All content on Eyewiki is protected by copyright law and the Terms of Service. This content may not be reproduced, copied, or put into any artificial intelligence program, including large language and generative AI models, without permission from the Academy.
Background:
Posterior Capsular Rent (PCR), is a breach in the posterior capsule of the crystalline lens during cataract surgery. A common yet feared complication of cataract surgery, posterior capsular rent may lead to sub-optimal visual outcomes If not recognized early or managed appropriately. The consequences of this complication vary with the stage at which it occurred or was recognized, the coexisting pathologies in the operated eye, expertise of the operating surgeon and the facilities available to manage it. Consequent vitreous loss is also a major determinant of post-operative outcomes.
Incidence:
Incidence of PCR is variable and the reported numbers are between 0.2% to 14% whereas, the rate of vitreous loss is between 1 and 5%.[1] For experienced surgeons, the rate of PCR ranges from 0.45-5.2%.[2] [3]
Risk Factors:
Most factors that predispose a patient to a PCR can be identified in the pre-operative planning phase. These may be categorized into patient related, surgeon related, and machine related risk factors.
Patient Related:
General Factors
Restless, anxious, elderly, obesity, COPD (chronic obstructive pulmonary disease), demented patients or patients with inadvertent head movements.
Age: A steady rise in rent rate has been reported with age, with a much higher risk of vitreous loss and retained nuclear fragments in patients 88 years or older. [4][5]
Extraocular Factors
Deep set eyes with a prominent brow: These features make surgical maneuvering difficult. A temporal approach may be superior to a superior one in such cases. Deep set eyes may also allow pooling of irrigating solution over the ocular surface causing reflections and reducing the visibility during surgery. Conjunctival ballooning can occur with subconjunctival collection of irrigation fluid resulting from the construction of a posterior wound in phacoemulsification.
Exaggerated Bell’s phenomenon: Due to this the eye moves upwards and keeping the eye in the field of operating microscope may be difficult.
Intraocular Factors
Corneal opacities with visibility issues: Conditions like pterygia, Fuchs endothelial dystrophy, poor ocular surface, epithelial toxicity of topical drugs or povidone iodine, corneal scarring, prominent arcus, spheroidal degeneration, band keratopathy etc. may hinder visualization. It is advisable to slow down and pay careful attention to visualization during critical steps by focusing around the corneal problem. Light pipes can also be used to assist in visualization in cases with significant corneal pathology. Dense asteroid hyalosis, vitreous hemorrhage, retinal detachment, posteriorly displaced trypan blue dye (used to stain anterior capsule before capsulorrhexis) may also hinder visualization of the posterior capsule due to reduced retroillumination and red reflex. Displacing the globe, tilting the microscope and altering entry wound location can be done to maximize view in such cases.
Type of cataract: certain types of cataracts increase the potential for posterior capsular rupture. These are:
- Intumescent Cataract[6]
- Brunescent/Black Cataract
- Posterior Polar Cataract
- Cataract associated with posterior lenticonus or lentiglobus [7]
- Traumatic Cataract
- Post-vitrectomy cataract
Pseudoexfoliation: is associated with weak zonules, non-dilating pupils and a thick fibrosed posterior capsule increasing incidence of posterior capsular rupture and zonular dehiscence.
Small Pupils: reduce the working space and hinder visibility.[8] Causes are:
- Intraoperative Floppy Iris Syndrome (IFIS): history of alpha-1 blocker use
- Posterior Synechiae
- Long-term use of topical Pilocarpine
- Pseudoexfoliation
- Diabetes Mellitus
- Senescence
- Congenital: Coloboma, iridoschisis
Shallow Anterior Chamber:
In a crowded shallow Anterior chamber (AC) for example, in hypermetropes, the phaco needle is closer to the posterior capsule with a greater likelihood of PCR. Iris ballooning due to fluid flowing posteriorly may cause frequent iris prolapse. Possible solutions are a narrow incision with a longer tunnel, a more anterior incision and use of second instrument only when required.[9]
Deep Anterior Chamber:
High myopia: A trampolining posterior capsule due to thinner and more elastic tissues, lack of scleral rigidity and zonular laxity.
Post pars plana vitrectomy eyes: lack of vitreous support.
Lens Iris Diaphragm retropulsion syndrome (LIDRS): The mechanism is reverse pupillary block with excessive anterior chamber deepening. If a pronounced post-occlusion surge occurs, a sudden shallowing of AC may follow and PC Rent may occur.
Solutions: This rapid fluctuation in AC depth may be controlled by keeping BSS (Balanced Salt Solution) bottle height, vacuum and flow rate settings lower than usual. Alternatively, a second instrument such as an iris hook can be used to manually lift the iris from the surface of the lens to distribute pressure. [10]
Machine Related:
- Unfamiliar devices
- Poorly controlled machine settings
- Sudden intraoperative machine dysfunction
- Surge
- Suboptimal alignment or inadequate brightness of operating microscope leading to poor visualization
- Interruption of infusion flow causing sudden AC collapse (bottle finishing midway, loose irrigation line, or kinks in tubing)
Surgeon Related:
Inexperienced Surgeons:
PCR incidence in phacoemulsification for experienced surgeons is 0.45-3.6 % [2] compared to that performed by surgeons newly converting from extracapsular cataract extraction to phacoemulsification, reported to be 4.8% to 11%. [2][4] Resident performed surgeries have had documented rates between 0.8 to 8.9%. [11][12][13][14]
Anatomical Considerations:
The Lens
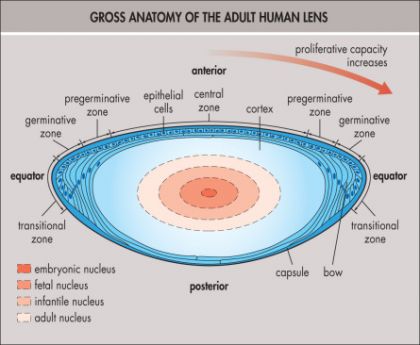
The human crystalline lens is a biconvex transparent structure which is enclosed in a thin capsular bag and is located in front of the vitreous body and behind the iris. It is suspended on all sides with zonules which support it and connect it to the ciliary body. The lens continues to grow throughout life.
Structure:
Histologically, It is composed of three structures
1. Capsule
· An elastic basement membrane made of Type IV collagen and glucosaminoglycans which is produced by epithelial cells of the lens.
· Thickest at the anterior and posterior pre-equatorial zones (20 microns) and thinnest in the central posterior pole (2-4 microns). This thinness of the posterior capsule makes it vulnerable to rupture. [15]
· Anterior capsule thickness increases with age while the posterior capsule diminishes with age except for posterior polar region which remains stable.[16]
· Posterior capsule functions like a physical barrier between the anterior and posterior segments of the eye and limits spread of inflammation and infection between the two. [17]
2. Anterior subcapsular epithelium
· A single layer of undifferentiated, nucleated columnar cells.
· Metabolically active cells which carry out all cellular functions including RNA, DNA, lipid and protein synthesis and generation of ATP.[18]
3. Lens substance (Cortex and nucleus)
· Composed of a soft cortical peripheral part and a central firm nucleus
· When the lens becomes more firm with age, it can be seen as concentrically arranged laminae made of ribbon-like fibers.[19]
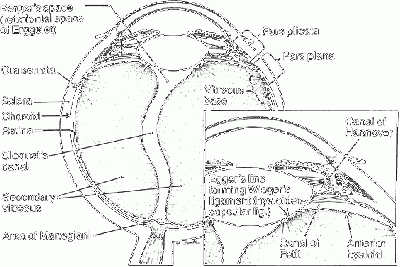
The Retrolental Region:
It has two potential spaces
· Space of Berger/Errgelet’s space: Central
· Petit’s canal: Peripheral
· Weiger’s Ligament: The two spaces are concentric and are separated by a false ligament which is in reality a circular adhesion of the anterior hyaloid membrane to the posterior capsule.[20]
· Egger’s Line: This line of adhesion is often termed Egger’s line and the strength of this adhesion diminishes with age.[21]
The Vitreous[21][22]
A colorless, gel-like structure, the vitreous is about 4 ml and occupies 80% of the volume of the eye.
Boundaries:
It is bound anteriorly by the retrozonular part of the anterior segment and the posterior surface of the lens anteriorly. Posteriorly, from the posterior pole to periphery it is bounded by the optic disc, the retina and the pars plana of the ciliary body.
Surfaces:
The anterior and posterior surfaces of the vitreous are known as the anterior and posterior hyaloid faces respectively.
Zones:
To simplify, the vitreous can be divided into two zones
Central Zone: is more liquified, has fewer number of collagen fibrils and less hyalocytes as compared to the outer zone. It is traversed by a fluid-filled S-shaped canal (Cloquet’s canal) which extends from the optic nerve to the posterior surface of the lens.
Outer Zone: Vitreous collagen is primarily found in the outer zone known as the cortex, which is a shell like structure that is nearly absent in front of the optic nerve and macula.
Attachments:
There are localized condensations or adhesions of the vitreous with Internal limiting membrane of the retina and the pars plana. These adhesions vary in strength are from strongest to weakest are:
· Ora Serrata (vitreous base)
· Posterior lens capsule (hyaloideocapsular ligament of Weiger)
· Margins of the optic disc
· Macula
· Along retinal vasculature (variable)
Margins of the Vitreous Base:
5mm posterior to the limbus in all meridians
Extends posterior to the ora serrata for about 2 mm in the temporal and 3 mm in the nasal quadrants.
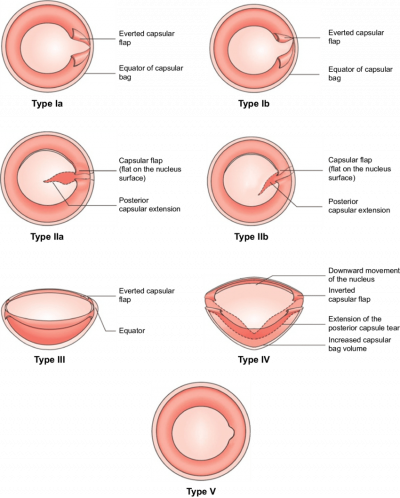
Classification of capsular tears[23]
| Types of Capsular Tears According to Location | |
| IA | Perpendicular Pre-equatorial |
| IB | Acute-Angled Pre-equatorial |
| IIA | Perpendicular Post-Equatorial |
| IIB | Acute-Angled Post-Equatorial |
| III | Pre-equatorial with Argentinian flag sign |
| IV | Post-equatorial with Argentinian flag sign |
| V | Mini Punch |
Prevention of Posterior Capsular Rent:
Prophylaxis of a posterior capsular rent begins with comprehensive pre-operative evaluation and preparation. A continuous assessment of the intraoperative situation during phacoemulsification while keeping a low threshold of suspicion for a PCR may help in early recognition of this event.
Pre-operative Evaluation:
History:
- Previous ocular trauma
- Surgical procedures
- Ocular and systemic, recent or chronic use of medications
Examination:
- A thorough slit lamp exam to analyze ocular adnexa and surfaces, corneal transparency, AC depth, anterior and posterior capsule integrity, zonular status and grade of cataract. The fellow eye should also be examined (especially looking for complicated cataract surgery).
- Intraocular pressure
- Dilated fundoscopy for evaluation of posterior segment
- Specular microscopy for corneal endothelial health (If history of previous surgery, hard cataract, trauma or a very elderly patient)
Anesthesia:
Peribulbar anesthesia instead of topical is preferable in anxious patients. Sedation may help excessively anxious patients. In phacomorphic glaucoma, intravenous mannitol should be administered to reduce IOP (intraocular pressure) and vitreous upthrust. General anesthesia is preferred for children, patients with neurologic disorders or mentally unstable patients.
Identifying High Risk cases:
Pre-operative identification of the cases likely to evolve into unwanted complications can help the surgeon take special precautions preemptively.
Systemic:
- Musculoskeletal problems hampering prolonged supine positioning of the patient
- Neurological disorders affecting patient cooperation
- Cardiopulmonary conditions causing anxiety and breathing problems during surgery
- A short neck or obesity can increase vitreous pressure leading to instability of the anterior chamber
- Use of medications such as alpha blockers responsible for Intra-operative Floppy Iris Syndrome.
Ocular:
- Smaller palpebral fissures or deep-set eyes causing difficulties in placing speculum. Pooling of fluid which commonly occurs in deep sockets, causes reflections which compromise visibility and limit surgeon’s maneuverability
- Excessively shallow or Deep AC
- Non-dilating pupil or synechiae
- Fibrotic or breached capsule. Capsules in children are more elastic posing a challenge during capsulorhexis.
- Subluxated lens, zonular dehiscence or weakening e.g., pseudoexfoliation.
- Ocular comorbidities like silicone oil-filled vitrectomized eyes, glaucoma, previous trauma or uveitis.
Surgical Strategies to Minimize Risk of a Posterior Capsule Rent
Draping and Speculum
The drape should not allow exposure of lashes and should not obscure the surgical field. The speculum should be of appropriate size and should ideally be lightweight as to avoid undue pressure on the globe leading to increased posterior segment pressure against the lens.
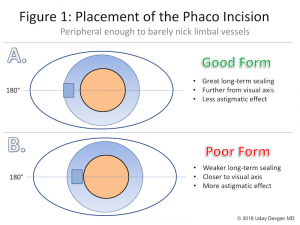
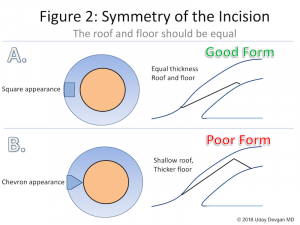
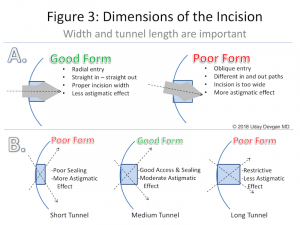
Wound construction
Precise wound construction is pivotal in determining the course of events in phacoemulsification. Triplanar incisions are ideal since they resist leakage and are self-sealing. Too small an incision can cause wound burn and localized corneal edema. The sleeve size should be compatible with the wound width to make gliding of instruments easy. Inappropriately wide incisions lead to chamber instability, especially during high flow states and during emulsification of the last nuclear remnant. Such an incision can be sutured and another site chosen to reconstruct a proper incision.
[Video Credit- Uday Devgan. cataractcoach.com]
Phaco-tip and Sleeve
The probe tip and sleeve should match each other in size as well as with the wound size. Greater bevel angulation facilitates sculpting whereas, a flat tip is more suited for occlusion.
Maintenance of Intraoperative visibility
Use of a microscope with good stereoscopic depth perception and coaxial illumination is recommended. Pooling of fluid should be prevented by ensuring proper head positioning allowing for escape of fluid. Two percent Hydroxypropyl Methylcellulose is found to be superior to Balanced Salt Solution (BSS) to prevent corneal drying and haze.[24]
Non-Dilating pupils
Intracameral adrenaline, Malyugin ring, B-Hex ring, other pupil expansion devices or iris hooks can greatly help enlarge pupils intraoperatively. Posterior synechiae can be released by instilling Ophthalmic Viscosurgical Device (OVD) and using Sinskey hooks to break adhesions. A well-dilated pupil prevents PCR by allowing a wider view of the rhexis margins and the posterior capsule.
Handling Anterior Capsule
An intact Continuous Curvilinear Capsulorhexis (CCC) is fundamental in preventing a capsular breach. To minimize complications, beginners should aim for a larger capsulorhexis (5-5.5mm) and use Trypan Blue to stain the anterior capsule despite a good fundal glow. Vannas scissors may be required to cut the fibrotic areas in the anterior capsule during capsulorhexis to prevent lateral extension.
Small fibrotic central plaques may be included within the rhexis margins.
Posterior run-offs are more likely to occur in myopic eyes with deeper bags. For capsulorhexis in intumescent cataracts with high intra-lenticular pressure, techniques such as two-stage capsulorhexis, excessive use of viscocohesive OVDs, trypan blue dye and chandelier endoillumination systems have been described.[25][26][27][28] Femtosecond-laser assisted capsulotomy is considered safer and more convenient in intumescent cataract cases.[29]
Zonular insertions on the anterior capsule may sometimes be long enough to be traversed by the leading capsulorhexis margin resulting in zonular disinsertion.[30] Presence of anterior zonules can easily be determined on a pre-operative slit-lamp exam with a well-dilated pupil.
Pediatric capsules are highly elastic and use of Utrata forceps or micro-capsulorhexis forceps may be preferable over traditional cystotome needles to attain better control. Similarly, forceps may allow for greater control in subluxated lenses where CCC should be centered on the lens rather than on the pupil to avoid decentration.
Hydrodissection
This step should be instituted with great caution. Before commencing, the anterior chamber should be decompressed by allowing outflow of some of the OVD through the incision. To prevent a sudden increase in intralenticular pressure, small amounts of fluid should be introduced beneath the anterior capsule and the nucleus gently tapped to release trapped fluid. Aggressive hydrodissection in the presence of a small capsulorhexis can lead to Capsular Block Syndrome and consequently a PCR. Cortical cleaving hydrodissection reduces the need for cortical removal thereby preventing capsule rupture.
Hydrodelineation
This step separates the nucleus from the epinucleus, which acts like a protective shell during trenching, cracking and emulsification, and prevents inadvertent direct entry of the posterior capsule into the probe tip. A thorough hydrodelineation with multiple rings enhances safety by providing cushioning especially in posterior polar cataracts. [31]
Grooving
A Posterior breach is more likely to occur in very soft or very hard cataracts. Beginners with less foot pedal control, use of a burst phaco mode and a very deep linear trench are risk factors for a PCR. A change in red reflex seen in the floor of the trench is a good sign of adequate groove depth.
Irrigation and Aspiration
The I/A probe tip (opening) should not be directed towards the posterior capsule to prevent its aspiration or rupture. Sleeve-tip distance should be adjusted and special care should be taken to not let the anterior chamber collapse in eyes with fragile posterior capsules e.g, posterior polar cataracts. Cases with inadequate hydrodissection may require extensive I/A further increasing the risk. Accidental aspiration of PC into the I/A tip may result in visible converging folds (Spider Sign). On identification, they should be released immediately using the reflux function of the foot pedal.
Management of Subluxated Lens
<3 clock hours of subluxation- Capsular Tension Ring (CTR) is required to redistribute force on intact zonules and keep the capsular bag stretched. CTRs should be avoided in the presence or anterior or posterior capsular tears.
>3 clock hours of subluxation- Capsular Tension Segments (CTS) or Cionni Rings are needed to stabilize the lens.
IOL Insertion
Before inserting the IOL, the capsular bag should be filled with OVD to push the posterior capsule away followed by a slow and controlled injection. Automated injectors are preferable to manual ones which should be thoroughly examined for smoothness before loading and injecting the IOL. For sulcus placement, cohesive OVDs should be injected behind the iris to make space
Lower fluidics and Slow Phacoemulsification
Low aspiration flow rate, phaco power and vacuum settings and infusion pressure allow for reduced pressure differences during various steps of surgery, decreasing risk of complications like PC rents, iris tears and vitreous prolapse. [32] This may be especially useful for inexperienced surgeons. Decreasing parameters while emulsifying the last nuclear piece in a hard cataract, in posterior polar cataracts, zonular laxity and IFIS all reduce the risk of complications.
Management of Posterior Capsular Rent
The precise management varies greatly with the timing and extent of rupture and the intraocular circumstances at that point. The following management steps are a compilation of various ideas and experiences presented in textbooks and articles. Choice of approach may vary considerably depending on surgeon’s comfort and expertise and the facilities available.
Early Recognition
Recognizing the occurrence of a Posterior Capsule Rent early is vital to its optimum management and outcome.
- Pupil snap sign (sudden pupillary constriction during hydrodissection)[33]
- Sudden deepening of Anterior chamber
- Striae of the posterior capsule (usually due to extended anterior capsular tear) [34]
- Improved Red Reflex (especially If localized)[35]
- Tremulousness of the nucleus and difficulty in rotating a previously mobile nucleus due to vitreous within capsular bag
- Movements of pupillary margin inconsistent with AC manipulation by instruments
- Excess sideways tilt of the lens or IOL
- Strands of vitreous in AC
If the above signs are missed and phacoemulsification is continued, other signs may indicate presence of a PCR. Sculpting may become more difficult and there may be tilting of the nucleus, eventually leading to a drop into the posterior segment. The behavior of the nucleus after a PCR depends on the size of breach, AC pressure and status of the vitreous. Peaking of the pupil due to vitreous entrapment in the wound is another sign of vitreous loss. Confirming presence with a Weck-Cel was previously recommended, though should be avoided if possible as this results in traction on the retina and possible retinal breaks.[36]
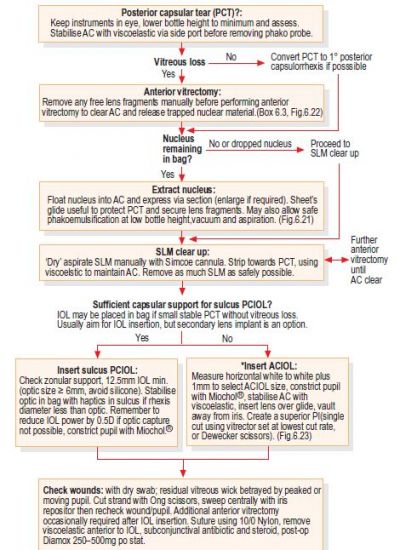
Principles of Management:[37]
Stabilize the Anterior Chamber
Whenever a PCR is suspected, it is imperative to not withdraw the instruments from the AC. This can lead to a collapsed AC with possible extension of the tear, vitreous prolapse and displacement of lens fragments into the posterior segment.
If there is no imminent danger of the nucleus fragments falling into the posterior chamber, the second instrument should be carefully withdrawn and viscoelastic injected through the side port incision.
Switch to Low Fluidics
Machine settings should be significantly reduced once a case gets complicated with a PCR.
· Flow Rate: Lower flow rate feeds less fluid and fragments into the probe, increases surgeon’s control over the case and narrows intraocular pressure variations. The risk of nuclear fragments falling posteriorly due to fluid flow also decreases.
· Irrigation Pressure: High pressures can also push lens fragments posteriorly. Bottle height should be lowered to reduce turbulence within the AC.
· Vacuum: High vacuum can lead to higher post occlusion surge attracting a sudden large volume of fluid from the AC into the tubing, causing the AC to collapse and again lead to lens drop.
I/A should also be done on significantly low settings.
Lens Emulsification in AC using Viscoshield Barrier
A large amount of dispersive viscoelastic can be injected through the posterior capsule to create an effective barrier between the vitreous and contents of the AC. This allows for safer phacoemulsification of the remaining lens fragments in the AC.
Vitreous Detection Before I/A
Presence of vitreous can be ascertained by injecting triamcinolone (approx. 10 times diluted) into the AC.[38] It vitreous is detected, careful anterior vitrectomy should be done. Before Irrigation and Aspiration it is crucial to ascertain the absence of vitreous in the AC, since undue forces on vitreous strands can lead to retinal traction and detachment.
Management of Posterior Capsule Rent without Vitreous Loss
The primary aim in such a case is to (a) Prevent enlargement of tear (b) Prevent vitreous prolapse into AC
1. Immediately cease both the Phacoemulsification and aspiration functions without removing any instruments from the eye. The irrigation should be kept on (footpedal position 1) to prevent AC from collapsing.
2. A dispersive OVD should be introduced through the side port without retracting the phaco probe. The positive pressure fully forms the AC, prevents the tear from enlarging and vitreous from coming forward. A cohesive OVD has limited use in this situation.
3. If there is danger of losing nuclear fragments to the PS (posterior Segment), aspiration should be kept on (footpedal position 2) to keep the fragment engaged while OVD is injected below the fragment.
4. If nucleus is soft and most of it has been emulsified, the surgeon can proceed with phaco using copious amounts of OVD (viscodispersive) to plug the PC tear and tamponade the vitreous. This can be replenished repeatedly till all fragments have safely been removed.
5. If the nucleus is hard or a large chunk remains, conversion to extracapsular Cataract Extraction (ECCE) or manual Small Incision Cataract Surgery (MSICS) is an option, versus vitreous removal if present in the anterior chamber followed by a scaffolding technique depending on the remaining capsular support.
6. In order to reduce vitreous hydration, all machine parameters should be reduced and I/A done with great care.
7. Once the cortex has been removed, a small amount of diluted triamcinolone should be administered to rule out vitreous prolapse.[38] If herniated vitreous is found, the surgeon should proceed with anterior vitrectomy.
8. If vitreous prolapses at a stage when nucleus or cortex are present, the vitreous should be removed first before attempting phacoemulsification or I/A. This is mandatory to avoid vitreous traction and retinal tears.
9. An appropriate IOL can be implanted thereafter.
Posterior Continuous Curvilinear Capsulorhexis (PCCC)
A small central or midperipheral PCR with a tag can be extended circumferentially to a form a PCCC. This technique was introduced by Gimbel and Neuhann to prevent the formation of Posterior Capsular Opacification (PCO).[39] It gives the posterior capsule strength in the same way as the anterior capsule.[40] It is imperative to fill up the AC just enough to flatten the PC. Overfilling may push the tag posteriorly into the vitreous, making it difficult to initiate the capsulorhexis. The technique is performed using micro-capsulorhexis forceps and is possible only If the PCR is away from the equator. Once an intact PCCC is achieved, IOL can safely be implanted in the capsular bag.
[Video Credit- Dr. Pradip Mohanta]
Management of Posterior Capsule Rent with Vitreous Loss
1. In case of a PCR with vitreous prolapse OVD should first be injected before removing the phaco probe.
2. Nuclear fragments If present should be moved superiorly to a safer zone using OVD.
3. Bimanual Anterior Vitrectomy is carried out to remove all prolapsed vitreous from the AC using a limbal or pars plana approach, depending on surgeon comfort.
4. Once vitrectomy is complete, management is the same as in the above case.
5. If large amounts of nucleus remains, It may be safer to convert to a non-phacoemulsification technique i.e, MSICS or ECCE. [41]
6. If the nucleus has already been divided into multiple pieces, It can be expressed using OVD or delivered with a wire Vectis If the pieces are sufficiently large and no vitreous in present in the anterior chamber. Phacoemulsification should not be attempted in the anterior chamber since It could lead to permanent endothelial decompensation.
7. If sections or whole of the nucleus fall into the vitreous cavity, the surgeon should not insert the phacoemulsification probe or other instruments posteriorly to retrieve them. They should be retrieved by performing a pars plana vitrectomy.
Guiding Principles of Anterior Vitrectomy
The goal of anterior vitrectomy is to clear the Anterior chamber and wound from prolapsed and incarcerated vitreous to prevent traction on the retina and to allow for proper IOL placement. Any remaining strands can lead to disastrous consequences such as endophthalmitis, cystoid macular edema (CME), retinal tears or detachment. [38]
Instrumentation
Vitreous cutters are either biaxial or coaxial systems. Both have their respective advantages and disadvantages. In general, bimanual cutters allow for better patient and surgeon satisfaction.
| Vitrectomy Systems | Advantages | Disadvantages |
| Bimanual | -Financially feasible; easily available equipment.
-Separate Irrigation and vitrector leading to better fluidics and low turbulence -Better tamponade on vitreous through irrigation |
-Difficulty in performing the procedure due to lack of hydration.
-Inadequate irrigation may compromise procedure -additional sideport incision needed |
| Coaxial | Easy set up | Irrigation and cutter in the same systems because of which vitreous gets pushed away from the cutter
Vitreous volume expansion Greater risk of enlargement of PCR Larger amounts of vitreous removal |
Technique
A low volume sub-tenon block can be given before the procedure If the patient was initially given topical anesthesia. Ideally, the cut rate should be highest while the vacuum, Aspiration Flow Rate and bottle height are kept low. A high cut rate for the vitreous and a closed chamber causes vitreous to flow seamlessly into the cutter with less traction induced posteriorly. The phaco wound should be sutured to form the anterior chamber and prevent leakage. A peripheral clear corneal incision for the AC maintainer should be made. Once the vitrector is introduced through a side port incision and a closed system is ascertained, vitrectomy can be initiated. The vitrector should be angled downwards through the PCR while avoiding damaging other intraocular structures.
While performing bimanual vitrectomy, the irrigation should be kept above the cutter and its direction should be towards the angle to cause laminar flow and prevent vitreous hydration. Diluted triamcinolone administration is advisable to visualize vitreous strands better and to ensure complete removal from the wound and AC. Once the vitreous has been cleared, cortical removal and IOL placement can be done. Alternatively, intracameral acetylcholine can be administered to constrict the pupil to rule out vitreous wicks in the wound.
[Video Credit- Uday Devgan. cataractcoach.com]
IOL Selection and Placement
In case of an intact capsulorhexis, sulcus placement of a three-piece IOL is the most preferable method. A single-piece IOL is not angulated and its sharp edges can cause iris chafing causing a host of long term problems. Forceps or injector can be used depending on the surgeon’s comfort and expertise. Optic capture of the IOL through the CCC should be done to stabilize it. An in the bag single-piece IOL placement can be opted for in case of a small posterior rent with successful achievement of a posterior continuous curvilinear capsulorhexis (PCCC).
Since an IOL placed in the ciliary sulcus is anterior to that in the bag, the power needs to be reduced by approximately 1/20th of the power in Diopters (about 0-1.5D). [42] A more accurate nomogram can be found here: Bag to Sulcus Conversion of IOL Power
| Surgical Scenarios | IOL Options |
| Intact capsular bag with a small PCR and no vitreous prolapse | Multi-piece IOL in sulcus |
| Small PCR with successful PCCC | Single-piece IOL in the bag |
| Larger PCR but intact capsulorhexis | Multi-piece IOL in sulcus with optic capture in the capsular bag |
| Insufficient capsular support for PCIOL placement | Plan scleral fixated IOL or iris sutured IOL in same sitting or secondary IOL (ACIOL possible but less preferable) |
| Dropped Nucleus | Clear the anterior segment of vitreous, perform good cortical clean up, put IOL in the primary setting as described previously, do not fish the dropped lens matter, suture the wound; Refer to Vitreo-Retinal surgeon |
An intracameral cefuroxime dose of 1mg in 0.1ml is recommended as in all cases of cataract surgery. [43] This becomes even more relevant in a complicated case with a PCR since the risk of endophthalmitis is six times as high.[44]
Post-Operative Management
- Good patient communication and managing expectations
- Including topical Non-Steroidal Anti-Inflammatory (NSAID) drugs to the standard post-op regimen reduces risk of CME. Addition of a topical IOP lowering agent can also be considered.
- Regular post op follow up
Post-Operative Complications associated with a Posterior Capsular Rent: [45]
Early Complications
- Striate Keratopathy
- Corneal edema
- Glaucoma e.g, retained viscoelastic, pigment dispersion from excessive manipulation, vitreous in the angle etc.
- Uveitis
- Endophthalmitis
Late Complications:
- Pseudophakic Bullous Keratopathy
- Glaucoma
- Epithelial downgrowth
- Increased risk of retina detachment
References
- ↑ Chakrabarti, A., & Nazm, N. (2017). Posterior capsular rent: Prevention and management. Indian journal of ophthalmology, 65(12), 1359.
- ↑ Jump up to: 2.0 2.1 2.2 Vajpayee RB, Sharma N, Dada T, Gupta V, Kumar A, Dada VK, et al. Management of posterior capsule tears. Surv Ophthalmol. 2001;45:473–88.
- ↑ Greenberg PB, Tseng VL, Wu WC, Liu J, Jiang L, Chen CK, et al. Prevalence and predictors of ocular complications associated with cataract surgery in United States veterans. Ophthalmology. 2011;118:507–14.
- ↑ Jump up to: 4.0 4.1 Narendran N, Jaycock P, Johnston RL, et al. The Cataract National Dataset electronic multicentre audit of 55,567 operations: risk stratification of posterior capsule rupture and vitreous loss. Eye (Lond). 2009;23:31–7. (Pubmed:18327164).
- ↑ Berler DK. Intraoperative complications during cataract surgery in the very old. Trans Am Ophthalmol Soc. 2000;98:127–30; discussion 130–132.
- ↑ Brazitikos PD, Tsinopoulos IT, Papadopoulos NT, Fotiadis K, Stangos NT. Ultrasonographic classification and phacoemulsification of white senile cataracts. Ophthalmology. 1999;106:2178–83.
- ↑ Crouch Jr ER, Parks MM. Management of posterior lenticonus complicated by unilateral cataract. Am J Ophthalmol. 1978;85:503–8
- ↑ Hashemi H, Mohammadpour M, Jabbarvand M, et al. Incidence of and risk factors for vitreous loss in resident-performed phacoemulsification surgery. J Cataract Refract Surg. 2013;39:1377–82.
- ↑ Benjamin L. Cataract surgery in eyes with shallow anterior chamber. In: Chakrabarti A, editor. Cataract surgery in diseased eyes. JAYPEE; 2014. p. 57–60.
- ↑ Chakrabarti A. Phacoemulsification in vitectomozed eyes. In: Chakrabarti A, editor. Cataract surgery in diseased eyes. JAYPEE; 2014. p. 122–31.
- ↑ Zaidi FH, Corbett MC, Burton BJL, Bloom PA. Raising the benchmark for the 21st century – the 1000 cataract operations audit and survey: outcomes, consultant-supervised training and sourcing NHS choice. Br J Ophthalmol. 2007;91:731–6.
- ↑ Najjar DM, Awwad ST. Cataract surgery risk score for residents and beginning surgeons. J Cataract Refract Surg. 2003;29:2035–6.
- ↑ Randleman JB, Wolfe JD, Woodward M, Lynn MJ, Cherwek DH, Srivastava SK. The resident surgeon phacoemulsification learning curve. Arch Ophthalmol. 2007;125:1215–9.
- ↑ Rutar T, Porco TC, Naseri A. Risk factors for intraoperative complications in resident-performed phacoemulsification surgery. Ophthalmology. 2009;116:431–6.
- ↑ Gray H. The organs of the senses and the common integument. Peripheral organs of the special senses. The organ of sight. The refracting media. In: Lewis WH, editor. Anatomy of the human body. 20th ed. Philadelphia: Lea & Febiger; 1918.
- ↑ Barraquer RI, Michael R, Abreu R, Lamarca J, Tresserra F. Human lens capsule thickness as a function of age and location along the sagittal lens perimeter. Invest Ophthalmol Vis Sci. 2006;47:2053–60.
- ↑ Beyer TL, Vogler G, Sharma D, et al. Protective barrier effect of the posterior lens capsule in exogenous bacterial endophthalmitis – An experimental primate study. Invest Ophthalmol Vis Sci. 1984;25:108–12.
- ↑ Brown JC, Blecher MH, Glasser DB. Lens and cataract. Basic and clinical science course. American academy of Ophthalmology, San Francisco; 2017–2018.
- ↑ Koretz JF, Handelman GH. How the human eye focuses. Sci Am. 1988;259:92–9.
- ↑ Tolentino FI, Schepens CL, Mackenzie Freeman H. Biomicroscopic appearance of the normal vitreous body. Vitreoretinal disorders: diagnosis and management. Philadelphia: W.B. Saunders; 1976.
- ↑ Jump up to: 21.0 21.1 Sebag J. Anatomy and pathology of the vitreo-retinal interface. Eye. 1992;6:541–52.
- ↑ Hogan MJ, Alvarado JA, Weddell JE. Histology of the human eye. Philadelphia: Saunders; 1971.
- ↑ Parkash, Rohit Om, et al. "Flap motility as a sign of posterior capsule rupture in peripherally extended anterior capsular tears." Clinical Ophthalmology (Auckland, NZ) 11 (2017): 1445.
- ↑ Chen YA, Hirnschall N, Findl O. Comparison of corneal wetting properties of viscous eye lubricant and balanced salt solution to maintain optical clarity during cataract surgery. J Cataract Refract Surg. 2011;37:1806–8.
- ↑ Bhattacharjee K, Bhattacharjee H, Goswami BJ, Sarma P. Capsulorhexis in intumescent cataract. J Cataract Refract Surg. 1999;25:1045–7.
- ↑ Jacob S, Agarwal A, Agarwal A, Agarwal S, Chowdhary S, Chowdhary R, Bagmar AA. Trypan blue as an adjunct for safe phacoemulsification in eyes with white cataract. J Cataract Refract Surg. 2002;28:1819–25.
- ↑ Kara-Junior N, de Santhiago MR, Kawakami A, Carricondo P, Hida WT. Mini-rhexis for white intumescent cataracts. Clinics. 2009;64:309–12.
- ↑ Vajpayee RB, Bansal A, Sharma N, Dada T, Dada VK. Phacoemulsification of white hypermature cataract. J Cataract Refract Surg. 1999;25:1157–60.
- ↑ Conrad-Hengerer I, Hengerer FH, Joachim SC, Schultz T, Dick HB. Femtosecond laser-assisted cataract surgery in intumescent white cataracts. J Cataract Refract Surg. 2014;40:44–50.
- ↑ Koch DD, Liu JF. Zonular encroachment on the anterior capsular zonular-free zone. Am J Ophthalmol. 1988;106:491–2.
- ↑ Vajpayee RB, Sinha R, Singhvi A, Sharma N, Titiyal JS, Tandon R. ‘Layer by layer’ phacoemulsification in posterior polar cataract with pre-existing posterior capsular rent. Eye (Lond). 2008;22(8):1008–10.
- ↑ Osher RH. Slow motion phacoemulsification approach. J Cataract Refract Surg. 1993;19:667 [letter].
- ↑ Yeoh R. The ‘pupil snap’ sign of posterior capsule rupture with hydrodissection in phacoemulsification. Br J Ophthalmol. 1996;80(5):486.
- ↑ Reddy MK. Complications of cataract surgery. Indian J Ophthalmol. 1995;43:201–9.
- ↑ Jacobs PM. Vitreous loss during cataract surgery: prevention and optimal management. Eye (Lond). 2008;22:1286–9.
- ↑ Blomquist PH, Rugwani RM. Visual outcomes after vitreous loss during cataract surgery performed by residents. J Cataract Refract Surg. 2002;28(5):847–52.
- ↑ Chakrabarti, A. Posterior Capsular Rent.
- ↑ Jump up to: 38.0 38.1 38.2 Couch SM, Bakri SJ. Use of triamcinolone during vitrectomy surgery to visualize membranes and vitreous. Clin Ophthalmol. 2008;2(4):891–6.
- ↑ Gimbel H, Neuhann T. Developments, advantages, and methods of the continuous circular capsulorhexis technique. J Cataract Refract Surg. 1990;16:31–7.
- ↑ Gimbel HV. Posterior capsule tears using phacoemulsification-causes, prevention and management. Eur J Implant Refract Surg. 1990;2:63–9.
- ↑ Dada T, Sharma N, Vajpayee RB, Dada VK. Conversion from phacoemulsification to extracapsular cataract extraction: incidence, risk factors, and visual outcome. J Cataract Refract Surg. 1998; 24(11):1521–4.
- ↑ Steel D. Refractive changes associated with vitreoretinal surgery. In: Lois N, Wong D, editors. Complications of vitreo-retinal surgery. Philadelphia: Lippincott Williams & Wilkins; 2013. p. 379–86.
- ↑ García-Sáenz MC, Arias-Puente A, Rodríguez-Caravaca G, Bañuelos JB. Effectiveness of intracameral cefuroxime in preventing endophthalmitis after cataract surgery. Ten-year comparative study. J Cataract Refract Surg. 2010;36(2):203–7.
- ↑ Cao, He, et al. "Risk factors for acute endophthalmitis following cataract surgery: a systematic review and meta-analysis." PloS one 8.8 (2013): e71731.
- ↑ Jaffe NS, Jaffe MS, Jaffe GF. Cataract surgery and its complications. 6th ed. St. Louis: Mosby; 1997.