West Nile Retinopathy
All content on Eyewiki is protected by copyright law and the Terms of Service. This content may not be reproduced, copied, or put into any artificial intelligence program, including large language and generative AI models, without permission from the Academy.
West Nile virus retinopathy is a subtype of multifocal chorioretinitis caused by the West Nile virus.
Disease Entity
West Nile virus retinopathy
Disease
West Nile virus retinopathy is a subtype of multifocal chorioretinitis caused by the West Nile virus. The virus was first isolated from the West Nile district of Uganda in 1937. In USA, the first appearance was in 1999 at New York.
Etiology
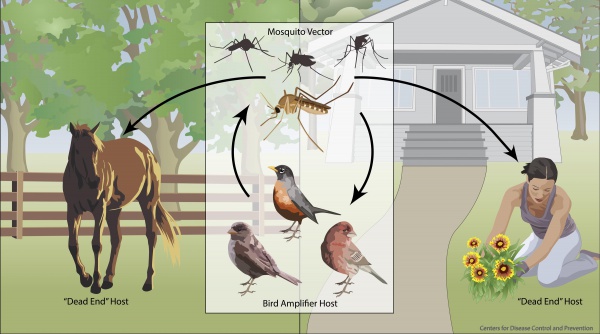
The West Nile virus (WNV) is a single-stranded RNA flavivirus of the Japanese encephalitis serogroup. It is a zoonotic disease most commonly transmitted by the bite of the Culex genus of mosquitoes (especially Culex pipiens). WNV reservoirs are wild birds though horses and humans act as “dead end” hosts (Figure 1).
Alternate routes of human infection have been documented in the case of blood transfusions, organ transplants, exposure in a laboratory setting, and vertically during pregnancy. However, the vast majority of cases of WNV infection have been from infected mosquitoes.
Risk Factors
There are several risk factors associated with WNV infection. Chief among them is living in an area in which the virus is prevalent. Originally, the virus was limited to sites on major bird migratory pathways including Africa, some of Europe, the Middle East, West Asia, and Australia. Since 1999, when the virus was introduced into the United States, WNV has spread from Canada to Venezuela.
In the United States, a higher concentration of WNV infections has been present in the western states (e.g. Montana, Colorado, Wyoming). However, new information indicates that infection is increasing, with Centers for Disease Control data indicating 5674 cases of infection and 286 deaths attributable to the virus in 2012.
Additional risk factors include the following: age > 50; certain medical conditions (cancer, diabetes, hypertension, kidney disease, and solid-organ transplants); lack of proper mosquito protection; and exposure to mosquitoes during the summer months (the time of year during which outbreaks are most common). Recent research suggests that a mutation of the chemokine receptor type 5 (CCR5) gene, which has a protective effect against HIV predisposes a homozygote to developing neurologic manifestations.
General Pathology
After becoming infected with WNV, there is usually an incubation period that can last from two to fifteen days. Though 80% of WNV infections in humans are subclinical, the remaining 20% typically manifest as a febrile illness. Constitutional symptoms – fever, headaches, fatigue, myalgias, malaise, nausea, emesis, anorexia, and rash – are common.
In approximately 1% of all WNV infections, West Nile neuroinvasive disease develops. This manifestation is associated with meningoencephalitis, a poliomyelitis-like syndrome, muscular weakness, and acute flaccid paralysis. There are rare non-neurologic complications of WNV infection that tend to affect those with suppressed immune function more often. These complications include fulminant hepatitis, pancreatitis, optic neuritis, viral hemorrhagic fever, and chorioretinitis.
Of the ocular manifestations of WNV infection, asymptomatic multifocal chorioretinits is the most common (80%). Other ocular complications of WNV infection have been described in the literature: anterior uveitis, retinal vasculitis, optic neuritis, subconjunctival hemorrhage, sixth cranial nerve palsy, nystagmus, and congenital chorioretinal scarring.
The ocular involvement may be hematogenous (involvement of choroid) or neural via transmission from the central nervous system to the retinal nerve fiber layer (linear scars appear along the districution of the nerve fiber layer).
Pathophysiology
Although the exact molecular pathophysiology of WNV infection remains an area of active study, recent research supports the hypothesis that WNV likely gains access to cells through interaction with toll-like receptor 3 (Tlr3) and CCR5. It is likely that WNV infection causes a Tlr3-induced inflammatory response, weakening the blood brain barrier. Thus, the virus is able to penetrate into the brain and cause its more devastating pathologies. As a member of the Flaviviridae family, WNV is a single-stranded RNA virus that causes host cells to produce progeny virions using host proteins.
Diagnosis
In 80% of cases, a patient will have an asymptomatic course of ocular WNV infection. It is more likely that chorioretinal findings will be observed after the patient has already been diagnosed with WNV infection.
Diagnosis is typically made through history, physical examination, and serology.
History
Classically, the patient has been previously diagnosed with a past WNV infection and has no reported ocular symptoms.
Physical examination
Typical symptoms of WNV infection are acute headache, weakness, myalgia, arthralgia, gastrointestinal symptoms, and maculopapular rash. If neurological involvement is present, symptoms and signs of meningitis and encephalitis may be present.
Signs
Chorioretinal WNV is typically found to exhibit linearly arranged or scattered chorioretinal lesions. On slit lamp examination, active lesions appear circular, deep, and creamy. Inactive lesions are atrophic and partially pigmented with a targetoid appearance. Representative images are available at
https://www.medscape.com/viewarticle/575934_10, https://www.retinalphysician.com/issues/2007/may-2007/uveitis-diagnosis,-management,-and-treatment, and http://retinagallery.com/displayimage.php?album=search&cat=0&pid=8669#top_display_media
Symptoms
In the case of active infection or lesions along the macula, the patient may report decreased visual acuity, floaters, conjunctival injection, visual field defects, and diplopia.
Persistent visual loss may be due to foveal chorioretinal scar, choroidal neovascularization, vitreous hemorrhage, tractional retinal detachment, ischemic maculopathy, optic atrophy, and retrogeniculate damage.
Clinical diagnosis
A high degree of suspicion is required to make the diagnosis of WNV retinopathy in patients who have not been previously diagnosed with WNV infection. Suspect those who have had recent exposure to mosquitoes, blood transfusion, organ transplantation, and travel through endemic areas.
Diagnostic procedures
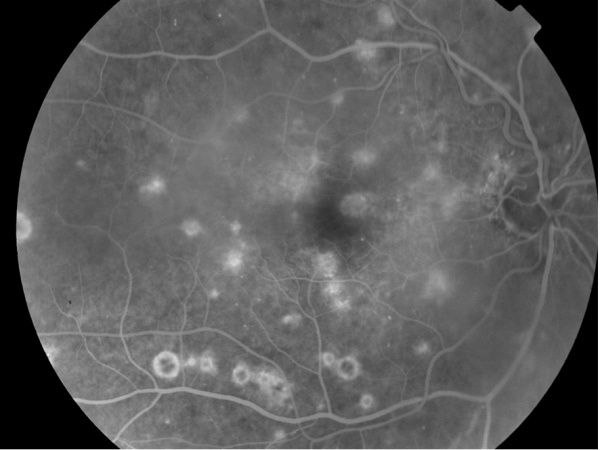
Fluorescein angiography may demonstrate early hypofluorescence and late staining of active lesions and central hypofluorescence and peripheral hyperfluorescence of inactive lesions (Figure 2).
Indocyanine green angiography typically demonstrates more lesions than can be appreciated by fluorescein angiography.
Autofluorescence may highlight the old scars better than fundus examination of fundus photograph.
Laboratory test
Serologic testing of blood or cerebrospinal fluid for WNV IgM and neutralizing antibodies often successfully identifies active WNV infection. Specimens may not be positive by IgM prior to eight days of infection. A positive IgG test in the absence of IgM simply indicates that a flavivirus infection has occurred, not that an active infection is present.
Other diagnostic tests include
- plaque reduction neutralization test (PRNT),
- immunoflourescence (IFA)
- nucleic acid amplification test (NAT)
For screening blood before transfusion these tests are available
- Procleix® WNV Assay
- Procleix® TIGRIS®
- cobas TaqScreen WNV test for screening donated tissues including blood
Differential diagnosis
There are a wide variety of other infections that present similarly to WNV infection, including those that can cause meningitis and/or encephalitis. The following are common causes of illnesses that present in similar ways: HSV-1 encephalitis, enterovirus, Legionnaire’s disease, Rocky Mountain spotted fever, EBV, HHV-6, Japanese encephalitis, encephalopathy due to systemic illnesses (e.g. subacute bacterial endocarditis, systemic lupus erythematosus), and drug-induced aseptic meningitis.
Management
General treatment
There is currently no proven treatment for WNV infection. For systemic infection, therapy consists in supportive treatment. In severe cases, hospitalization for the administration of intravenous fluids and respiratory support may be necessary.
Should choroidal neovascularization occur, anti-VEGF therapy may be warranted.
Complications
Through rather uncommon, complication from intraocular WNV infection may occur in the form of chorioretinal scars, choroidal neovascularization, vitreous hemorrhage, tractional retinal detachment, ischemic maculopathy, optic atrophy, and retrogeniculate damage.
Prevention
Primary prevention of infection is the mainstay of WNV infection control. Wearing protective clothing, using mosquito repellent, and using mosquito larvicides are proven strategies for decreasing the incidence of infection. A vaccine for WNV is currently under development but has not gone through clinical trials.
Prognosis
Prognosis for WNV retinopathy is very good. In the vast majority of cases, the ocular disease is self-limited and visual acuity returns to baseline.
Additional Resources
- Centers for Disease Control and Prevention. Centers for Disease Control and Prevention, 09 July 2013. Web. 10 July 2013. http://www.cdc.gov/westnile/index.html
- "West Nile Virus." WHO. N.p., n.d. Web. 10 July 2013. http://www.who.int/mediacentre/factsheets/fs354/en/
References
1. Bakri SJ, Kaiser PK. Ocular manifestations of west nile virus. Curr Opin Ophthalmol. 2004; 15:537-540.
2. Davis LE, DeBiasi R, Goade DE, et al. West nile virus neuroinvasive disease. Ann Neurol. 2006; 60:286-300.
3. Garg S, Jampol LM. Systemic and intraocular manifestations of west nile virus infection. Surv Ophthalmol. 2005; 50:3-13.
4. Glass WG, Lim JK, Cholera R, Pletnev AG, Gao JL, Murphy PM. Chemokine receptor CCR5 promotes leukocyte trafficking to the brain and survival in west nile virus infection. J Exp Med. 2005; 202:1087-1098.
5. Glass WG, McDermott DH, Lim JK, et al. CCR5 deficiency increases risk of symptomatic west nile virus infection. J Exp Med. 2006; 203:35-40.
6. Khairallah M, Ben Yahia S, Ladjimi A, et al. Chorioretinal involvement in patients with west nile virus infection. Ophthalmology. 2004; 111:2065-2070.
7. Khairallah M, Yahia SB, Letaief M, et al. A prospective evaluation of factors associated with chorioretinitis in patients with west nile virus infection. Ocul Immunol Inflamm. 2007; 15:435-439.
8. Wang T, Town T, Alexopoulou L, Anderson JF, Fikrig E, Flavell RA. Toll-like receptor 3 mediates west nile virus entry into the brain causing lethal encephalitis. Nat Med. 2004; 10:1366-1373.
9. Yahia SB, Khairallah M. Ocular manifestations of west nile virus infection. Int J Med Sci. 2009; 6:114-115.