Cerulean Cataract
All content on Eyewiki is protected by copyright law and the Terms of Service. This content may not be reproduced, copied, or put into any artificial intelligence program, including large language and generative AI models, without permission from the Academy.
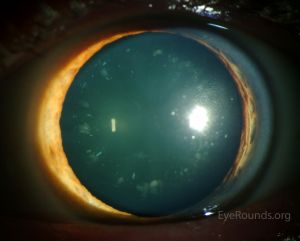
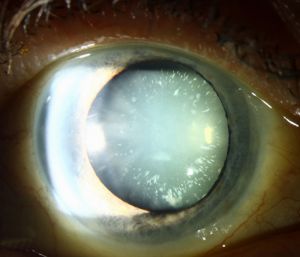
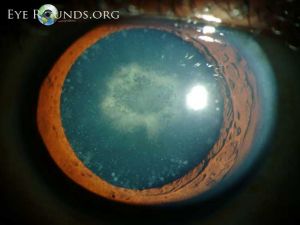
Cerulean cataracts are inherited, developmental cataracts characterized by blue and white opacifications scattered in the nucleus and cortex of the lens. They usually do not affect vision until adulthood and can be diagnosed by slit lamp examinations. In patients for whom cerulean cataracts cause visual impairment, treatment is with surgical cataract removal and artificial lens placement.
Cerulean Cataract
Disease
Etiology/Pathophysiology
Cerulean cataracts are inherited as an autosomal dominant trait. At least four loci for the cerulean cataract phenotype have been mapped – congenital cataract-1 (CCA1;17q24), CCA2 (22q11.2-q12.2), CCA3 (2q33-q35), and CC4 (16q22-q23).[1] Multiple causative mutations have been identified, including mutations in the beta-B2-crystallin gene (CRYBB2)[2], gamma-D-crystallin gene (CRYGD)[3], V-MAF avian musculoaponeurotic fibrosarcoma oncogene homolog gene (MAF)[4], and the major intrinsic protein of lens fiber gene (MIP).[1]
Diagnosis
Presentation
Cerulean cataracts can be present at birth or develop during infancy/early childhood.[5] [6] Progression of cerulean cataracts can be slow and may not become significant until adulthood, when patients begin to notice a gradual decrease in vision in both eyes.[7] However, cerulean cataracts can cause visually impairment early on in some patients resulting in nystagmus and amblyopia if left undiagnosed and untreated.[6][8]
Physical examination
Cerulean cataracts usually present as bilateral, blue-white opacities that begin to form in the fetal nucleus after birth.[5] Patients can be diagnosed via slit lamp examination, which would reveal tiny blue and/or white opacities in the superficial layer of the lens nucleus.[9]
Differential diagnosis
The differential diagnosis for cerulean cataracts includes other congenital, developmental and traumatic cataracts. These can be distinguished by physical examination, family history, and laboratory tests.
Management
Serial evaluations and surgery
There are no known treatments to prevent the formation and progression of cerulean cataracts. Serial eye evaluations and eventual cataract surgery if they are causing visual impairment are the standard of care.[8]
References
- ↑ Jump up to: 1.0 1.1 Xiao X, Li W, Wang P, et al. Cerulean cataract mapped to 12q13 and associated with a novel initiation codon mutation in MIP. Mol Vis. 2011;17:2049-2055.
- ↑ Litt M, Carrero-Valenzuela R, LaMorticella DM, et al. Autosomal Dominant Cerulean Cataract Is Associated with a Chain Termination Mutation in the Human β-Crystallin Gene CRYBB2. Human Molecular Genetics. 1997;6(5):665-668. doi:10.1093/hmg/6.5.665
- ↑ Nandrot E, Slingsby C, Basak A, et al. Gamma-D crystallin gene (CRYGD) mutation causes autosomal dominant congenital cerulean cataracts. Journal of Medical Genetics. 2003;40(4):262-267. doi:10.1136/jmg.40.4.262
- ↑ Vanita V, Singh D, Robinson PN, Sperling K, Singh JR. A novel mutation in the DNA-binding domain of MAF at 16q23.1 associated with autosomal dominant “cerulean cataract” in an Indian family. American Journal of Medical Genetics Part A. 2006;140A(6):558-566. doi:10.1002/ajmg.a.31126
- ↑ Jump up to: 5.0 5.1 Ram J, Singh A. Cerulean cataract. QJM: An International Journal of Medicine. 2019;112(9):699-699. doi:10.1093/qjmed/hcz038
- ↑ Jump up to: 6.0 6.1 Cerulean cataract | Genetic and Rare Diseases Information Center (GARD) – an NCATS Program. Accessed September 19, 2021. https://rarediseases.info.nih.gov/diseases/9508/cerulean-cataract/cases/30241#ref_3241
- ↑ Kumawat D, Jayaraman N, Sahay P, Chandra P. Multicoloured lenticular opacities in a case of cerulean cataract. BMJ Case Reports CP. 2019;12(4):e230167. doi:10.1136/bcr-2019-230167
- ↑ Jump up to: 8.0 8.1 Pediatric Cataracts: Overview. American Academy of Ophthalmology. Published November 11, 2015. Accessed September 19, 2021. https://www.aao.org/disease-review/pediatric-cataracts-overview
- ↑ Armitage MM, Kivlin JD, Ferrell RE. A progressive early onset cataract gene maps to human chromosome 17q24. Nat Genet. 1995;9(1):37-40. doi:10.1038/ng0195-37