Sarcoidosis, Mycosis Fungoides, and contact lens-associated keratitis are in the differential diagnosis.
- Bacterial etiologies: Syphilis, Lyme Disease, Tuberculosis, Leprosy, Brucellosis, Leptospirosis
- Viral etiologies: Herpesviridae (Herpes simplex, Herpes Zoster, Epstein- Barr), HLTV-1, Mumps, Measles, Vaccinia, Variola
- Parasitic etiologies: Onchocerciasis, Acanthamoeba, Leishmaniasis, Trypanosomiasis, Microsporidiosis
- Immune etiologies: Cogan syndrome, sarcoidosis, mycosis fungoides, contact lens-associated keratitis, intracorneal foreign body, heavy metals, autoimmune disease, e.g., rheumatoid arthritis, granulomatosis with polyangiitis (formerly known Wegener's)
The overall incidence of interstitial keratitis is rare, responsible for only 3% of all keratoplasties performed in the United States. Together, Herpetic and Syphilitic disease are responsible for greater than 50% of cases of interstitial keratitis in the United States.
Risk Factors
The risk factors for interstitial keratitis vary according to the underlying etiology.
General Pathology
Intralamellar infiltration of lymphocytes with extensive neovascularization in the absence of overlying ulceration. The endothelium is unaffected in the early stages but may experience decompensation later in the course along with concomitant findings of stromal and epithelial edema. Other late findings include ghost vessel formation and corneal scarring.
Pathophysiology
The pathophysiology of interstitial keratitis is unknown but varies significantly by etiology. The majority of bacterial and parasitic interstitial keratitis is a result of direct invasion of the corneal stroma and the host's allergic/immune reaction to foreign antigens. The reaction is similar in viral interstitial keratitis with the identification of viral antigens in the corneal stroma, but whether this represents active infection is less clear.
Diagnosis
The purpose of the comprehensive evaluation of a patient with interstitial keratitis is to assess the degree and location of active inflammation in the cornea and elsewhere in the eye, as well as to determine the underlying etiology of the disease. The comprehensive evaluation (history and physical) includes those components of the comprehensive medical eye evaluation specifically relevant to the diagnosis and treatment of interstitial keratitis as noted below.
History
A comprehensive medical and ocular history including the previous history of herpetic infections, other previous infections, an exposure history to insect bites, animals, environment, a travel history to endemic regions of disease, sexually transmitted diseases, trauma, contact lens wear, medication use, and constitutional symptoms including fever, hearing loss, shortness of breath.
Physical examination
A thorough and complete eye exam should be performed on any patient suspected of having interstitial keratitis. The general health of the eye should be assessed, and appropriate ancillary tests should be done to assess corneal curvature, astigmatism, and thickness.
- Visual acuity with and without current correction at distance and when appropriate at near
- Measurement of best-corrected visual acuity
- Ultrasound pachymetry
- External examination (lids, lashes, lacrimal apparatus, orbit)
- Examination of ocular alignment and motility
- Assessment of pupillary function
- Measurement of intraocular pressure (IOP)
- Slit-lamp biomicroscopy of the anterior segment with specific reference for signs of previous inflammation, active ulceration, or epithelial defect
- Dilated examination of the eye for the assessment of lens, vitreous, optic nerve, macula, and peripheral retina
- Assessment of relevant aspects of the patient’s mental and physical status
Signs
- Conjunctival injection
- Corneal haze: diffuse, sectoral, central, or circumferential
- White cell infiltration without significant necrosis or suppuration
- Stromal neovascularization
- Ghost vessels when the disease is quiescent
- Lipid keratopathy
Symptoms
Decreased vision highly dependent on the extent and location of involvement. Significant photophobia and pain are highly characteristic.
Clinical diagnosis
Interstitial keratitis is a clinical diagnosis based on the finding of non-ulcerative, stromal keratitis with deep stromal neovascularization.
Diagnostic procedures
- Slit-lamp biomicroscopy to determine the depth and pattern of stromal inflammation and associated ocular and corneal abnormalities
- Corneal pachymetry and intraocular pressure measurement
- Confocal microscopy in suspected cases of atypical infectious keratitis, primarily fungal and acanthamoeba related
Laboratory test
Ancillary testing should be directed by the history and physical exam. While syphilis is becoming a less common etiology for infectious keratitis, it still represents a significant number of cases and is easily treated, making the inclusion of an RPR/VDRL + FTA-ABS/ MHA-TP or equivalent tests worthwhile. Other bacterial etiologies are rare causes of interstitial keratitis, and testing for them should be based on specific findings either in the history or physical exam. Contact lens-associated keratitis may be either immune-mediated or infectious. Immune-mediated contact lens-associated interstitial keratitis is usually self-limiting. If it does not resolve as expected, confocal microscopy or corneal culture/histology should be considered. Other parasitic infections may be identified through isolation or identification from other bodily fluids. Serologic testing for most viral etiologies is often unhelpful given the widespread exposure of the vast majority of the population to these pathogens. Acute and convalescent titers and other specific tests may have some value if the diagnosis is uncertain or the resolution is delayed.
Cogan syndrome represents a special indication for audiology testing (50% experience irreversible deafness) with follow-up MRI scans of the inner ear looking for vestibular or cochlear inflammation if indicated. This disorder is most common in young adults and should be suspected if any hearing deficits are noted by history in a patient with mild peripheral interstitial keratitis. Ocular signs usually begin with sectoral conjunctival injection followed by subepithelial, peripheral keratitis. The interstitial keratitis which subsequently develops is described as anterior stromal or superficial. They may also manifest findings of systemic inflammation including Takayasu's arteritis, myalgia, polyarthralgia, and be found in conjunction with other autoimmune diseases. Circulating autoantibodies against both the inner ear and cornea have been identified although the correlation with disease and treatment is variable.
Differential diagnosis
- Entities which cause corneal stromal clouding where the source is not primarily stromal inflammation
- Acute ulcerative keratitis either infectious (bacterial, fungal, parasitic, viral) or immune
- Endothelial disease resulting in secondary corneal edema or stromal haze
- Traumatic corneal scarring
Management
The primary goals in the treatment of interstitial keratitis are to control local inflammation to prevent pain and visually significant scarring and to identify an underlying cause to reduce systemic sequelae of the disease.
General treatment
For many common forms of interstitial keratitis, corneal inflammation represents primarily an immune response rather than active infection. Therefore, treatment of the corneal manifestations may often occur independently or in parallel to the treatment of its underlying systemic cause. Prominent exceptions are rare forms of interstitial keratitis in infectious diseases as are seen in Lyme disease, mycobacterial infections, certain bacterial pathogens, as well as most parasitic diseases, where definitive therapy of the underlying disease is integral to the safe resolution of the corneal infection.
As an example, Syphilitic interstitial keratitis is a result of congenital syphilis in 90% of cases and usually occurs in the pre-teen or early teen years manifesting in the majority of patients as a bilateral, painful, visually limiting keratitis. If left untreated, photophobia is the most striking symptom often confining the patient to dark environments until its spontaneous resolution months to years. Whether the syphilis is congenital or acquired, treatment of the systemic disease will have little effect on the corneal inflammation but should be vigorously pursued to limit systemic complications.
Medical therapy
Topical corticosteroids are the mainstay of therapy for the most common forms of interstitial keratitis and are effective both for alleviating acute symptoms of pain, discomfort and blurred vision and for reducing scarring and neovascularization. They are the primary therapy for syphilitic interstitial keratitis, most immune forms of interstitial keratitis, and are used either in conjunction with antivirals (Herpes Simplex, Herpes Zoster) or alone in the viral-mediated interstitial keratitis. Systemic corticosteroids are often required in the treatment of systemic manifestations of sarcoidosis, and their prompt use is critical in avoiding permanent loss of hearing inability.
Corticosteroids have a role in the local therapy of most forms of bacteria-mediated interstitial keratitis, but all will require specific systemic antibiotics to cure the underlying disease. With the exception of syphilis, all of the other forms of bacteria-mediated interstitial keratitis benefit from the treatment of the underlying systemic infection.
Similarly, most viral-mediated interstitial keratitis will respond briskly to the addition of topical corticosteroids, but may benefit from the adjunctive use of topical or systemic anti-virals, specifically trifluridine in Herpes Simplex-mediated interstitial keratitis or acyclovir/valacyclovir in Herpes simplex and Herpes Zoster. The HEDS study specifically concluded that treatment of 'Herpes Simplex Virus-mediated interstitial keratitis resulted in less persistence/progression of the disease and faster recovery than placebo, but noted no difference in visual outcome or recurrence of disease at 6 months [1]. The addition of oral acyclovir vs. placebo in patients already on topical corticosteroids and trifluridine showed no improvement in resolution time or final visual outcome but did result in more patients experiencing visual improvement at 6 months follow-up [2]. Prophylactic treatment of Herpes Simplex Virus stromal keratitis patients with 400mg of Acyclovir twice daily, however, resulted in a statistically significant reduction of ocular (14% treated vs. 28% placebo) and orofacial recurrences during a 12-month treatment and 6-month follow-up [3]
Parasitic forms of interstitial keratitis require appropriate topical (Acanthamoeba) or specific systemic anti-parasitic therapy. The use of corticosteroids is controversial in Acanthamoeba keratitis (Dart) and is not routinely indicated in any of the other parasite-mediated interstitial keratitides.
Onchocerciasis manifests as centripetal sclerosing keratitis from the limbal migration of a filarial parasite, Onchocerca volvulus. Although there is a direct invasion of the parasite, the blinding interstitial keratitis exacerbated with the effective treatment and death of the parasite is thought to be due to an immune reaction to the release of its endosymbiont Wolbachia sp., suggesting that specific therapy against this bacteria may reduce the corneal complications.
Medical follow up
Patients treated for interstitial keratitis should have their corticosteroids reduced to the lowest effective dosage based on a lack of active inflammation or neovascularization on clinical examination. Patients should also be monitored for side effects of corticosteroid therapy, including steroid-induced elevation of intraocular pressure and cataract formation. With the exception of syphilitic interstitial keratitis, other bacterial, parasitic, and some viral interstitial keratitis should be monitored for response to systemic antibiotic therapy.
Surgery
With the exception of Acanthamoeba, the etiology of interstitial keratitis is either a systemic immune process, a systemic infection, or, in the case of some Herpesviridae, a regional infection not restricted to the cornea. Acute surgical intervention in the form of therapeutic keratoplasty would not be expected to be curative, and since, by definition, interstitial keratitis is largely non-ulcerative, and the need for urgent tectonic keratoplasty in its treatment is limited. Optical keratoplasty does have a role once the underlying disease has been effectively controlled or treated. These patients are at elevated risk of rejection because of the history of inflammation, as well as the concomitant neovascularization that is characteristic of most forms of interstitial keratitis.
Surgical follow-up
Routine follow-up is essential in patients who undergo keratoplasty to ensure proper wound healing, prompt and appropriate removal of sutures, and to monitor for sequelae of the surgical procedure or post-operative medications including glaucoma, cataract formation, and secondary infection. Special attention in these higher-risk patients is required to monitor for rejection and recurrence of previous inflammation, especially if the original cause of interstitial keratitis remains uncertain. Visual rehabilitation in the form of spectacle, contact lens fitting, or refractive surgery is likely in most post-keratoplasty patients.
Complications
See surgical follow-up.
Prognosis
The prognosis for interstitial keratitis is good if recognized and treated early before significant corneal scarring has occurred. Because of the lack of ulceration, even with mild loss of tissue, patients can often see reasonably well after resolution of the keratitis. The prognosis for patients requiring keratoplasty is more guarded.
References
- ↑ Wilhelmus KR, Gee L, Hauck WW, et al. Herpetic eye disease study. A controlled trial of topical corticosteroids for herpes simplex stromal keratitis. Ophthalmology 1994; 101:1883–1895; discussion 95–96.
- ↑ Barron BA, Gee L, Hauck WW, et al:Herpetic eye disease study. A controlled trial of oral acyclovir for herpes simplex stromal keratitis. Ophthalmology 1994; 101:1871–1882.
- ↑ Herpetic Eye Disease Study Group. Acyclovir for the prevention of recurrent herpes simplex virus eye disease. N Engl J Med. 1998 Jul 30;339(5):300-6.
- Pepose JS, Margolis TP, LaRussa P, Pavan-Langston D.Ocular complications of smallpox vaccination. Am J Ophthalmol. 2003 Aug;136(2):343-52.
- Apple DJ and Rabb MF. Ocular Pathology. Clinical Applications and Self-Assessment. 4th ed. 1991.
- Tu EY. Interstitial Keratitis in Principles and Practice of Ophthalmology 3e, Jakobiec ed 2007.
- Tu EY, Joslin CE, Sugar J, Booton GC, Shoff ME, Fuerst PA. The Relative Value of Confocal Microscopy and Superficial Corneal Scrapings in the Diagnosis of Acanthamoeba Keratitis. Cornea 2008;27(7):764-772.
- Schwartz, G. S., A. R. Harrison, et al. (1998). "Etiology of immune stromal (interstitial) keratitis." Cornea 17(3): 278-81.
- Dart JK, Saw VP, Kilvington S. Acanthamoeba keratitis: diagnosis and treatment update 2009 Am J Ophthalmol. 2009, Oct;148(4):487-499
- Kiss S, Damico FM, Young LH: Ocular manifestations and treatment of syphilis. Semin Ophthalmol 2005; 20:161–167.
- Knox CM, Holsclaw DS: Interstitial keratitis.Int Ophthalmol Clin 1998; 38:183–195.
- Orsoni JG, Zavota L, Vincenti V, et al: Cogan syndrome in children: early diagnosis and treatment is critical to prognosis. Am JOphthalmol 2004; 137:757–758.
- Lennarson P, Barney NP. Interstitial keratitis as presenting ophthalmic sign of sarcoidosis in a child. J Pediatr Ophthalmol Strabismus 1995; 32:194–196.
- Pearlman EaL, JH. Keratitis due to Onchocerciasis. Ophthalmol Clin 1994;7:641–648.
- Liesegang TJ. Corneal complications from herpes zoster ophthalmicus. Ophthalmology 1985; 92:316–324.
- Matoba AY, Wilhelmus KR, Jones DB. Epstein-Barr viral stromal keratitis.Ophthalmology 1986; 93:746–751.
- Herpetic Eye Disease Study Group. Oral acyclovir for herpes simplex virus eye disease: effect on prevention of epithelial keratitis and stromal keratitis. Arch Ophthalmol 2000; 118:1030–1036.