Incision Construction
All content on Eyewiki is protected by copyright law and the Terms of Service. This content may not be reproduced, copied, or put into any artificial intelligence program, including large language and generative AI models, without permission from the Academy.
Introduction
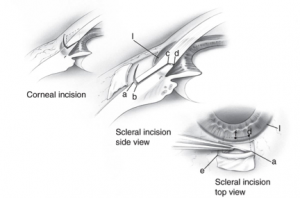
Cataract incision construction is of utmost importance in phacoemulsification cataract surgery. The initial wound construction influences the fluidic balance of the anterior segment, lays the foundation for all additional steps of the surgery, and plays an important role in the immediate post-operative period when the wound is most unstable[1]. Additionally, the creation of a watertight, self-sealing wound helps to prevent subsequent infections[2][3]. Conversely, a poorly constructed wound increases the risk of surgical and post-operative complications[4][5]. A properly executed phaco incision allows for a smoother operation, faster recovery, and improved visual outcomes for the patient. Various surgical approaches to wound construction have been utilized in cataract surgery (Figure 1a-b), with scleral and corneal incisions being the two main methods[6]. The purpose of this article is to review the advantages and disadvantages of scleral versus corneal incisions in cataract surgery, and offer recommendations on wound construction based on available literature.
Paracentesis[7]
The purpose of creating a paracentesis is two-fold: (1) to provide entry access for anesthetic agents or viscoelastic material into the anterior chamber, and (2) to accommodate a bimanual technique of phacoemulsification.
The paracentesis incision should be appropriately placed away from the main phaco incision to provide an optimal angle for intraocular manipulation of instruments.
- The incision should be made radially towards the center of the eye, and include slight nicking of the surrounding limbal vessels, which will help seal and strengthen the healing incision.
Scleral vs. clear corneal incision
Clear corneal incision (CCI) has become the preferred method of wound construction in modern-day cataract surgery due to the several advantages that it confers over the traditional scleral method. A survey of the members of the American Society of Cataract and Refractive Surgery (ASCRS) in 2003 showed that CCI was utilized by 72% of the respondents, compared to 47% in 2000, 12.4% in 1995, and 1.5% in 1992[8]. The advantages of CCI include a self-sealing, sutureless wound, shorter procedure time, faster recovery, intraoperative wound stability, and decreased risk of bleeding due to minimal conjunctival manipulation compared to scleral tunnel incisions[9][10].
However, CCI is not without its limitations. Higher rates of post-operative endophthalmitis, wound leaking, increased loss of endothelial cells, and surgically-induced astigmatism (SIA) with CCI compared to scleral incision have been reported[6][10][11][12][13][14][15][16]. Although infrequent, post-operative endophthalmitis remains one of the most devastating concerns due to its substantial morbidity related to vision loss[17][18]. CCI has been well-studied to be one of the major risk factors for post-operative endophthalmitis, with CCI having a 3-5.88-fold increase of endophthalmitis compared to scleral tunnel incision[11][17][19]. Studies have suggested this risk to be related to post-operative wound defects such as wound leaks and loss of wound apposition, leading to the ingress of extraocular, bacterial-contaminated fluid into the anterior chamber and causing subsequent infection[11][20][21][22]. A laboratory study even demonstrated permeation of India Ink into the anterior chamber through a sutureless CCI in cadaveric globes[23], which supports the role of wound leak and poor wound closure in CCI-related endophthalmitis. This concern has led to the employment of various techniques to improve wound leaking and closure including stromal hydration, sutures, adhesives, bandages, and femtosecond laser[14][15][24][25]. However, an alternative explanation has also suggested that this reported increase in post-operative endophthalmitis might correlate with the paralleled transition from scleral tunnel incision to CCI since its introduction in 1992[26][27].
In addition to the increased risk of post-operative endophthalmitis, higher rates of SIA with CCI have been reported due to the closer proximity of CCI to the center of the cornea compared to scleral incisions[6][12][13]. The magnitude of SIA at different post-operative timepoints has varied in studies, ranging from 0.72-2.69D with CCI to 0.36-1.85D with scleral incision with 3.5-6.0 mm incision sizes[12][13]. The incision size, degree of pre-operative astigmatism, and chosen axis of incision also influences the degree of post-operative astigmatism, with 3.5 mm wounds[28]. Modifications to the CCI technique, such as axis, location, length, width, and shape of the incision, have been studied and recommended to reduce such risk of SIA[29][30][31]. However, SIA with CCI still occurs and remains one of the limitations compared to scleral tunnel incision. Case reports of corneal erosion, wound dehiscence following trivial trauma, and increased risk of Descemet membrane detachment with ragged CCI have also been reported[32][33][34][35][36]. Despite its disadvantages, CCI remains a popular method of wound construction due to the overall advantages it confers for the surgeon during operation and the patient during recovery. Table 1 summarizes the comparison between CCI and scleral tunnel incision.
| Table 1 | Advantages | Disadvantages |
|---|---|---|
| Scleral tunnel incision | - Lower rates of endophthalmitis compared to CCI
- Utilized in manual small incision cataract surgery (MSICS), which is more cost-effective than phacoemulsification in developing countries[37][38] - Preferred method in microcornea[39] -Preferred method in cases with low endothelial count[8] |
- Requires conjunctival incision, which increases the risk of bleeding[27]
- Slower visual recovery - Hyphema from scleral vessels[27] - Oarlocking with tight scleral incisions[27] |
| Clear corneal incision (CCI) | - Self-sealing, sutureless wound
- Shorter procedure time - Faster visual recovery - Can be used as a refractive tool for pre-existing astigmatism[40][41] - Can be used with topical anesthesia[42] - Wound stability during surgery - Decreased risk of bleeding due to minimal to no conjunctival manipulation - Lower incidence of hyphema, which is more favorable for patients on anticoagulants[42] |
- Higher rates of endophthalmitis
- Higher rates of regular and irregular surgically-induced astigmatism - Wound leaking - Increased loss of endothelial cells and endothelial gap - Wound dehiscence following trivial trauma - Induction of irregular astigmatism - Descemet membrane detachment with ragged CCI - Corneal erosion - Hypotony[42] |
Cataract incision construction
Though CCI remains the preferred method in cataract surgery, there are various configurations in the method itself. This includes the axis, location, size, shape, form, and architecture of the incision, which can affect the degree of SIA, wound strength, and healing[28][43]. No configuration has been determined to be completely safe. As a result, the benefits and limitations of each configuration should be considered when constructing the corneal incision. Attention to individual cases, including the presence of pre-operative astigmatism, should also be considered[42]. While an astigmatically-neutral incision should be used if there is no pre-operative astigmatism, an astigmatic-inducing incision should be used if there is pre-operative astigmatism[6].
Location
The position of the corneal incision influences the degree of SIA due to the differing distances of each location to the central visual axis[44]. Common incision positions include temporal, superior, and nasal.
- Temporal incisions induce a lower degree of SIA than superior ones due to the closer proximity of the superior limbus to the central visual axis, and offer better surgical accessibility for the surgeon due to decreased brow obstruction[44].
- Temporal and nasal incisions have comparable corneal and astigmatic changes 8-weeks post-operatively, though the changes in the nasal approach are greater in the early postoperative period. As a result, temporal vs. nasal approach can be selected based on surgeon preference[45].
The presence of pre-existing astigmatism also influences the position and number of corneal incisions used[42][46].
- Superior incision is recommended for with-the-rule astigmatism (>1.5 D) and steep axis at 90 degrees.
- Temporal incision is recommended for:
- <1.5 D and steep axis at 90 degrees,
- negligible astigmatism, or
- against-the-rule astigmatism <0.75 D and steep axis at 180 degrees.
- Nasal incision is recommended for >0.75 D of astigmatism and steep axis at 180 degrees.
- Paired opposite CCI at the temporal and nasal positions can reduce pre-existing corneal astigmatism (>1.5 D) compared to a single CCI[47].
While the meridian is used to determine the position of the incision, landmarks such as the terminal vessels in the limbal arcade and CCI distances anterior to the limbus are used to determine the precise location of the initial incision[46]. CCI distances ranging from 0.5-1.5 mm anterior to the limbus have been found to be safe and effective in phacoemulsification[48].
- The incision should include slight nicking of the surrounding limbal blood vessels, which will help seal and strengthen the healing incision. A vascular, near-clear incision is preferred over a non-vascular, true clear incision, as a non-vascular incision can lead to a weaker wound that requires a longer healing time due to a delayed fibroblastic response compared to vascular incisions[49].
Size
Incision width: The width of the phaco incision should be matched to the gauge of the phaco probe, the irrigation/aspiration tip, and the IOL injector. Foldable IOLs have allowed for smaller incisions, which reduce the risk of SIA. However, larger IOLs may require larger incisions at the expense of increased SIA[42].
- An incision width of 1.8-2.2 mm has relatively small SIA with no difference in visual acuity or corneal astigmatism[50].
- Decreasing incision size from 3.2 mm to 2.2 mm or 1.8 mm reduces astigmatic effects, but further reduction from 2.2 mm to 1.8 mm has limited benefit[51][52].
- Enlarging the incision width to 4.5-5.0 mm reduces pre-existing against-the-rule astigmatism[41].
Incision length (tunnel length): Shorter-length incisions (<1.75 mm)[53] induce smaller SIA compared to longer-length incisions (>1.75 mm)[53] of same width and localization[53][54][55].
Shape and Form
The incision shape describes the internal incision or tunnel shape, while the form describes the depth of the incision[42].
- CCI with a square or near-square shape has greater stability (absent hypotony or wound leakage) than a rectangular one[49][56].
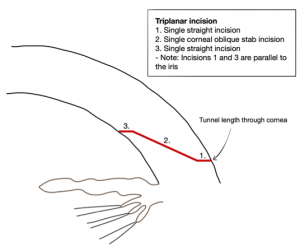
Architecture
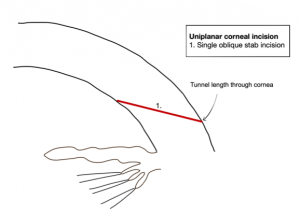
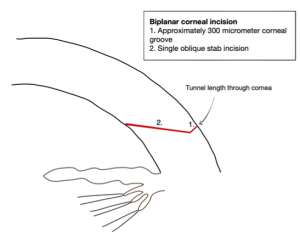
The incision architecture describes the number of incision planes that are used to construct the wound (Figure 2a-c). Uniplanar vs. biplanar vs. multiplanar (e.g triplanar) incisions have their own distinct advantages and disadvantages. Regardless of the incision architecture that is chosen, all should produce a well-balanced, watertight, and stable incision.
- Multiplanar incisions maintain better wound closure than uniplanar or biplanar incisions. This decreases the risk of the ingress of bacterial-contaminated fluid, and subsequent post-operative endophthalmitis[1][57].
Axis
The chosen incision axis also influences the degree of SIA. The ideal corneal incision will be at the axis of patient’s astigmatism to reduce post-operative astigmatism in conjunction with IOL choice.
- Incision at the steepest meridian reduces post-operative astigmatism in patients with pre-operative corneal astigmatism >0.50 D[30][40].
Alternative approaches
Femtosecond lasers are a popular integration into many parts of ocular surgery, ranging from the creation of the main corneal incision to lens disassembly (Figure 3). There is growing literature suggesting that femtosecond-assisted wound creation may result in wounds with better morphology and integrity than manual CCIs, thereby enhancing safety and outcomes[1][58]. The reverse side-cut CCI, which is associated with decreased wound leakage, is also unique to the femtosecond laser technique[1]. There is no definitive literature, yet, suggesting this as a mainstay modality. Femtosecond lasers are also an additional expense during surgery, so a cost-benefit analysis should be undertaken at this stage between individual surgeons[59].
Videos:
Video Credit: Uday Devgan MD - cataractcoach.com (Permission granted)
References
- ↑ 1.0 1.1 1.2 1.3 Donnenfeld E, Rosenberg E, Boozan H, Davis Z, Nattis A. Randomized prospective evaluation of the wound integrity of primary clear corneal incisions made with a femtosecond laser versus a manual keratome. J Cataract Refract Surg. 2018;44(3):329-335. doi:10.1016/j.jcrs.2017.12.026.
- ↑ Elkady B, Piñero D, Alió JL. Corneal incision quality: microincision cataract surgery versus microcoaxial phacoemulsification. J Cataract Refract Surg. 2009;35(3):466-474. doi:10.1016/j.jcrs.2008.11.047.
- ↑ Sykakis E, Karim R, Kinsella M, Bhogal M, Patel S, Parmar DN. Study of fluid ingress through clear corneal incisions following phacoemulsification with or without the use of a hydrogel ocular bandage: a prospective comparative randomised study. Acta Ophthalmol. 2014;92(8):e663-e666. doi:10.1111/aos.12436.
- ↑ Rodrigues R, Santos MSD, Silver RE, Campos M, Gomes RL. Corneal incision architecture: VICTUS femtosecond laser vs manual keratome. Clin Ophthalmol. 2019;13:147-152. Published 2019 Jan 10. doi:10.2147/OPTH.S181144.
- ↑ Teixeira A, Salaroli C, Filho FR, et al. Architectural analysis of clear corneal incision techniques in cataract surgery using Fourier-domain OCT. Ophthalmic Surg Lasers Imaging. 2012;43(6 Suppl):S103-S108. doi:10.3928/15428877-20121003-02.
- ↑ 6.0 6.1 6.2 6.3 Kohnen T. Corneal shape changes and astigmatic aspects of scleral and corneal tunnel incisions. J Cataract Refract Surg. 1997;23(3):301-302. doi:10.1016/s0886-3350(97)80168-x.
- ↑ Gupta R. (2017) The Paracentesis Incision. In: Phacoemulsification Cataract Surgery. Springer, Cham. https://doi.org/10.1007/978-3-319-59924-3_13.
- ↑ 8.0 8.1 Leaming DV. Practice styles and preferences of ASCRS members--2003 survey. J Cataract Refract Surg. 2004;30(4):892-900. doi:10.1016/j.jcrs.2004.02.064.
- ↑ Al Mahmood AM, Al-Swailem SA, Behrens A. Clear corneal incision in cataract surgery. Middle East Afr J Ophthalmol. 2014;21(1):25-31. doi:10.4103/0974-9233.124084.
- ↑ 10.0 10.1 Ho FL, Salowi MA, Bastion MC. The Effect of Eye Patching on Clear Corneal Incision Architecture in Phacoemulsification: A Randomized Controlled Trial. Asia Pac J Ophthalmol (Phila). 2017;6(5):429-434. doi:10.22608/APO.2016198.
- ↑ 11.0 11.1 11.2 Cooper BA, Holekamp NM, Bohigian G, Thompson PA. Case-control study of endophthalmitis after cataract surgery comparing scleral tunnel and clear corneal wounds. Am J Ophthalmol. 2003;136(2):300-305. doi:10.1016/s0002-9394(03)00202-2.
- ↑ 12.0 12.1 12.2 Olsen T, Dam-Johansen M, Bek T, Hjortdal JO. Corneal versus scleral tunnel incision in cataract surgery: a randomized study. J Cataract Refract Surg. 1997;23(3):337-341. doi:10.1016/s0886-3350(97)80176-9.
- ↑ 13.0 13.1 13.2 Archana S, Khurana AK, Chawla U. A comparative study of sclero-corneal and clear corneal tunnel incision in manual small-incision cataract surgery. Nepal J Ophthalmol. 2011;3(1):19-22. doi:10.3126/nepjoph.v3i1.4273.
- ↑ 14.0 14.1 Matossian C, Makari S, Potvin R. Cataract surgery and methods of wound closure: a review. Clin Ophthalmol. 2015;9:921-928. Published 2015 May 22. doi:10.2147/OPTH.S83552.
- ↑ 15.0 15.1 Masket S, Hovanesian JA, Levenson J, et al. Hydrogel sealant versus sutures to prevent fluid egress after cataract surgery. J Cataract Refract Surg. 2014;40(12):2057-2066. doi:10.1016/j.jcrs.2014.03.034.
- ↑ Jin KH, Kim TG. Relationship between early structural changes at cornea incision sites and surgical outcomes after phacoemulsification. Int J Ophthalmol. 2019;12(7):1139-1145. Published 2019 Jul 18. doi:10.18240/ijo.2019.07.14.
- ↑ 17.0 17.1 Cao H, Zhang L, Li L, Lo S. Risk factors for acute endophthalmitis following cataract surgery: a systematic review and meta-analysis. PLoS One. 2013;8(8):e71731. Published 2013 Aug 26. doi:10.1371/journal.pone.0071731.
- ↑ Safneck JR. Endophthalmitis: A review of recent trends. Saudi J Ophthalmol. 2012;26(2):181-189. doi:10.1016/j.sjopt.2012.02.011.
- ↑ Colleaux KM, Hamilton WK. Effect of prophylactic antibiotics and incision type on the incidence of endophthalmitis after cataract surgery. Can J Ophthalmol. 2000;35(7):373-378. doi:10.1016/s0008-4182(00)80124-6.
- ↑ Maxwell DP, Diamond JG. Infectious Endophthalmitis Following Sutureless Cataract Surgery-Reply. Arch Ophthalmol. 1992;110(7):915. doi:10.1001/archopht.1992.01080190019010.
- ↑ McDonnell PJ, Taban M, Sarayba M, et al. Dynamic morphology of clear corneal cataract incisions. Ophthalmology. 2003;110(12):2342-2348. doi:10.1016/S0161-6420(03)00733-4.
- ↑ Herretes S, Stark WJ, Pirouzmanesh A, et al. Inflow of ocular surface fluid into the anterior chamber after phacoemulsification through sutureless corneal cataract wounds. American Journal of Ophthalmology. 2005 Oct;140(4):737-740. DOI: 10.1016/j.ajo.2005.03.069.
- ↑ Sarayba, M. A. et al. (2004) ‘Inflow of ocular surface fluid through clear corneal cataract incisions: a laboratory model’, American Journal of Ophthalmology, 138(2), pp. 206–210. doi: 10.1016/j.ajo.2004.03.012.
- ↑ Tong AY, Gupta PK, Kim T. Wound closure and tissue adhesives in clear corneal incision cataract surgery. Curr Opin Ophthalmol. 2018;29(1):14-18. doi:10.1097/ICU.0000000000000431.
- ↑ Uy HS, Kenyon KR. Surgical outcomes after application of a liquid adhesive ocular bandage to clear corneal incisions during cataract surgery. J Cataract Refract Surg. 2013;39(11):1668-1674. doi:10.1016/j.jcrs.2013.04.041.
- ↑ West ES, Behrens A, McDonnell PJ, Tielsch JM, Schein OD. The incidence of endophthalmitis after cataract surgery among the U.S. Medicare population increased between 1994 and 2001. Ophthalmology. 2005;112(8):1388-1394. doi:10.1016/j.ophtha.2005.02.028.
- ↑ 27.0 27.1 27.2 27.3 Fine IH. Clear corneal incisions. Int Ophthalmol Clin. 1994;34(2):59-72. doi:10.1097/00004397-199403420-00005.
- ↑ 28.0 28.1 Theodoulidou S, Asproudis I, Athanasiadis A, Kokkinos M, Aspiotis M. Comparison of surgically induced astigmatism among different surgeons performing the same incision. Int J Ophthalmol. 2017;10(6):1004-1007. Published 2017 Jun 18. doi:10.18240/ijo.2017.06.26.
- ↑ Piao, J., & Joo, C. K. (2020). Site of clear corneal incision in cataract surgery and its effects on surgically induced astigmatism. Scientific reports, 10(1), 3955. https://doi.org/10.1038/s41598-020-60985-5.
- ↑ 30.0 30.1 Song W, Chen X, Wang W. Effect of steep meridian clear corneal incisions in phacoemulsification. Eur J Ophthalmol. 2015;25(5):422-425. doi:10.5301/ejo.5000575.
- ↑ Özyol E, Özyol P. Analyses of surgically induced astigmatism and axis deviation in microcoaxial phacoemulsification. Int Ophthalmol. 2014;34(3):591-596. doi:10.1007/s10792-013-9858-8.
- ↑ Eun-Ji Kim M, Kim DB. Cataract incision-related corneal erosion: recurrent corneal erosion as a result of clear corneal cataract surgery [published online ahead of print, 2020 Jul 31]. J Cataract Refract Surg. 2020;10.1097/j.jcrs.0000000000000345. doi:10.1097/j.jcrs.0000000000000345.
- ↑ Kim KH, Kim WS. Aniridia after blunt trauma and presumed wound dehiscence in a pseudophakic eye. Arq Bras Oftalmol. 2016;79(1):44-45. doi:10.5935/0004-2749.20160013.
- ↑ Sophocleous S. Doctor, where is my iris?. BMJ Case Rep. 2016;2016:bcr2016214957. Published 2016 May 5. doi:10.1136/bcr-2016-214957.
- ↑ Titiyal JS, Kaur M, Ramesh P, et al. Impact of Clear Corneal Incision Morphology on Incision-Site Descemet Membrane Detachment in Conventional and Femtosecond Laser-Assisted Phacoemulsification. Curr Eye Res. 2018;43(3):293-299. doi:10.1080/02713683.2017.1396616.
- ↑ Singhal D, Sahay P, Goel S, Asif MI, Maharana PK, Sharma N. Descemet membrane detachment. Surv Ophthalmol. 2020;65(3):279-293. doi:10.1016/j.survophthal.2019.12.006.
- ↑ Haldipurkar SS, Shikari HT, Gokhale V. Wound construction in manual small incision cataract surgery. Indian J Ophthalmol. 2009;57(1):9-13. doi:10.4103/0301-4738.44491.
- ↑ Hennig A, Kumar J, Yorston D, Foster A. Sutureless cataract surgery with nucleus extraction: outcome of a prospective study in Nepal. Br J Ophthalmol. 2003;87(3):266-270. doi:10.1136/bjo.87.3.266.
- ↑ Khokhar S, Gupta S, Tewari R, et al. Scleral tunnel phacoemulsification: Approach for eyes with severe microcornea. Indian J Ophthalmol. 2016;64(4):320-322. doi:10.4103/0301-4738.182949.
- ↑ 40.0 40.1 Rho CR, Joo CK. Effects of steep meridian incision on corneal astigmatism in phacoemulsification cataract surgery. J Cataract Refract Surg. 2012;38(4):666-671. doi:10.1016/j.jcrs.2011.11.031.
- ↑ 41.0 41.1 Rao SN, Konowal A, Murchison AE, Epstein RJ. Enlargement of the temporal clear corneal cataract incision to treat pre-existing astigmatism. J Refract Surg. 2002;18(4):463-467.
- ↑ 42.0 42.1 42.2 42.3 42.4 42.5 42.6 Mohan, Shalini & Khokhar, Sudarshan & Aggarwal, Anand & Panda, Anita. (2007). Wound Construction. DOS Times. 13. 13-21.
- ↑ Ernest P, Tipperman R, Eagle R, et al. Is there a difference in incision healing based on location?. J Cataract Refract Surg. 1998;24(4):482-486. doi:10.1016/s0886-3350(98)80288-5
- ↑ 44.0 44.1 Nikose AS, Saha D, Laddha PM, Patil M. Surgically induced astigmatism after phacoemulsification by temporal clear corneal and superior clear corneal approach: a comparison. Clin Ophthalmol. 2018;12:65-70. Published 2018 Jan 3. doi:10.2147/OPTH.S149709.
- ↑ Hayashi K, Sato T, Yoshida M, Yoshimura K. Corneal shape changes of the total and posterior cornea after temporal versus nasal clear corneal incision cataract surgery. Br J Ophthalmol. 2019;103(2):181-185. doi:10.1136/bjophthalmol-2017-311710.
- ↑ 46.0 46.1 Tejedor J, Murube J. Choosing the location of corneal incision based on preexisting astigmatism in phacoemulsification. Am J Ophthalmol. 2005;139(5):767-776. doi:10.1016/j.ajo.2004.12.057.
- ↑ Razmjoo H, Koosha N, Vaezi MH, Rahimi B, Peyman A. Corneal astigmatism change and wavefront aberration evaluation after cataract surgery: "Single" versus "paired opposite" clear corneal incisions. Adv Biomed Res. 2014;3:163. Published 2014 Aug 19. doi:10.4103/2277-9175.139126.
- ↑ Wang L, Zhao L, Yang X, Zhang Y, Liao D, Wang J. Comparison of Outcomes after Phacoemulsification with Two Different Corneal Incision Distances Anterior to the Limbus. J Ophthalmol. 2019;2019:1760742. Published 2019 Aug 19. doi:10.1155/2019/1760742.
- ↑ 49.0 49.1 Ernest PH, Lavery KT, Kiessling LA. Relative strength of scleral corneal and clear corneal incisions constructed in cadaver eyes. J Cataract Refract Surg. 1994;20(6):626-629. doi:10.1016/s0886-3350(13)80651-7.
- ↑ Yang J, Wang X, Zhang H, Pang Y, Wei RH. Clinical evaluation of surgery-induced astigmatism in cataract surgery using 2.2 mm or 1.8 mm clear corneal micro-incisions. Int J Ophthalmol. 2017;10(1):68-71. Published 2017 Jan 18. doi:10.18240/ijo.2017.01.11.
- ↑ Febbraro JL, Wang L, Borasio E, et al. Astigmatic equivalence of 2.2-mm and 1.8-mm superior clear corneal cataract incision. Graefes Arch Clin Exp Ophthalmol. 2015;253(2):261-265. doi:10.1007/s00417-014-2854-5.
- ↑ Yu YB, Zhu YN, Wang W, Zhang YD, Yu YH, Yao K. A comparable study of clinical and optical outcomes after 1.8, 2.0 mm microcoaxial and 3.0 mm coaxial cataract surgery. Int J Ophthalmol. 2016;9(3):399-405. Published 2016 Mar 18. doi:10.18240/ijo.2016.03.13.
- ↑ 53.0 53.1 53.2 Hayashi K, Yoshida M, Hirata A, Yoshimura K. Changes in shape and astigmatism of total, anterior, and posterior cornea after long versus short clear corneal incision cataract surgery. J Cataract Refract Surg. 2018;44(1):39-49. doi:10.1016/j.jcrs.2017.10.037.
- ↑ Wilczynski M, Supady E, Wierzchowski T, Zdzieszynski M, Omulecki W. The effect of corneal tunnel length in patients after standard phacoemulsification through a 2.75 mm incision on surgically induced astigmatism, corneal thickness and endothelial cell density. Klin Oczna. 2016;117(4):236-242.
- ↑ Sonmez S, Karaca C. The effect of tunnel length and position on postoperative corneal astigmatism: An optical coherence tomographic study. Eur J Ophthalmol. 2020;30(1):104-111. doi:10.1177/1120672118805875.
- ↑ Masket S, Belani S. Proper wound construction to prevent short-term ocular hypotony after clear corneal incision cataract surgery. J Cataract Refract Surg. 2007;33(3):383-386. doi:10.1016/j.jcrs.2006.11.006.
- ↑ May W, Castro-Combs J, Camacho W, Wittmann P, Behrens A. Analysis of clear corneal incision integrity in an ex vivo model. J Cataract Refract Surg. 2008;34(6):1013-1018. doi:10.1016/j.jcrs.2008.01.038.
- ↑ Mastropasqua L, Toto L, Mastropasqua A, et al. Femtosecond laser versus manual clear corneal incision in cataract surgery. J Refract Surg. 2014;30(1):27-33. doi:10.3928/1081597x-20131217-03.
- ↑ Moshirfar M, Churgin DS, Hsu M. Femtosecond laser-assisted cataract surgery: a current review. Middle East Afr J Ophthalmol. 2011;18(4):285-291. doi:10.4103/0974-9233.90129.