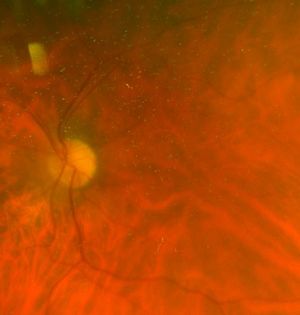
Foveal Hypoplasia
All content on Eyewiki is protected by copyright law and the Terms of Service. This content may not be reproduced, copied, or put into any artificial intelligence program, including large language and generative AI models, without permission from the Academy.
Disease entity
ICD 9:
Fundus coloboma 743.52
Other congenital anomalies of posterior segment 743.59
ICD 10:
Other congenital anomalies of posterior segment of eye Q14.8
Disease
Foveal hypoplasia is an ocular abnormality in which the foveal pit either fails to develop or does not completely develop. It is associated with poor visual acuity and nystagmus. [1] It may present in isolation or be associated with other conditions such as albinism, coloboma, optic nerve hypoplasia, retinopathy of prematurity, and aniridia. [2][3] Risk factors that affect foveal development, such as prematurity, can lead to foveal hypoplasia.[2]
Although the foveal pit does not fully develop in foveal hypoplasia, there is developmental plasticity in the retina that allows cone cells in the foveal area to change their own shape to allow for a higher cone cell density in the fovea.[4] This, in the most basic sense, allows the central area of the eye and brain to increase the sampling rate of each photon entering the retina.[4]
History
Early descriptions of foveal hypoplasia in the early 1900s were in association with other diseases, particularly hereditary nystagmus, while assessing for retinal abnormalities with ophthalmoscopy.[5][6] Other conditions were later discovered to be associated with foveal hypoplasia, including macular aplasia, macular coloboma, and albinism.[5][7][8] Later use of imaging techniques such as fundus fluorescein angiography to identify absent foveal avascular zones (FAZ) have found cases of isolated foveal hypoplasia without associated nystagmus or comorbidities.[9]
Epidemiology
A cross-sectional study by Noval et al. with 286 healthy children found that up to 3% of children had an anatomically underdeveloped foveal pit (fovea plana) bilaterally on OCT (optical coherence tomography).[10] Because foveal hypoplasia is commonly associated with several ocular diseases, there does not seem to be a widely accepted standalone statistic on its prevalence or incidence in isolation. However, many studies show the frequency of foveal hypoplasia occurrence in associated diseases.[2]
Risk Factors and Associations
Numerous risk factors are associated with foveal hypoplasia, including developmental and genetic factors. It is hypothesized that fetal endothelial cell and macular pigmentary development are key to the proper development of the foveal pit.[1][4][11][12][13][14]
- Prematurity is a significant risk factor due to the associated abnormal development of retinal vasculature and deficiency of macular pigmentation in these patients.[2][11] Prior studies have shown a small FAZ in patients with a history of prematurity.
- Foveal hypoplasia is also associated with
Genetics
A multicenter observational study of 907 patients found that the most common genetic etiology for typical foveal hypoplasia was albinism (67.5%), followed by PAX6 (21.8%), SLC38A8 (6.8%), and FRMD7 (3.5%) gene variants.[18] Multiple gene mutations have been linked to ocular albinism, with and without cutaneous involvement.
PAX6 variants can cause autosomal dominant foveal hypoplasia and affect embryonic eye development, resulting in a range of phenotypes, the most common being aniridia.[19]
SLC38A8 variants are responsible for an autosomal recessive form of foveal hypoplasia.[20] SLC38A8 codes for a glutamine transporter mainly expressed in the photoreceptor layer of the retina. Missense mutations of SLC38A8 can cause defects in the sixth transmembrane domain of photoreceptors, resulting in the underdevelopment of the retina.[21]
FRMD7 variants are often associated with idiopathic infantile nystagmus and have an X-linked inheritance pattern. The role of FRMD7 is not well understood, but knockdown models in mice suggest that it regulates neuronal outgrowth and development.[22][23]
Other genes cause ocular diseases associated with foveal hypoplasia as a symptom of the main disease. For example, several genes cause achromatopsia, which is partial or total agenesis of cone cells in the retina and associated with foveal hypoplasia.[24][25] Melanin synthesis genes, retinal vascularization genes, and procollagen genes, which cause albinism, familial exudative vitreoretinopathy, and stickler syndrome, respectively, all also have a high association with having foveal hypoplasia as an ocular symptom of their disease.[18][26][27][28][29][30]
General Pathology
There are hypotheses on why the fovea fails to develop in these diseases, although the mechanism is not fully known. One theory is that in fetal development, the foveal avascular zone does not develop, and instead, astrocytes induce retinal endothelial cell migration across the fovea, which inhibits the foveal pit from forming.[2][6][12] The concentration of vessels is thought to cause the retinal tissue to have a fragile and stiff quality, reducing malleability for the formation of the foveal pit.[12] However, in disease conditions such as congenital achromatopsia, the foveal avascular zone is present while the patient still presents with foveal hypoplasia, suggesting that the foveal avascular zone is necessary for normal foveal development, but not sufficient to prevent foveal hypoplasia altogether.[13]
Other hypotheses for the development of foveal hypoplasia suggest a cellular disruption in the mechanical development of retinal neurons forming the foveal pit. The ganglion cell layer and inner nuclear layer are displaced centrifugally, while cone cells in the outer nuclear layer are migrated centrally to the fovea during foveal development.[12][31] During this process, Muller cells aid in vertical traction of the Henle fibers, causing vertical stability of the foveal pit, and astrocytes retract laterally, causing widening of the pit.[5] This suggests cellular disruption to this mechanical process in specific diseases may lead to foveal hypoplasia.
Diagnosis
Signs and Symptoms
Foveal hypoplasia is commonly seen with conditions such as albinism, nystagmus, achromatopsia, iris abnormalities like aniridia, cataracts, and other ocular abnormalities.[32] Histologic analysis and OCT studies have revealed that the structure of the macula resembles peripheral retinal tissue with the persistence of ganglion and nuclear layers.[1][31] Ophthalmoscopy shows the absence of the foveal pit and FAZ.[2][4][9] Most patients with foveal hypoplasia have poorer than normal visual acuity ranging between 20/50 to 20/200.[9][32]
Diagnostic Procedures/ Testing
Historically, the diagnosis of foveal hypoplasia was made in nystagmus evaluation with the ophthalmoscopic examination of the fundus. Findings commonly included the absence of foveal pigmentation and the foveal reflex.[6][9] Introduction of fluorescein angiography allowed for visualization of the FAZ, and a small or absent FAZ coupled with ophthalmoscopy support the diagnosis of foveal hypoplasia.[9] Non-invasive imaging such as OCT and OCT-A (optical coherence tomography angiography) are now commonly used to evaluate the morphology and vasculature of the retina, respectively. In foveal hypoplasia, OCT evaluation reveals the absence of the foveal pit and the persistence of inner retinal layers through the area where the foveal center is expected.[2][32] OCT-A evaluation of the FAZ shows an absence at the superficial capillary plexus and a variable decrease in the deep capillary plexus.[33][34]
A summary of diagnostic test findings is described below:
| Diagnostic test | Findings |
|---|---|
| Ophthalmoscopy | Absent foveal pigmentation and foveal reflex |
| Fluorescein angiography | Absence or decreased foveal avascular zone |
| Optical coherence tomography | Absence of foveal pit
Persistence of inner retinal layers through the expected area of the fovea |
| Optical coherence tomography angiography | Absence of FAZ at the superficial capillary plexus
Absence or decrease of FAZ at the deep capillary plexus |
Grading
OCT provides high-resolution images of the retinal layers and has aided in the morphologic characterization of foveal hypoplasia. In 2011, Thomas MG et al. developed an OCT-based grading system for foveal hypoplasia based on unique foveal specialization features visible on OCT ( see table ).[35] This Leicester Grading system has been validated in several studies[18] and has recently also been shown to predict future vision in children with infantile nystagmus.[36]
| Grade | Morphologic features | Associated BCVA (LogMAR) |
|---|---|---|
| Normal |
|
0.26 |
| 1a |
|
0.41 |
| 1b |
|
0.65 |
| 2 |
|
0.60 |
| 3 |
|
0.74 |
| 4 |
|
1.01 |
| Atypical |
|
0.93 |
Management
The management includes management of the associated ocular and systemic disorders, refractive correction, management of amblyopia, and low vision aids.[37] Patients with significant cataracts may benefit from cataract surgery though visual outcome is limited by the severity of foveal hyoplasia. Genetic counseling and analysis for genetic mutations form an important part of management in cases.
References
- ↑ Jump up to: 1.0 1.1 1.2 Duke-Elder, S. System of Ophthalmology 1963 3 St Louis CV Mosby.
- ↑ Jump up to: 2.0 2.1 2.2 2.3 2.4 2.5 2.6 Kondo, H. Foveal hypoplasia and optical coherence tomographic imaging. Taiwan J Ophthalmol. 2018 Oct-Dec;8(4):181-188. doi: 10.4103/tjo.tjo_101_18. PMID: 30637189; PMCID: PMC6302563.
- ↑ Bringmann, A., Syrbe, S., Görner, K., Kacza, J., Francke, M., Wiedemann, P., Reichenbach, A. The primate fovea: Structure, function and development, Progress in Retinal and Eye Research, Volume 66, 2018, Pages 49-84, ISSN 1350-9462, https://doi.org/10.1016/j.preteyeres.2018.03.006.
- ↑ Jump up to: 4.0 4.1 4.2 4.3 Provis, J.M., Dubis, A.M., Maddess, T., Carroll, J. Adaptation of the central retina for high acuity vision: cones, the fovea, and the avascular zone. Prog Retin Eye Res. 2013 Jul;35:63-81. doi: 10.1016/j.preteyeres.2013.01.005. Epub 2013 Mar 15. PMID: 23500068; PMCID: PMC3658155.
- ↑ Jump up to: 5.0 5.1 5.2 Koyanagi, Y. Über den genetischen Zusammenhang zwischen dem hereditären Nystagmus und Bulbus Albinismus. Klin Monatsbl Augenheilkd 1927; 79: 43- 8.
- ↑ Jump up to: 6.0 6.1 6.2 Holm, E. Hereditary nystagmus. Acta Ophthalmol (Kbh) 1926; 4: 20-7.
- ↑ Uemura Y, Kumanomido A. [The study on macular findings in "amblyopic eye". Report I. Macular coloboma, macular aplasia, and macular hypoplasia]. Nippon Ganka Gakkai Zasshi. 1961 Nov 10;65:2264-71. Japanese. PMID: 13923531.
- ↑ Curran, R.E., & Robb, R.M. Isolated foveal hypoplasia. 1976; Archives of ophthalmology, 94 1, 48-50 .
- ↑ Jump up to: 9.0 9.1 9.2 9.3 9.4 Oliver, M.D., Dotan, S.A., Chemke, J., Abraham, F.A. Isolated foveal hypoplasia. Br J Ophthalmol. 1987 Dec;71(12):926-30. doi: 10.1136/bjo.71.12.926. PMID: 3427001; PMCID: PMC1041346.
- ↑ Noval S., Freedman S.F., Asrani S., El-Dairi M.A. Incidence of fovea plana in normal children. J AAPOS. 2014 Oct;18(5):471-5. doi: 10.1016/j.jaapos.2014.07.157. Epub 2014 Sep 27. PMID: 25266830.
- ↑ Jump up to: 11.0 11.1 Bernstein, P.S., Sharifzadeh, M., Liu, A., Ermakov, I., Nelson, K., Sheng, X., Panish, C., Carlstrom, B., Hoffman, R.O., Gellermann, W. Blue-light reflectance imaging of macular pigment in infants and children. Invest Ophthalmol Vis Sci. 2013 Jun 10;54(6):4034-40. doi: 10.1167/iovs.13-11891. PMID: 23652486; PMCID: PMC3680006.
- ↑ Jump up to: 12.0 12.1 12.2 12.3 Springer, A., & Hendrickson, A. Development of the primate area of high acuity. 1. Use of finite element analysis models to identify mechanical variables affecting pit formation. 2004. Visual Neuroscience, 21(1), 53-62. doi:10.1017/S0952523804041057
- ↑ Jump up to: 13.0 13.1 Rachel E. Linderman, Michalis Georgiou, Erica N. Woertz, Jenna A. Cava, Katie M. Litts, Sergey Tarima, Ranjan Rajendram, Jan M. Provis, Michel Michaelides, Joseph Carroll; Preservation of the Foveal Avascular Zone in Achromatopsia Despite the Absence of a Fully Formed Pit. Invest. Ophthalmol. Vis. Sci. 2020;61(10):52. doi: https://doi.org/10.1167/iovs.61.10.52.
- ↑ Mintz-Hittner, H.A., Knight-Nanan, D.M., Satriano, D.R., Kretzer, F.L. A small foveal avascular zone may be a historic mark of prematurity. Ophthalmology. 1999 Jul;106(7):1409-13. doi: 10.1016/S0161-6420(99)00732-0. PMID: 10406630.
- ↑ Creel, D.J., Summers, C.G., King, R.A. Visual anomalies associated with albinism. Ophthalmic Paediatr Genet. 1990;11:193–200.
- ↑ Kohli P, Mishra C, Baliga G, Rajan RP. Multimodal imaging of congenital retinal macrovessel with secondary foveal hypoplasia. BMJ Case Rep. 2022;15(4):e249563. Published 2022 Apr 20. doi:10.1136/bcr-2022-249563
- ↑ Tripathy K, Bypareddy R, Chawla R. Congenital retinal macrovessel may be associated with unilateral foveal hypoplasia/small foveal avascular zone. Can J Ophthalmol. 2019;54(1):139. doi:10.1016/j.jcjo.2018.06.018
- ↑ Jump up to: 18.0 18.1 18.2 Kuht, H.J., Maconachie, G.D.E., Han, J., Kessel, L., van Genderen, M.M., McLean, R.J., Hisaund, M., Tu, Z., Hertle, R.W., Gronskov, K., Bai, D., Wei, A., Li, W., Jiao, Y., Smirnov, V., Choi, J.H., Tobin, M.D., Sheth, V., Purohit, R., Dawar, B., Girach, A., Strul, S., May, L., Chen, F.K., Heath Jeffery, R.C., Aamir, A., Sano, R., Jin, J., Brooks, B.P., Kohl, S., Arveiler, B., Montoliu, L., Engle, E.C., Proudlock, F.A., Nishad, G., Pani, P., Varma, G., Gottlob, I., Thomas, M.G. Genotypic and Phenotypic Spectrum of Foveal Hypoplasia: A Multicenter Study. Ophthalmology. 2022 Jun;129(6):708-718. doi: 10.1016/j.ophtha.2022.02.010. Epub 2022 Feb 11. PMID: 35157951; PMCID: PMC9341240.
- ↑ Ehrenberg, M., Bagdonite-Bejarano, L., Fulton, A.B., Orenstein, N., Yahalom, C. Genetic causes of nystagmus, foveal hypoplasia, and subnormal visual acuity- other than albinism. Ophthalmic Genet. 2021 Jun;42(3):243-251. doi: 10.1080/13816810.2021.1888128. Epub 2021 Feb 17. PMID: 33594928.
- ↑ Poulter JA, Al-Araimi M, Conte I, et al. Recessive mutations in SLC38A8 cause foveal hypoplasia and optic nerve misrouting without albinism. Am J Hum Genet. 2013;93(6):1143-1150. doi:10.1016/j.ajhg.2013.11.002
- ↑ Kuht, H.J., Han, J., Maconachie, G.D.E., Park, S.E., Lee, S.T., McLean, R., Sheth, V., Hisaund, M., Dawar, B., Sylvius, N., Mahmood, U., Proudlock, F.A., Gottlob, I., Lim, H.T., Thomas, M.G. SLC38A8 mutations result in arrested retinal development with loss of cone photoreceptor specialization. Hum Mol Genet. 2020 Nov 4;29(18):2989-3002. doi: 10.1093/hmg/ddaa166. PMID: 32744312; PMCID: PMC7645707.
- ↑ Thomas, M.G., Crosier, M., Lindsay, S., Kumar, A., Araki, M., Leroy, B.P., McLean, R.J., Sheth, V., Maconachie, G., Thomas, S., Moore, A.T., Gottlob, I. Abnormal retinal development associated with FRMD7 mutations. Hum Mol Genet. 2014;23:4086–93.
- ↑ Betts-Henderson J, Bartesaghi S, Crosier M, Lindsay S, Chen HL, Salomoni P, Gottlob I, Nicotera P. The nystagmus-associated FRMD7 gene regulates neuronal outgrowth and development. Hum Mol Genet. 2010 Jan 15;19(2):342-51. doi: 10.1093/hmg/ddp500. Epub 2009 Nov 5. PMID: 19892780.
- ↑ Thiadens AA, Somervuo V, van den Born LI, Roosing S, van Schooneveld MJ, Kuijpers RW, et al. Progressive loss of cones in achromatopsia: An imaging study using spectral-domain optical coherence tomography. Invest Ophthalmol Vis Sci. 2010;51:5952–7.
- ↑ Sundaram V, Wilde C, Aboshiha J, Cowing J, Han C, Langlo CS, et al. Retinal structure and function in achromatopsia: Implications for gene therapy. Ophthalmology. 2014;121:234–45.
- ↑ Thomas MG, Kumar A, Kohl S, Proudlock FA, Gottlob I. High-resolution in vivo imaging in achromatopsia. Ophthalmology. 2011;118:882–7.
- ↑ 24. Kondo H. Complex genetics of familial exudative vitreoretinopathy and related pediatric retinal detachments. Taiwan J Ophthalmol. 2015;5:56–62.
- ↑ 25. Yonekawa Y, Thomas BJ, Drenser KA, Trese MT, Capone A., Jr Familial exudative vitreoretinopathy: Spectral-domain optical coherence tomography of the Vitreoretinal interface, retina, and choroid. Ophthalmology. 2015;122:2270–7.
- ↑ 26. Richards AJ, Baguley DM, Yates JR, Lane C, Nicol M, Harper PS, et al. Variation in the vitreous phenotype of stickler syndrome can be caused by different amino acid substitutions in the X position of the type II collagen gly-X-Y triple helix. Am J Hum Genet. 2000;67:1083–94.
- ↑ 27. Matsushita I, Nagata T, Hayashi T, Kimoto K, Kubota T, Ohji M, et al. Foveal hypoplasia in patients with stickler syndrome. Ophthalmology. 2017;124:896–902.
- ↑ Jump up to: 31.0 31.1 Hendrickson AE. Primate foveal development: a microcosm of current questions in neurobiology. Invest Ophthalmol Vis Sci. 1994 Jul;35(8):3129-33. PMID: 8045707.
- ↑ Jump up to: 32.0 32.1 32.2 Recchia FM, Carvalho-Recchia CA, Trese MT. Optical Coherence Tomography in the Diagnosis of Foveal Hypoplasia. Arch Ophthalmol. 2002;120(11):1587–1588.
- ↑ Sánchez-Vicente, José L. MD; Contreras-Díaz, Miguel MD; Llerena-Manzorro, Laura MD; Rueda, Trinidad PhD; López-Herrero, Fernando MD; Molina-Socola, Fredy E. MD; Muñoz-Morales, Ana PhD; Rodríguez de la Rúa-Franch, Enrique PhD. FOVEAL HYPOPLASIA: DIAGNOSIS USING OPTICAL COHERENCE TOMOGRAPHY ANGIOGRAPHY. Retinal Cases & Brief Reports: Spring 2018 - Volume 12 - Issue 2 - p 122-126 doi: 10.1097/ICB.0000000000000440
- ↑ Pakzad-Vaezi K, Keane PA, Cardoso JN, Egan C, Tufail A. Optical coherence tomography angiography of foveal hypoplasia. Br J Ophthalmol. 2017 Jul;101(7):985-988. doi: 10.1136/bjophthalmol-2016-309200. Epub 2016 Nov 29. PMID: 27899366.
- ↑ Thomas MG, Kumar A, Mohammad S, Proudlock FA, Engle EC, Andrews C, Chan WM, Thomas S, Gottlob I. Structural grading of foveal hypoplasia using spectral-domain optical coherence tomography a predictor of visual acuity? Ophthalmology. 2011 Aug;118(8):1653-60. doi: 10.1016/j.ophtha.2011.01.028. Epub 2011 Apr 29. Erratum in: Ophthalmology. 2011 Oct;118(10):1910. PMID: 21529956; PMCID: PMC5648335.
- ↑ Rufai SR, Thomas MG, Purohit R, Bunce C, Lee H, Proudlock FA, Gottlob I. Can Structural Grading of Foveal Hypoplasia Predict Future Vision in Infantile Nystagmus?: A Longitudinal Study. Ophthalmology. 2020 Apr;127(4):492-500. doi: 10.1016/j.ophtha.2019.10.037. Epub 2019 Nov 4. PMID: 31937464; PMCID: PMC7105819.
- ↑ Foveal Hypoplasia 1. Hereditary Ocular Disease Database (https://disorders.eyes.arizona.edu).