CMV Endotheliitis
All content on Eyewiki is protected by copyright law and the Terms of Service. This content may not be reproduced, copied, or put into any artificial intelligence program, including large language and generative AI models, without permission from the Academy.
Disease Entity
Cytomegalovirus (CMV) endotheliitis is a disorder involving inflammation of the corneal endothelium triggered by CMV.
Disease
CMV endotheliitis is defined as inflammation of the corneal endothelial cells caused by a CMV infection. The disease is characterized by the presence of small to medium-sized keratic precipitates (KPs) on slit-lamp examination. Corneal edema and increased intraocular pressure (IOP) are often present, the latter likely due to concurrent inflammation of the trabecular meshwork[1]. Severe and untreated infections can damage the corneal endothelium, causing persistent corneal edema[2].
Corneal endotheliitis can be classified clinically into four types: linear, sectoral, disciform, and diffuse. This classification is based on the distribution of the KPs and the pattern of overlying stromal and epithelial edema. In linear endotheliitis, the KPs are present in a linear distribution, and the corneal edema is localized. In sectoral endotheliitis, the KPs and corneal edema are also localized; however, the KPs are spread across a broader region than in linear disease. Disciform endotheliitis is characterized by a round or disc-shaped area of KPs and overlying edema. Finally, the corneal edema is spread over the entire cornea in diffuse endotheliitis, with dispersed KPs across the endothelial surface[1][3][4].
Endotheliitis caused by CMV often presents with linear or coin-shaped KPs, which may be accompanied by mild anterior chamber inflammation[4]. It is typically unilateral and may occur in immunocompetent individuals[5]. Retrospective case analyses of CMV-associated ocular infections indicate that the anterior segment is predominantly involved in immunocompetent individuals, with additional posterior segment involvement in the immunocompromised[6].
Etiology
Corneal endotheliitis can be broadly defined as a spectrum of disorders in which the corneal endothelium is the primary site of inflammation. Initially presumed to be an autoimmune process, growing evidence suggests that the disease is often caused by viral infections, including herpes simplex virus (HSV), varicella zoster virus (VZV), or cytomegalovirus (CMV)[1][7]. Significant correlations have been found between CMV viral load in the aqueous humor and anterior segment inflammation severity, as measured by clinical parameters, including the grade of anterior chamber cells, IOP elevation, corneal endothelial cell loss, and the incidence of recurrent inflammation episodes[8][9]. Primary cultured corneal endothelial cells have been found to be capable of supporting CMV infection and replication[10].
Human cytomegalovirus is a ubiquitous herpes family virus, with seroprevalence rates varying widely from 45-100% across the world. The highest rates generally occur in South America, Asia, and Africa, with the lowest rates recorded in Europe and the United States (U.S.) However, substantial variation exists. An inverse correlation between prevalence rates and socioeconomic status has also been established[11]. Like other herpes viruses, CMV infection is typically characterized by an asymptomatic primary infection. The virus then establishes a lifelong latent infection in the trabecular meshwork and ciliary body that can be periodically reactivated, causing recurrent disease[12]. Most ocular manifestations in immunocompetent individuals are due to the reactivation of latent CMV.
Risk Factors
Most reports of CMV-related anterior segment disease are from Asian countries, primarily involving Chinese and Japanese patient populations[7][13]. The Japan corneal endotheliitis study is the largest case-series of CMV endotheliitis to date, with 109 reported cases. The disease was most prevalent in middle-aged and older patients (mean age of 66.9 years), with male predominance (80.2% of cases). Prior corneal transplantation was also found to be linked with CMV endotheliitis, implicating CMV endotheliitis as either a possible cause of graft failure or as the primary cause of endothelial dysfunction that prompted corneal transplantation[4]. It has also been suggested that topical corticosteroid therapy and local immunosuppression may be a risk factor for the reactivation of CMV endotheliitis[7].
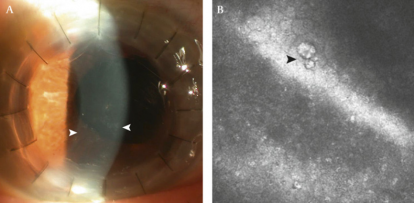
General Pathology
A distinctive “owl’s eye” morphology is pathognomonic of CMV infection: large endothelial cells with nuclei possessing a central area of high reflectivity, surrounded by a halo of low reflectivity, can be found on the corneal endothelial surface through in vivo confocal microscopy[14]. (Figure 1) These confocal findings may be reversible after the resolution of endotheliitis and may help monitor the effects of treatment[15].
Mild stromal edema, with a loss of corneal endothelial cells, has been reported after prolonged CMV endotheliitis in histologic analysis of both native cornea tissue[2] and failed penetrating keratoplasty grafts[16]. Notably, histopathology demonstrated a lack of prominent inflammation as well as an absence of reactive leukocytes in the corneal stroma. (Figure 2)
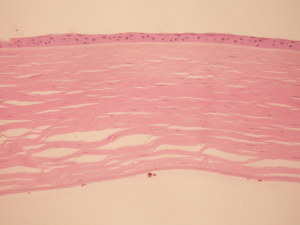
Pathophysiology
It is hypothesized that the pathophysiology of CMV associated endotheliitis is similar to HSV endotheliitis. Latent HSV infection can intermittently reactivate anywhere along the trigeminal ganglion. A small amount of virus may be shed into the anterior chamber of the eye via the nerve branches that supply the trabecular meshwork, iris and ciliary body[17][18][19]. It has been proposed that anterior chamber associated immune deviation (ACAID) may play a role in HSV-1 corneal endotheliitis[20]. HSV-1 particles that are released into the anterior chamber are captured by indigenous antigen-presenting cells, which in turn, generate an immunogenic signal that induces virus specific ACAID. Various cytokines, including IL-6 and IL-8, have been found in the aqueous humor of patients with the disease.[21]
In an experiment by Zheng et al, anterior chamber priming with viral antigen in a rabbit model produced ACAID by this proposed mechanism[22]. CMV, which has been clinically noted to establish a reservoir in the trabecular meshwork and ciliary body, may induce a similar ACAID response[20]. This may explain why some patients experience a recurrence of CMV endotheliitis after stopping topical anti-inflammatory treatment, but respond well to a treatment regimen that includes antiviral medication.
The
Primary Prevention
There is no known method for primary prevention of CMV endotheliitis. In cases of corneal transplantation performed due to CMV endotheliitis, antivirals can be used to prevent a recurrence. See Prognosis section below.
Diagnosis
In general, corneal endotheliitis is primarily a clinical diagnosis, with KPs and overlying corneal edema often accompanied by a mild anterior chamber reaction[1]. The detection of CMV DNA in the aqueous fluid (via quantitative polymerase chain reaction [PCR]), in the presence of the above clinical findings, is confirmatory for CMV-associated endotheliitis. The PCR test may only be positive during the early stages of infection due to a decrease in viral load to below detectable levels later in the disease course. PCR analysis of the sample for HSV and VZV should also be performed[23]. Testing for CMV serologies and antibodies in the aqueous fluid should also be considered.
In some instances, when the diagnosis is difficult to confirm, the Goldmann-Witmer coefficient (GWC) can be used to assess the etiology of the present antibodies. Antibody level in the aqueous and/or vitreous fluid is compared with the antibody level in the serum, multiplied by total serum globulins. A value greater than 3 indicates that the detected antibodies were formed in the eye and did not passively diffuse from the serum[1]. However, the test is of limited use in immunocompromised patients due to their overall low antibody production[23]. Using both PCR and GWC analysis can improve diagnostic accuracy[24].
In vivo confocal microscopy can be used to detect characteristic morphological changes in the cornea and detect findings specific to CMV, such as the presence of an “owl’s eye”[1][14]. Recently, anterior segment spectral-domain optical coherence tomography (SD-OCT) in confirmed CMV endotheliitis cases showed high reflectivity (76.95%) of posterior corneal lesions and saw-tooth appearing protruding structures, presumed KPs, which resolved gradually with treatment[25].
History
A patient presenting with CMV endotheliitis may describe photophobia and/or decreased vision, typically in one eye. These symptoms are non-specific and may be associated with various etiologies of ocular inflammation. Additionally, patients who develop bullae from corneal edema may suffer from eye pain or foreign body sensation.
Physical Examination
As described above, slit-lamp examination demonstrates KPs with overlying corneal edema, present in several different patterns. The degree of anterior chamber cells may be mild as compared to the corneal findings. Conjunctival injection may be present. At initial presentation and again as indicated, dilated fundus examination should be performed, and posterior segment involvement should be ruled out.
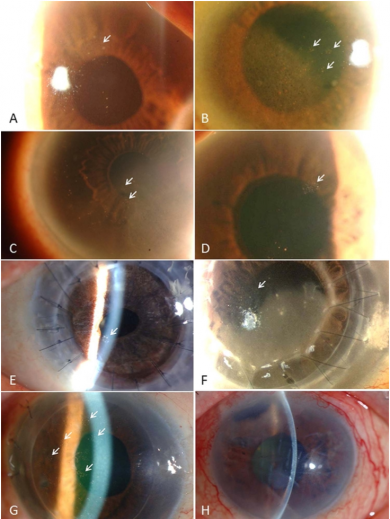
Signs
Clinical signs of CMV endotheliitis include:
- Small to medium-sized, non-granulomatous, and often pigmented KPs. They appear in a typical coin-shaped or linear pattern. (Figure 3)
- Coin-shaped KPs tend to be more common, occurring in 70.6% of eyes in the Japan Endotheliitis study[4]: this has a high positive predictive value for CMV endotheliitis.
- Most often KPs are located in the Arlt’s triangle, but may also appear elsewhere.
- Corneal edema
- Associated ocular hypertension, likely secondary to concurrent trabeculitis.
- Mild anterior chamber inflammation.
- Posterior synechiae formation is rare.
Symptoms
See History section above for a discussion of presenting symptoms in patients with CMV endotheliitis.
Diagnostic Procedures
Based on the level of clinical suspicion for CMV endotheliitis and a complete eye exam, including slit-lamp biomicroscopy and IOP measurement, the following diagnostic procedures may be considered. See Diagnosis section above for further discussion:
- PCR (polymerase chain reaction) analysis of the aqueous fluid
- Aqueous (+/- serum) CMV antibody analysis via ELISA (Enzyme-Linked Immunosorbent Assay)
- In vivo confocal microscopy
- Anterior segment OCT
Laboratory Test
See Diagnosis section above for a discussion of laboratory tests that may be used for CMV endotheliitis.
Differential Diagnosis
Corneal endotheliitis can also be caused by HSV, VZV[26], mumps[27], and autoimmune disease. It is important to exclude HSV and VZV via PCR analysis of the aqueous fluid, as the treatment differs from CMV.
CMV endotheliitis is one anterior segment manifestation of clinical CMV infection. The Japan corneal endotheliitis study described ocular hypertension and anterior uveitis as commonly associated features of CMV endotheliitis[4].
The spectrum of CMV ocular manifestations include: hypertensive anterior uveitis, severe chronic anterior uveitis with recurrent episodes of iritis, elevated IOP resembling Posner-Schlossman syndrome (PSS), as well as anterior chamber cells, KPs, and iris atrophy resembling Fuchs’ heterochromic iridocyclitis (FHI)[11][28]. Previously considered idiopathic, cases of PSS and FHI cases have now been linked with CMV, raising the question of whether these conditions may have a viral etiology[6][29].
The diagnosis of CMV can be challenging, as the initial findings of ocular inflammation are often treated with topical corticosteroids. While topical corticosteroids may lead to temporary improvement of inflammation, when unaccompanied by antiviral treatment, topical corticosteroids can aggravate viral activity. In turn, this has the potential to intensify corneal damage and endothelial cell loss. In many cases, CMV endotheliitis is only diagnosed after the patient fails to respond to topical steroid therapy.
Differentiating CMV endotheliitis from immunologic corneal graft rejection in corneal transplant patients can also be difficult as clinical signs closely resemble one another. Subtle differences, however, have been noted between CMV + and CMV - eyes presenting with endothelial rejection after penetrating keratoplasty. CMV positive cases included the extensive presence of pigmented KPs, usually arranged in coarse clumps in the troughs of Descemet membrane folds, as well as the absence of vascularization of the donor[30].
Episodes of allograft rejection unresponsive to steroids or unexplained endothelial cell loss in a relatively quiet eye post corneal transplantation should prompt an aqueous tap to rule out CMV by PCR[31]. Non-viral anterior uveitis is typically more commonly associated with low IOP, so hypertensive anterior uveitis should raise concern of possible viral etiology. Distinguishing between graft rejection and viral endotheliitis is vital. The use of intensive topical and/or systemic steroid immunosuppression (without antivirals) may exacerbate CMV activity, thereby worsening the course of CMV endotheliitis and leading to endothelial cell loss and graft failure. A high index of suspicion for CMV endotheliitis is advised, especially in patients of Asian descent[32].
Management
In the Japan Endotheliitis study, 95% of the participants received topical ganciclovir, systemic ganciclovir or valganciclovir, or a combination of topical and systemic antiviral therapy[4]. Combined systemic and topical therapy appeared to be more effective in reducing KPs and corneal edema, but this was not statistically significant compared to other treatments. After treatment, a considerable portion of patients (36 %) experienced a recurrence and needed to be placed back on therapy. The study also reported that 5.8% of patients did not respond to any treatment, signifying that severe corneal endothelial damage in CMV endotheliitis may be irreversible even with antiviral treatment.
Other studies have used real-time PCR testing before and after treatment to show decreased CMV DNA following treatment with systemic and topical ganciclovir combined with a topical steroid[8][33]. In cases of severe infection, marked by persistent corneal edema and significant endothelial cell loss, corneal transplantation may be necessary. Endothelial keratoplasty (DSAEK or DMEK) may be considered.
Medical Therapy
Treatment algorithms for CMV infections of the anterior segment have been reported in the literature[3][7]. For CMV endotheliitis, current treatment regimens are aimed at targeting both the infectious and inflammatory components. Most commonly, this is approached by combining topical corticosteroids with: (1) topical antiviral treatment, (2) systemic antiviral treatment, or (3) combined topical + systemic antiviral treatment. Anti-glaucoma medication may also be necessary to control elevated IOP.
Potential antiviral regimens are listed below, although the dosing and frequency varies in the literature:
- Topical ganciclovir
- First line: 0.15% ganciclovir gel 5 times daily (therapeutic dosing) with subsequent taper to prophylactic dosing at TID.
- Second line (if failed ganciclovir gel): compounded ganciclovir (0.5% - 2%)
- Systemic antiviral treatment (suggested dosing below, but recommend considering co-management with Infectious Disease specialist):
- First line: Oral valganciclovir 900 mg BID (therapeutic dose) with subsequent taper to prophylactic dosing at 900 mg QD.
- Second line (if failed PO valganciclovir treatment): Intravenous 5 mg/kg ganciclovir BID
- Third line: Intravenous foscarnet 60 mg/kg TID or cidofovir are alternatives but can cause significant nephrotoxicity
CMV endotheliitis treatment with topical ganciclovir gel (0.15%), without concomitant systemic antiviral therapy, was first reported by Pavan-Langston et al. In a small prospective interventional case series of immunocompetent patients with CMV endotheliitis, Koizumi et al. found that topical ganciclovir gel alone led to clinical improvements and lowered aqueous CMV viral load[34]. The authors found that there were some cases, however, where ganciclovir gel was insufficient. Notably, this occurred in patients who had either high viral CMV load or low aqueous ganciclovir concentrations. In subsequent studies, Fan et al. reported good outcomes with topical ganciclovir 0.5%, and Hwang et al with topical ganciclovir 2%[35][36]. The use of maintenance topical ganciclovir to prevent repeat failure of endothelial transplant regrafts in CMV endotheliitis patients has also been reported[37][38]. Lastly, there have also been reports of CMV endotheliitis successfully treated with intravitreal ganciclovir injections[39][40].
While further studies are needed to determine the optimal regimen and duration of therapy, most authors continue prophylactic antiviral therapy for at least 3 months after clinical resolution. Many authors also report rechecking the aqueous for the absence of CMV via PCR before discontinuing treatment.
Medical Follow Up
Patients placed on systemic valganciclovir or ganciclovir need to have their kidney function closely monitored (serum creatinine), as well as regular complete blood count (CBC) with differential, as usage can results in kidney damage (including acute renal failure) and blood dyscrasias . Patients with a history of renal impairment require dosing according to their renal function as well as more frequent monitoring. If myelosuppression, nephrotoxicity, or pancytopenia occur, systemic treatment should be discontinued.
Surgery
If treatment for CMV endotheliitis is not initiated early enough or if the patient fails to respond to medical therapy, the severity of corneal endothelial damage may warrant surgical intervention. Corneal transplantation is indicated in instances of irreversible corneal decompensation. In recent years, successful cases of endothelial keratoplasty (DSAEK and DMEK) in this setting have been reported[32][37]. There should be 90 days of inactive inflammation in order to safely proceed with surgical intervention.
It can be challenging to differentiate post-procedure CMV endotheliitis from graft rejection. Both conditions may present with anterior chamber inflammation, KPs, and corneal edema. Graft rejection treatment involving high dose corticosteroids can also lead to an elevated IOP. If CMV endotheliitis is not ruled out, steroid treatment can result in reactivation of latent CMV (or exacerbation of active CMV infection) with progression to subsequent graft failure[41].
Surgical Complications
Despite preoperative and postoperative anti-viral coverage, recurrence of CMV endotheliitis represents a significant risk. Prolonged postoperative use of systemic and/or topical immunosuppressants can cause reactivation of latent CMV harbored in anterior segment structures[31][41]. Long-term treatment with oral valganciclovir or topical ganciclovir is recommended to reduce graft failure rates[42].
Overall Complications
As previously noted, CMV endotheliitis can lead to irreversible corneal endothelial damage. This can lead to the need for a cornea transplant[31].
A diagnosis of CMV endotheliitis should also be entertained before consideration of corneal transplant surgery in order to reduce the risk of post-procedure graft rejection. A retrospective histological study of failed penetrating keratoplasty procedures revealed that CMV was not just confined to the endothelium, but extended into the corneal stroma, where it acted as a reservoir for graft infection after a keratoplasty procedure[43]. The usage of post-op topical corticosteroids may further exacerbate this process by allowing latent CMV to reactivate and proliferate.
Prognosis
CMV endotheliitis can recur at a rate of 5 - 60% after the discontinuation of therapy[4][7][44]. Maintenance therapy with oral valganciclovir or topical ganciclovir in combination with corticosteroids may be required. Although systemic valganciclovir is effective in preventing disease recurrence[45], its long-term usage may be limited by serious side effects such as pancytopenia, myelosuppression, and nephrotoxicity. Topical ganciclovir has been shown to have favorable clinical outcomes for preventing recurrence, especially in milder forms of CMV endotheliitis[35][46][47].
Prevention
See Prognosis section above.
References
- ↑ 1.0 1.1 1.2 1.3 1.4 1.5 Alfawaz A. Cytomegalovirus-related corneal endotheliitis: A review article. Saudi J Ophthalmol. 2013;27(1):47-49.
- ↑ 2.0 2.1 Hwang Y-S, Hsiao C-H, Tan H-Y, Chen K-J, Chen T-L, Lai C-C. Corneal endotheliitis. Ophthalmology. 2009;116(1):164-164.e1.
- ↑ 3.0 3.1 Moshirfar M, Murri MS, Shah TJ, et al. A Review of Corneal Endotheliitis and Endotheliopathy: Differential Diagnosis, Evaluation, and Treatment. Ophthalmol Ther. 2019;8(2):195-213.
- ↑ 4.0 4.1 4.2 4.3 4.4 4.5 4.6 Koizumi N, Inatomi T, Suzuki T for the Japan Corneal Endotheliitis Study Group, et al. Clinical features and management of cytomegalovirus corneal endotheliitis: analysis of 106 cases from the Japan corneal endotheliitis studyBritish Journal of Ophthalmology 2015;99:54-58.
- ↑ Inoue Y. Review of clinical and basic approaches to corneal endotheliitis. Cornea. 2014;33 Suppl 11:S3-S8. doi:10.1097/ICO.0000000000000228
- ↑ 6.0 6.1 Chee SP, Jap A. Presumed fuchs heterochromic iridocyclitis and Posner-Schlossman syndrome: comparison of cytomegalovirus-positive and negative eyes. Am J Ophthalmol. 2008 Dec;146(6):883-9.e1
- ↑ 7.0 7.1 7.2 7.3 7.4 Kumar A. and Mehta J.S. Diagnosis and Management of CMV Endotheliitis. Current Ophthalmology Reports. 2019;7:98-109.
- ↑ 8.0 8.1 Miyanaga M, Sugita S, Shimizu N, et al . A significant association of viral loads with corneal endothelial cell damage in cytomegalovirus anterior uveitis. Br J Ophthalmol 2010;94:336–40.
- ↑ Kandori M, Miyazaki D, Yakura K, et al. Relationship between the number of cytomegalovirus in anterior chamber and severity of anterior segment inflammation. Japanese Journal of Ophthalmology. 2013;57(6):497-502. doi:10.1007/s10384-013-0268-2
- ↑ Mayumi Hosogai, Nobuyuki Shima, Yoko Nakatani, et al. Analysis of human cytomegalovirus replication in primary cultured human corneal endothelial cells. British Journal of Ophthalmology. 2015;99(11):1583-1590. doi:10.1136/bjophthalmol-2014-306486
- ↑ 11.0 11.1 Cannon MJ, Schmid DS, Hyde TB. Review of cytomegalovirus seroprevalence and demographic characteristics associated with infection. Rev Med Virol. 2010;20(4):202-213. doi:10.1002/rmv.655
- ↑ Schottstedt V, Blümel J, Burger R, et al. Human Cytomegalovirus (HCMV) - Revised. Transfus Med Hemother. 2010;37(6):365-375. doi:10.1159/000322141
- ↑ Chee S-P, Jap A. Treatment outcome and risk factors for visual loss in Cytomegalovirus endotheliitis. Graefe’s Archive of Clinical & Experimental Ophthalmology. 2012;250(3):383-389. doi:10.1007/s00417-011-1813-7
- ↑ 14.0 14.1 Shiraishi A, Hara Y, Takahashi M, et al. Demonstration of “Owl’s Eye” Morphology by Confocal Microscopy in a Patient With Presumed Cytomegalovirus Corneal Endotheliitis. American Journal of Ophthalmology. 2007;143(4):715-717. doi:10.1016/j.ajo.2006.11.026
- ↑ Kobayashi A, Yokogawa H, Higashide T, Nitta K, Sugiyama K. Clinical significance of owl eye morphologic features by in vivo laser confocal microscopy in patients with cytomegalovirus corneal endotheliitis. Am J Ophthalmol. 2012;153(3):445-453. doi:10.1016/j.ajo.2011.07.026
- ↑ Sonoyama H, Araki-Sasaki K, Osakabe Y, et al. Detection of cytomegalovirus DNA from cytomegalovirus corneal endotheliitis after penetrating keratoplasty. Cornea. 2010;29(6):683-685. doi:10.1097/ICO.0b013e3181c325e2
- ↑ Ohashi Y, Yamamoto S, Nishida K, Okamoto S, Kinoshita S, Hayashi K. Demonstration of herpes simplex virus DNA in idiopathic corneal endotheliopathy. Am J Ophthalmol. 1991;112:419–423.
- ↑ Liesegang TJ. Biology and molecular aspects of herpes simplex and varicella-zoster virus infections. Ophthalmology. 1992;99:781–799.
- ↑ Pavan–Langston D. Herpes simplex virus ocular infections: current concept of acute, latent and reactivated disease. Trans Am Ophthalmol Soc. 1990;88:727–796.
- ↑ 20.0 20.1 Koizumi N, Suzuki T, Uno T, et al. Cytomegalovirus as an etiologic factor in corneal endotheliitis. Ophthalmology. 2008;115(2):292-297.e3.
- ↑ Zhang XJ, Zhang JX, Qu Y, Peng RM, Zhang P, Hong J. Cytokine analysis of aqueous humor in patients with cytomegalovirus corneal endotheliitis. Graefes Arch Clin Exp Ophthalmol. 2024 Mar 6. doi: 10.1007/s00417-024-06417-w. Epub ahead of print. PMID: 38446197.
- ↑ Xiaodong Zheng, Masahiko Yamaguchi, Tomoko Goto, Shigeki Okamoto, Yuichi Ohashi; Experimental Corneal Endotheliitis in Rabbit. Invest. Ophthalmol. Vis. Sci. 2000;41(2):377-385.
- ↑ 23.0 23.1 Foster, C Stephen. “The Uveitis Syndromes - Infectious .” Diagnosis and Treatment of Uveitis , edited by Albert T Vitale , 2nd ed., vol. 1, JP Medical Ltd, 2013, pp. 446–453.
- ↑ Relvas LJM, Antoun J, de Groot-Mijnes JDF, et al. Diagnosis of Cytomegalovirus Anterior Uveitis in Two European Referral Centers. Ocular Immunology & Inflammation. 2018;26(1):116-121. doi:10.1080/09273948.2017.1411952
- ↑ Kobayashi R, Hashida N, Soma T, Koh S, Miki A, Usui S, Maeda N, Nishida K. Clinical Findings of Anterior Segment Spectral Domain Optical Coherence Tomography Images in Cytomegalovirus Corneal Endotheliitis. Cornea. 2017 Apr;36(4):411-414. doi: 10.1097/ICO.0000000000001103. PMID: 27941387.
- ↑ Maudgal P.C., Missotten L., De Clercq E., Descamps J. Varicella-zoster virus in the human corneal endothelium: a case report. Bull. Soc. Belge Ophtalmol. 1980;190:71–86
- ↑ Singh K., Sodhi P.K. Mumps-induced corneal endotheliitis. Cornea. 2004;23:400–402
- ↑ Chee SP, Bacsal K, Jap A, Se-Thoe SY, Cheng CL, Tan BH. Corneal endotheliitis associated with evidence of cytomegalovirus infection. Ophthalmology. 2007;114(4):798-803. doi:10.1016/j.ophtha.2006.07.057
- ↑ Chee S-P, Bacsal K, Jap A, Se-Thoe S-Y, Cheng CL, Tan BH. Clinical Features of Cytomegalovirus Anterior Uveitis in Immunocompetent Patients. American Journal of Ophthalmology. 2008;145(5):834-840. doi:10.1016/j.ajo.2007.12.015
- ↑ Chee, Soon-Phaik, Jap, Aliza, Wen Ling, Elaine, Ti, Seng-Ei. Cytomegalovirus-Positive Corneal Stromal Edema With Keratic Precipitates After Penetrating Keratoplasty: A Case-Control Study. Cornea. 2013;32(8):1094-1098. doi:10.1097/ICO.0b013e318283c887.
- ↑ 31.0 31.1 31.2 Anshu A, Chee SP, Mehta JS, Tan DTH. Cytomegalovirus endotheliitis in Descemet’s stripping endothelial keratoplasty. Ophthalmology. 2009;116(4):624–30.
- ↑ 32.0 32.1 Tan, Tien-En, Tan, Donald. Cytomegalovirus Corneal Endotheliitis After Descemet Membrane Endothelial Keratoplasty. Cornea. 2019;38(4):413-418. doi:10.1097/ICO.0000000000001847.
- ↑ Kandori M, Inoue T, Takamatsu F, et al. Prevalence and Features of Keratitis with Quantitative Polymerase Chain Reaction Positive for Cytomegalovirus. Ophthalmology. 2010;117(2):216-222.
- ↑ Koizumi N, Miyazaki D, Inoue T, et al. The effect of topical application of 0.15% ganciclovir gel on cytomegalovirus corneal endotheliitis. The British journal of ophthalmology. 2017;101(2):114-119. doi:10.1136/bjophthalmol-2015-308238
- ↑ 35.0 35.1 Fan N-W, Chung Y-C, Liu Y-C, Liu CJ-L, Kuo Y-S, Lin P-Y. Long-Term Topical Ganciclovir and Corticosteroids Preserve Corneal Endothelial Function in Cytomegalovirus Corneal Endotheliitis. Cornea. 2016;35(5):596-601. doi:10.1097/ICO.0000000000000791
- ↑ Hwang JH, Ha M, Park Y, Chung SH. The effect of topical ganciclovir and corticosteroid on cytomegalovirus corneal endotheliitis in Korean patients. Ocul Immunol Inflamm. 2019;27(2):338–44.
- ↑ 37.0 37.1 Basilous et al. Topical Ganciclovir for Prophylaxis of Cytomegalovirus Endotheliitis in Endothelial Keratoplasty. Cornea. 2019 Jan;38(1):120-122. doi: 10.1097/ICO.0000000000001797.
- ↑ Kitazawa K, Jongkhajornpong P, Inatomi T, Koizumi N, Kayukawa K, Wakimasu K, et al. Topical ganciclovir treatment post-Descemet’s stripping automated endothelial keratoplasty for patients with bullous keratopathy induced by cytomegalovirus. Br J Ophthalmol. 2018;102(9):1293–7.
- ↑ Choi et al. A case of CMV endotheliitis treated with intravitreal ganciclovir injection. Korean J Ophthalmol. 2013 Apr;27(2):130-2. doi: 10.3341/kjo.2013.27.2.130. Epub 2013 Feb 27.
- ↑ Yu et al. Clinical Evaluation of Intravitreal Injection of Ganciclovir in Refractory Corneal Endotheliitis.
Ocul Immunol Inflamm. 2020;28(2):270-280. doi: 10.1080/09273948.2019.1573261. Epub 2019 Feb 26. - ↑ 41.0 41.1 ANG M, SNG CCA, SOON-PHAIK CHEE, TAN DTH, MEHTA JS. Outcomes of Corneal Transplantation for Irreversible Corneal Decompensation Secondary to Corneal Endotheliitis in Asian Eyes. American Journal of Ophthalmology. 2013;156(2):260-266. doi:10.1016/j.ajo.2013.03.020
- ↑ Fernández López E, Chan E. Descemet stripping automated endothelial keratoplasty outcomes in patients with cytomegalovirus endotheliitis. Cornea. 2017;36(1):108–112.
- ↑ Chan A, Mehta J, Al Jajeh I, Iqbal J, Anshu A, Tan D. Histological features of cytomegalovirus-related corneal graft infections, its associated features and clinical significance. Br J Ophthalmol. 2015;100(5):601–6.
- ↑ Koizumi N, Suzuki T, Uno T, Chihara H, Shiraishi A, Hara Y, et al. Cytomegalovirus as an etiologic factor in corneal endotheliitis. Ophthalmology. 2008;115:292–7.
- ↑ Wong VW, Chan CK, Leung DY, Lai TY. Long-term results of oral valganciclovir for treatment of anterior segment inflammation secondary to cytomegalovirus infection. Clin Ophthalmol. 2012;6: 595–600.
- ↑ Su CC, Wang IJ, Chen WL, Lin CP, His B, Hu FR. Topical ganciclovir treatment in patients with cytomegalovirus endotheliitis receiving penetrating keratoplasty. Clin Exp Ophthalmol. 2013;41: 339–47.
- ↑ Hwang YS, Shen CR, Chang SH, Lai CC, Liu CL, Chen KJ, et al. The validity of clinical feature profiles for cytomegaloviral anterior segment infection. Graefes Arch Clin Exp Ophthalmol. 2011;249(1):103–10.
47. Cone RE, Pais R. Anterior Chamber-Associated Immune Deviation (ACAID): An Acute Response to Ocular Insult Protects from Future Immune-Mediated Damage?. Ophthalmol Eye Dis. 2009;1:33-40. Published 2009 Nov 17.
48. Liliana Moreno Garcia MVF. Bilateral linear endotheliitis: Case report. Arq Bras Oftalmol. 2013;76(2):121-123.
49. Chu HY, Sun CC, Chuang WY, et al. Cytomegalovirus associated corneal endotheliitis after penetrating keratoplasty in a patient with Fuchs corneal endothelial dystrophy. Br J Ophthalmol. 2012;96(2):300-301. doi:10.1136/bjo.2010.182378
50. Amano S, Oshika T, Kaji Y, et al. Herpes simplex virus in the trabeculum of an eye with corneal endotheliitis. Am J Ophthalmol 1999;127(6):721-2.
51. Hsiao CH, Hwang YS, Chuang WY, et al. Prevalence and clinical consequences of cytomegalovirus DNA in the aqueous humour and corneal transplants. Br J Ophthalmol. 2018;bjophthalmol-2018-312196. doi:10.1136/bjophthalmol-2018-312196
52. Myhre, H.-A., et al. Incidence and outcomes of ganciclovir-resistant cytomegalovirus infections in 1244 kidney transplant recipients. Transplantation.2011;92:217– 223

