Vitreous Wick Syndrome
All content on Eyewiki is protected by copyright law and the Terms of Service. This content may not be reproduced, copied, or put into any artificial intelligence program, including large language and generative AI models, without permission from the Academy.
Disease Entity
Vitreous wick syndrome, "vitreous tug syndrome" as reported by Iliff[1], or vitreous touch syndrome, is characterized by vitreous bands or strands to the corneal wound which can be associated with cystoid macular edema (Irvine-Gass Syndrome).
Disease
Vitreous wick syndrome occurs after eye surgery or trauma and is characterized by microscopic wound breakdown accompanied by vitreous prolapse that develops into a vitreous 'wick'.
At first, vitreous wick syndrome was limited to anterior-segment procedures. Subsequently, however, posterior fistulous tracts with vitreous entrapment were reported after vitreoretinal surgery. Vitreous wick syndrome has also been identified as a potential cause of endophthalmitis after intravitreal injection of triamcinolone through the pars plana.[2]
With the rise of intravitreal drug delivery devices currently available, vitreous wick syndrome may become more common.
Etiology
Vitreous wick syndrome may result from the following situations:
- Routine cataract surgery with unrecognized posterior capsular rupture or zonular dehiscence with vitreous prolapse and adhesion to the surgical wound
- Complicated cataract surgery with posterior capsular rupture and inadequate anterior vitrectomy with adhesion to the surgical wound
- Transconjunctival/transscleral intravitreal injection of pharmacologic agents
- Sutureless small-gauge pars plana vitrectomy
- Inadequate prolapsed vitreous removal during repair of scleral or sclerocorneal lacerations or scleral melts
Risk Factors
Both in the United States and throughout the world, vitreous wick syndrome is rare. No age predisposition has been documented for this syndrome. No gender predisposition has been identified, and the condition has no apparent racial predilection.
Pathophysiology
Vitreous wick syndrome develops in the setting of trauma, either iatrogenic or non-iatrogenic. Vitreous wick syndrome of iatrogenic origin usually follows anterior-segment surgery, though it may also follow sub Tenon injection and muscle surgery if sclera is perforated. Microscopic wound breakdown has been hypothesized as the “point of no return” for the development of vitreous wick syndrome—a point emphasized by Ruiz and Teeters in their initial description.[3]
Corneal wound healing has been documented to be slower on the endothelial side (inner layers). Poor suturing technique is implicated as a major factor for wound breakdown. Tightly compressed corneal wound edges may demonstrate puckering and also may lead to enlargement of suture tracts, promoting tissue necrosis within the suture loop.[3]
Once communication between the posterior wound gap and the anterior wound defect occurs (subsequent to tissue necrosis from tight sutures), anterior aqueous fluid may egress; vitreous incarceration may also occur, producing the vitreous wick. Occasionally, complete sloughing of strangulated tissue within the suture loop may occur.[3]
Non-iatrogenic traumatic causes involve sharp injuries, intraocular foreign bodies, or blunt injuries causing increased intraocular pressure and inside out injury leading to open globe injury (rupture). Neetens et al reported an 8-year-old girl who was hit by a sharp object that perforated the upper lid and caused a black eye.[4] A surgeon repaired the palpebral wound, and the child was not referred to an ophthalmologist. The girl reported vision loss 2-3 weeks later. The injury resulted in a microperforation of the globe through the conjunctiva and sclera.
Diagnosis
History
Symptoms of vitreous wick syndrome may include the following:
- Pain
- Blurring of vision
- Itching or foreign body sensation
- Gush of warm fluid
The past ocular history is commonly notable for the following:
- Recent eye surgery
- Recent eye trauma
Physical examination
Gross findings on physical examination may include the following:
- Mucous threadlike substance protruding from a surgical site
- Corneal haze
- Eye redness
- Eye discharge
- Hypopyon
Slit-lamp findings may include the following:
- Externalized vitreous at the wound site
- Necrotic area around the vitreous wick
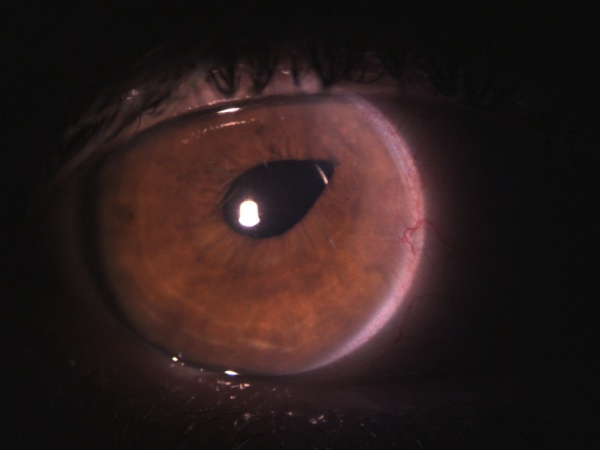
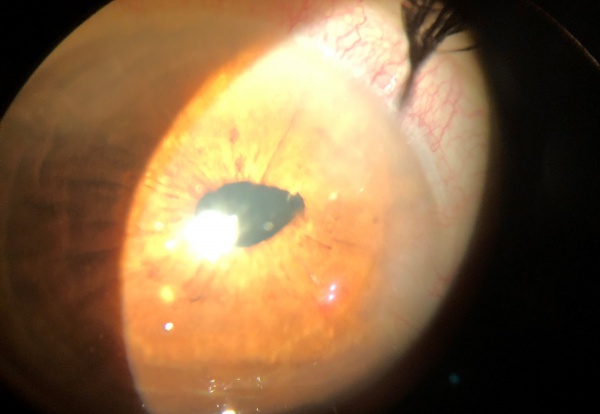
- Corectopia (peaked pupil, Figure 1)
- Presence or absence of cells and flare
- Positive Seidel test
- Corneal haze
- Hypopyon
Clinical diagnosis
Diagnostic procedures
In the Seidel test, a strip of fluorescein is placed on the area that is suspected of containing a leak, and the dye color is then observed in white light. If a leak is present, the dye changes from orange (concentrated) to green (diluted) and exhibits a waterfall-like effect at the leaking zone. The egress of fluid is noted best under blue light.
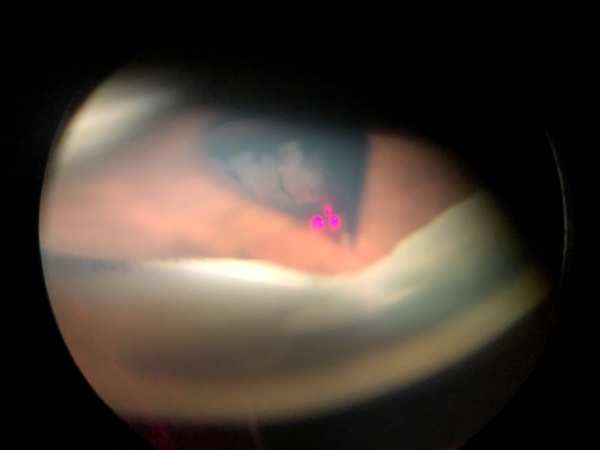
On slit-lamp biomicroscopy, the vitreous wick may have the appearance of a mucoid substance. The examiner may tease the wick with a cotton applicator or a cellulose sponge while taking note of synchronous movement of the iris or of the vitreous strand in the anterior chamber. A peaked pupil may also indicate a vitreous strand in the anterior chamber (Figure 1, 2).
Laboratory test
Depending on the presentation of vitreous wick syndrome specimens (eg, swab, vitreous wick, or aqueous) should be obtained from the external and internal eye for the following studies:
- Gram stain or Giemsa stain
- Cultures and sensitivities
- Calcofluor white (if fungal infection is suspected)
Differential diagnosis
All these different pathologies must be considered when presented with a vitreous wick syndrome:
- Bacterial or Postoperative Endophthalmitis
- Intraocular Foreign Body (IOFB)
- Iris Prolapse
Management
Treatment of vitreous wick syndrome is primarily surgical but may also include medical therapy as appropriate. The surgical approach to the management of this syndrome depends on the presentation (without any restrictions on diet indicated). Participation in strenuous activities and contact sports should be restricted until recovery is complete.
Medical therapy
The type of topical antibiotics used in the treatment of vitreous wick syndrome depends on the suspected infecting agent or the culture and sensitivity results.
In cases of endophthalmitis, medical therapy is initiated and selected according to its effectiveness against the suspected or confirmed (via culture and sensitivity results) infecting agents. Subconjunctival and intravitreal antibiotics should be given (and pars plana vitrectomy should be considered alongside medical therapy).
Surgical procedures
Laser therapy[5]
Patient Preparation
Explain the procedure beforehand to the patient and obtain informed consent. Remind the patient the procedure often requires more than one session.
When the vitreous strand or band passes through the pupil, treatment is often facilitated by administration of pilocarpine 2% every 10 minutes for 3 or 4 drops preoperatively. Inducing stretch of the vitreous through miosis facilitates identification of the strand. When the laser transects the strand the release of tension is shown more definitively.
Procedure
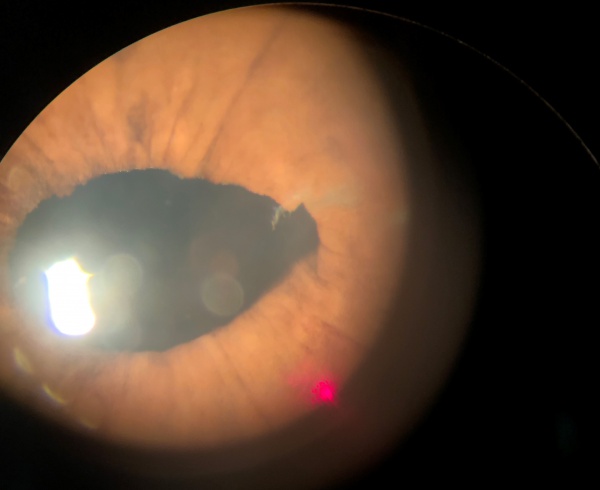
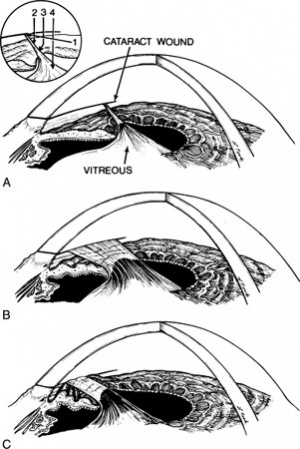
Figure 3 illustrates the three most common configurations of vitreous to the wound:
- A small discrete strand
- A broad band
- A band with either adhesions to the iris or iris entrapment behind the band.
The laser can be directed at a vitreous strand in 4 general areas (inset, Figure 3A):
- Pathway 1. The cataract wound is the most reliable landmark during vitreolysis because the vitreous band or strand has to terminate at that location. The cataract wound is visualized with a gonioscopy lens (pathway 1 on figure), and the laser can be fired at the wound area with a reasonable chance of successful vitreolysis. The energy settings are usually in the 6- to 12-mJ range to obtain adequate cutting power. Lower energy should be tried first. Disadvantages of this technique include the requirement of a gonioscopy lens, and the need to use a contact lens for the completion of the treatment session––even if a different approach is needed later in the session––because of the application of gonioscopy fluid to the cornea.
- Pathway 2. If the cornea is clear near the limbus and a vitreous strand can be visualized with clearance from the iris stroma, direct cutting without a contact lens or with a peripheral button Abraham lens may be successful along pathway 2. Usually 4 to 8 mJ is required. In the course of dozens to hundreds of shots along this pathway, considerable pigment may be liberated from the underlying iris stroma, which will ultimately obscure the surgeon's view. Misfocused shots can cause local damage to the underlying or overlying corneal stroma or underlying iris.
- Pathway 3. If directed at the vitreous passing over the iris collarette, pathway 3 can be helpful, especially when the vitreous has formed adhesions to the collarette, pulling it forward in a tent-like formation. Close proximity of vitreous and iris makes damage to the underlying iris stroma likely, but this may be clinically tolerable.
- Pathway 4. Pathway 4 is rarely successful if directed at the vitreous as it passes around the pupil. Vitreous traction components are poorly defined as they come around the pupil. The shock wave is ineffective at rupturing vitreous strands except directly at the laser focal point. Firing the laser immediately adjacent to the pupillary border causes low-grade capillary hemorrhage and release of pigment, obscuring visualization of the area.
Successful treatment releases the tension and converts a discrete strand or band to an amorphous gelatinous appearance. Observation of the change and any iris deformation is the best indicator of a successful release of tension. Hundreds of shots over several treatment sessions may be necessary to cut a large band. If cystoid macular edema (CME) does not respond to YAG laser vitreolysis alone, posterior sub Tenon triamcinolone, or intravitreal triamcinolone, or intravitreal steroid implants may be needed to control CME.
Postoperative laser care
Strong topical steroids (eg, prednisolone acetate 1% or dexamethasone 0.01%) are given 4 times daily until visual improvement occurs, typically in 2 to 3 months. Topical nonsteroidal anti-inflammatory drugs may be of benefit alone or with topical steroids. Ketorolac or diclofenac drops are usually administered 4 times daily. The addition of a systemic nonsteroidal anti-inflammatory drug can be considered in recalcitrant cases.
IOP elevation following vitreolysis has not been well documented. A beta-blocker or brimonidine drop at the time of treatment probably provides adequate prophylaxis.
Prophylactic Vitreolysis
With the availability of laser vitreolysis, the surgeon may treat vitreous strands to the wound in the absence of cystoid macular edema (CME) in an effort to prevent its later development. Only a large, long-term randomized treatment trial can determine the usefulness of this approach. Some patients with vitreous strands to the wound never acquire CME.
A baseline fluorescein angiogram and optical coherence tomography may be done to document the macular status in patients with vitreous to the wound and good visual acuity. If CME is detected, laser vitreolysis is probably indicated even in the presence of good acuity. If visual loss later develops with the new onset of CME, the baseline angiogram will be useful to further support therapeutic intervention.
Surgery
The precise surgical treatment varies with the circumstances. The following is a generalized procedural description.
Initially, the vitreous wick is excised or severed with Vannas-type scissors by lifting the exposed vitreous strand with a cotton-tipped applicator or fine nontoothed forceps. Alternatively, a suction-cutting instrument inserted into the anterior chamber may be used.
Vitrectomy may be performed via an anterior limbal approach or a closed posterior approach. It is imperative that no vitreous strand is left above the pupillary plane. To detect any remaining vitreous, one should sweep the anterior chamber with a spatula from a paracentesis site 90° away from the surgical wound.
Intracameral injection of pupil constrictors (carbachol intraocular solution) may help pull any remaining anterior-chamber vitreous wick back into the posterior segment. Intracameral preservative-free triamcinolone acetonide may help visualize vitreous strands.[6][7] A mobile (reacting), well-centered round pupil suggests clearance from any vitreous that is invading the anterior chamber. Adequate surgical closure is accomplished with nylon 10-0 sutures.
The patient is discharged on a regimen of topical antibiotics, either broad-spectrum or targeted on the basis of culture and sensitivity results. Nonsteroidal anti-inflammatory drug (NSAID)-containing ophthalmic drops are given to decrease cystoid macular edema and steroid drops are given to decrease inflammation. Pilocarpine ophthalmic drops are given to maintain pupillary constriction and prevent anterior segment migration of posterior vitreous during the acute healing phase.
Patients should receive follow-up care 1-2 days after surgery. If this initial follow-up examination identifies no problems, regular checkups should be scheduled for uneventful anterior segment surgeries. The use of an eye shield, especially at night, protects the globe from any untoward traumatic episodes.
Prognosis
The prognosis is mainly dependent on the presence or absence of cystoid macular edema.
Katzen, Fleischman, and Trokel reported the use of the Nd:YAG laser to lyse strands of vitreous to cataract wounds. Vision improved in all 14 patients treated. The presence of CME, however, was judged clinically, and the results of fluorescein angiography before and after laser treatment were not reported for 13 of the eyes.[8]
Because of the unpredictable natural history of aphakic CME, with erratic response to antiinflammatory agents and frequent spontaneous improvement, small and controlled series cannot unequivocally prove the efficacy of a given technique. Clinical experience, however, has confirmed a high rate of visual improvement after anterior segment vitreolysis, particularly in favorable cases. In Steinart and Wasson series of 29 patients[9], 22 had fluorescein angiographic confirmation of CME before laser treatment. The interval between cataract extraction and treatment averaged 10 months, with a range of 1 to 42 months; average follow-up after laser vitreolysis was 10 months, with a range of 3 to 27 months. No patient had loss of vision after laser vitreolysis. The visual acuity in 16 of the 29 patients (55%) improved by 2 or more lines, with stable acuity following treatments. Five (17%) patients' vision improved by at least 2 lines, but they experienced ongoing fluctuation of acuity. Vision in 8 patients (28%) showed less than 2 lines of improvement. Of these 8 patients: 2 had progressive maculopathy in addition to the CME (1 had an epiretinal membrane and one had progressive diabetic maculopathy); 2 had severe glaucoma with loss of central vision in addition to the CME; and 2 had persistent CME. Two patients were lost to follow-up without documentation of the basis for persistent unimproved acuity.[9]
Patients who did not respond to vitreolysis had the poorest pretreatment visual acuity measurements.[9]
Post-laser fluorescein angiography was performed on 9 eyes. Three eyes showed complete resolution of the CME; 2 showed improvement but persistent leakage; 1 showed an unimproved appearance of the fluorescein angiographic leakage but still experienced improved acuity; and 3 had persistent CME and less than 2 lines of visual improvement.[9]
References
- ↑ Iliff CE. Treatment of the vitreous-tug syndrome. Am J Ophthalmol. 1966 Nov;62(5):856-9.
- ↑ Chen SD, Mohammed Q, Bowling B, Patel CK. Vitreous wick syndrome--a potential cause of endophthalmitis after intravitreal injection of triamcinolone through the pars plana. Am J Ophthalmol. 2004 Jun. 137(6):1159-60; author reply 1160-1.
- ↑ 3.0 3.1 3.2 Ruiz RS, Teeters VW. The vitreous wick syndrome. A late complication following cataract extraction. Am J Ophthalmol. 1970 Oct. 70(4):483-90.
- ↑ Neetens A, Rubbens MC, Smets RM. Vitreous wick syndrome. Bull Soc Belge Ophtalmol. 1987. 223 Pt 2:41-5.
- ↑ https://www.aao.org/munnerlyn-laser-surgery-center/ndyag-laser-in-managing-postcataract-surgery-compl
- ↑ Couch SM, Bakri SJ. Use of triamcinolone during vitrectomy surgery to visualize membranes and vitreous. Clin Ophthalmol. 2008 Dec. 2(4):891-6.
- ↑ Schmidt JC, Chofflet J, Hörle S, Mennel S, Meyer CH. Three simple approaches to visualize the transparent vitreous cortex during vitreoretinal surgery. Dev Ophthalmol. 2008. 42:35-42.
- ↑ Katzen LE, Fleischman JA, Trokel S. YAG laser treatment of cystoid macular edema. Am J Ophthalmol. 1983 May;95(5):589-92.
- ↑ 9.0 9.1 9.2 9.3 Steinert RF, Wasson PJ. Neodymium:YAG laser anterior vitreolysis for Irvine-Gass cystoid macular edema.