Trisomy 13
All content on Eyewiki is protected by copyright law and the Terms of Service. This content may not be reproduced, copied, or put into any artificial intelligence program, including large language and generative AI models, without permission from the Academy.
Disease Entity
Trisomy 13, also known as Patau Syndrome, is a disorder of fetal maldevelopment with wide-ranging and often severe developmental manifestations, including ocular malformations. The disorder was first described by Patau, et al., in 1960 as "cerebral defects, apparent anophthalmia, cleft palate, hare lip, simian creases, trigger thumbs, polydactyly, and capillary hemangiomata."[1]
Etiology
Trisomy 13 is an aneuploidy (less or more than 46 chromosomes) in which three copies of chromosome 13—instead of the typical two—are present. Complete, mosaic, and partial forms of trisomy 13 exist.
A complete trisomy occurs when a chromosomal nondisjunction happens during meiosis, a process that occurs in sex cells before fertilization. In such cases, all cells in an organism fertilized by the aneuploid sex cell will harbor an aneuploid genome. Complete trisomy 13 is the most common form of the disorder, affecting approximately 80% patients.[2][3][4] A vast majority, approximately 91%, of trisomy 13 cases are due to nondisjunction in the female gamete.[5][6]
Translocation trisomy 13, “when part of chromosome 13 becomes attached (translocated) to another chromosome during the formation of eggs or sperm, or very early in fetal development” accounts for ~20% of cases. This results in two normal copies of chromosome 13 and an extra copy of chromosome 13 attached to another chromosome. Translocation trisomy 13 can be inherited.[3]
Mosaic trisomy 13 occurs when some cells contain euploid genomes and others contain aneuploid genomes. Mosaicism accounts for 5% of trisomy 13 cases.[7] The phenotypes of such cases are generally less severe. Mosaicism occurs due to nondisjunction during mitosis, the process of cell division during development. Only cells derived from the errant cell will harbor extra copies of the chromosome.[8]
Partial trisomy 13 occurs when cells contain two full copies plus an additional portion of chromosome 13. Phenotypes of partial trisomy 13 are also less severe than the complete form.[9]
Pathophysiology
Phenotypes that arise from aneuploidy are thought to occur due to abnormal levels of gene expression involving genes of the duplicated chromosome.[3][8] The following fetal developmental processes are thought to be disrupted during eye development in trisomy 13:[10]
- Organogenesis (third-fourth weeks): optic pit changes into the optic vesicle
- Potential abnormality in trisomy 13: anophthalmia
- End of fourth week: lens pit and vesicle appear, optic vesicle invaginates to form optic cup
- Potential abnormalities in trisomy 13: aphakia, congenital cystic eye, disturbed retinal lamination
- Sixth week: fetal fissures close; lens separates from surface and primary fibers form, retinal differentiation begins, tunica vasculosa begins
- Potential abnormalities in trisomy 13: coloboma, microphthalmia with orbital cysts, nuclear cataract
- Seventh-ninth weeks: secondary lens fibers form, secondary vitreous forms, neural crest cells grow into the anterior segment
- Potential abnormalities in trisomy 13: nuclear cataract, anomalies of the retina and/or vitreous, anterior segment dysgenesis
- Tenth-twelfth weeks: secondary lens fibers progress, development of ectodermal layers of iris and ciliary body begins
- Potential abnormalities in trisomy 13: zonular cataract, malformations of anterior segment
Epidemiology
The estimated prevalence of trisomy 13 is 1:5,300 in Europe[11] and 1:14,000 in the United States.[12] It is the third most common autosomal trisomy in newborns after trisomy 21 and trisomy 18[13][14] and is considered to be the most lethal of the three[15]. Only 6-12 % of infants with trisomy 13 survive beyond the first year of life. [4]It appears to affect females slightly more frequently than males.[3] The clearest risk factor for fetal aneuploidy is advanced maternal age, as the risk begins to increase significantly after a maternal age of 35 years for trisomies 13, 18, and 21.[14] Most cases are not inherited.[3]
Clinical Features
Systemic Manifestations
Complete trisomy 13 typically results in malformations of multiple organ systems and severe intellectual disability.[3] Common findings include: microcephaly, cleft lip and/or palate, polydactyly, hypotonia, failure to thrive, feeding difficulties, cutis aplasia (i.e. missing skin), capillary hemangiomas, low-set ears, deafness, cardiac, brain (holoprosencephaly), spinal cord, and genitourinary anomalies.[8][11][16] Trisomy 13 is the most common cause of holoprosencephaly, which causes severe abnormalities of midline facial structures.[17] Trisomy 13 has also been linked with paternal exposure to etoposide and cisplatin.[18] Many other systemic features of trisomy 13 have been described.[2][3]
Ocular Manifestations
The incidence of any particular ocular finding is difficult to ascertain due to the rarity of the disorder and the brief lifespan of affected individuals. These clinical signs have been reported in 80-99% of people with trisomy 13[2]:
These clinical signs have been reported in 30-79% of individuals with trisomy 13[2]:
- Abnormal eyelash morphology[2]
- Aplasia/hypoplasia of the iris (aniridia)[22]
- Congenital cataract 6% [11][16][20]
- Deeply set eye[2]
- Iris coloboma (usually inferonasal 33% [16] [20]Figure 2)
- Optic atrophy[2]
- Retinal dysplasia[23][24]
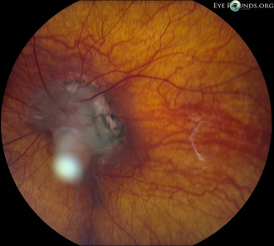
Other reported findings include:
- Acute elevation of intraocular pressure[7]
- Cartilaginous mass of the anterior segment[16][19]
- Ciliary body coloboma[24]
- Coats disease (retinal telangiectasias)[16]
- Congenital glaucoma/buphthalmos 1% [22][26][27][20]
- Corneal opacity[19]
- Cyclopia[17][23]
- Dysgenesis of the anterior segment[23][28]
- Epicanthal folds[29]
- Nasolacrimal duct obstruction[30]
- Optic nerve coloboma[31]
- Persistent fetal vasculature (PFV, Figure 3)[16][25]
- Persistent tunica vasculosa lentis (PTVL)[16]
- Sparse or absent eyebrows
- Upslanting palpebral fissures[21]
- Keratoconus[32]
Diagnosis
Trisomy 13 may be suggested or diagnosed with prenatal screening and testing using fetal ultrasound, biochemical and molecular testing of maternal blood, amniocentesis, and chorionic villus sampling.[2][33] It is associated with increased fetal nuchal translucency, decreased PAPP-A, and decreased β-hCG during first trimester screening.[34] Often, non-invasive prenatal testing is not sufficient for diagnosis, leading to more invasive amniocentesis and chorionic villus sampling. However, recent advancements in Artificial Intelligence methodologies of machine learning and deep learning may offer greater potential for non-invasive prenatal testing in the diagnosis of fetal aneuploidy.[35] Postnatal diagnostic genetic testing may be undertaken if features of the syndrome are suspected in an infant who was not diagnosed in the prenatal period.
Management
Given the limited life expectancy for most patients with trisomy 13, aggressive medical and surgical ophthalmic care has generally not been advised and palliative/supportive care options are preferred. However, for patients with better prognoses and eye conditions amenable to medical or surgical intervention, treatments may be considered.[16][30][36] In one study, among children who underwent surgery, 1-year survival was 68.6%.[37] More recent trends suggest more active medical and surgical management leading to increased survival rates and life expectancy in patients with trisomy 13.[20] More aggressive care downstream for other organ systems, including the eye, may also rise in the future. However, the American Association for Thoracic Surgery 2023 consensus statement advises individualized treatment plans, particularly for those with congenital heart disease, based on the severity of disease manifestations of the trisomic individual as a whole.[38]
Prognosis
Many cases of trisomy 13 end in spontaneous abortion (49% in one study[39]), and most of those born with the condition have a very limited life expectancy. Approximately 50% of affected infants do not survive beyond the first month, with a higher fatality rate for males.[2][40][41][4] According to one study, ten-year survival for trisomy 13 was 12.9%. In another study, among affected individuals who lived beyond one year, five-year survival was 84%.[41] The most common causes of death are cardiopulmonary arrest (69%), complications of congenital heart disease (13%), and pneumonia (4%).[40]
Summary
Trisomy 13, or Patau Syndrome, is a severe developmental disorder that results in anomalies in multiple organ systems and poor rate of survival. Ocular pathology is common and varies widely among individuals. Common ocular abnormalities include: anophthalmos/microphthalmos, congenital cataract, hypo- or hypertelorism, and inferonasal iris coloboma; but many other ocular findings have been reported.[11][16][19][21][23][22][31][30]
Additional Resources
- Chromosome Disorder Outreach (CDO): https://chromodisorder.org
- Hope For Trisomy: https://www.hopefortrisomy13and18.org
- Support Organization for Trisomy 18, 13, and Related Disorders (SOFT): https://trisomy.org
- Unique – Rare Chromosome Disorder Support Group: https://www.rarechromo.org
- National Institutes of Health: Genetic and Rare Diseases Information Center: https://rarediseases.info.nih.gov/diseases/7341/trisomy-13
- National Organization for Rare Disorders: https://rarediseases.org/rare-diseases/trisomy-13-syndrome/
References
- ↑ Patau K, Smith DW, Therman E, Inhorn SL, Wagner HP. Multiple congenital anomaly caused by an extra autosome. Lancet. 1960;1(7128):790-793. doi:10.1016/s0140-6736(60)90676-0
- ↑ Jump up to: 2.0 2.1 2.2 2.3 2.4 2.5 2.6 2.7 2.8 Trisomy 13. GARD (Genetic and Rare Diseases Information Center). Published April 25, 2016. Accessed December 8, 2020. https://rarediseases.info.nih.gov/diseases/7341/trisomy-13#ref_2976
- ↑ Jump up to: 3.0 3.1 3.2 3.3 3.4 3.5 3.6 Trisomy 13 Syndrome. NORD (National Organization for Rare Disorders). Published 2007. Accessed March 28, 2021. https://rarediseases.org/rare-diseases/trisomy-13-syndrome/
- ↑ Jump up to: 4.0 4.1 4.2 Noriega MA, Siddik AB. Trisomy 13. 2023 Aug 13. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2023 Jan–. PMID: 32644517.
- ↑ Bugge M, Collins A, Hertz JM, et al. Non-disjunction of chromosome 13. Hum Mol Genet. 2007;16(16):2004-2010. doi:10.1093/hmg/ddm148
- ↑ Hall HE, Chan ER, Collins A, et al. The origin of trisomy 13. Am J Med Genet A. 2007;143A(19):2242-2248. doi:10.1002/ajmg.a.31913
- ↑ Jump up to: 7.0 7.1 Jinawath, N. et al. Mosaic trisomy 13: understanding origin using SNP array. Journal of Medical Genetics 48, 323–326 (2010).
- ↑ Jump up to: 8.0 8.1 8.2 Korf BR, Irons MB. Cell Division and Chromosomes. In: Human Genetics and Genomics. Vol 4th ed. Wiley-Blackwell; 2012.
- ↑ Koole FD, Velzeboer CM, van der Harten JJ. Ocular abnormalities in Patau syndrome (chromosome 13 trisomy syndrome). Ophthalmic Paediatr Genet. 1990;11(1):15-21. doi:10.3109/13816819009012944
- ↑ Koole FD, Velzeboer CM, van der Harten JJ. Ocular abnormalities in Patau syndrome (chromosome 13 trisomy syndrome). Ophthalmic Paediatr Genet. 1990;11(1):15-21. doi:10.3109/13816819009012944
- ↑ Jump up to: 11.0 11.1 11.2 11.3 11.4 Springett A, Wellesley D, Greenlees R, et al. Congenital anomalies associated with trisomy 18 or trisomy 13: A registry-based study in 16 European countries, 2000-2011. Am J Med Genet A. 2015;167A(12):3062-3069. doi:10.1002/ajmg.a.37355
- ↑ Rasmussen SA, Wong L-YC, Yang Q, May KM, Friedman JM. Population-based analyses of mortality in trisomy 13 and trisomy 18. Pediatrics. 2003;111(4 Pt 1):777-784. doi:10.1542/peds.111.4.777
- ↑ Parker SE, Mai CT, Canfield MA, et al. Updated National Birth Prevalence estimates for selected birth defects in the United States, 2004-2006. Birth Defects Res Part A Clin Mol Teratol. 2010;88(12):1008-1016. doi:10.1002/bdra.20735
- ↑ Jump up to: 14.0 14.1 Savva GM, Walker K, Morris JK. The maternal age-specific live birth prevalence of trisomies 13 and 18 compared to trisomy 21 (Down syndrome). Prenat Diagn. 2010;30(1):57-64. doi:10.1002/pd.2403
- ↑ Springett, A., Wellesley, D., Greenlees, R., Loane, M., Addor, M. C., Arriola, L., Bergman, J., Cavero-Carbonell, C., Csaky-Szunyogh, M., Draper, E. S., Garne, E., Gatt, M., Haeusler, M., Khoshnood, B., Klungsoyr, K., Lynch, C., Dias, C. M., McDonnell, R., Nelen, V., O'Mahony, M., … Morris, J. K. (2015). Congenital anomalies associated with trisomy 18 or trisomy 13: A registry-based study in 16 European countries, 2000-2011. American Journal of Medical Genetics. Part A, 167A(12), 3062-3069.
- ↑ Jump up to: 16.0 16.1 16.2 16.3 16.4 16.5 16.6 16.7 16.8 16.9 Lueder GT. Clinical ocular abnormalities in infants with trisomy 13. Am J Ophthalmol. 2006;141(6):1057-1060. doi:10.1016/j.ajo.2005.12.048
- ↑ Jump up to: 17.0 17.1 Kruszka P, Muenke M. Syndromes associated with holoprosencephaly. Am J Med Genet C Semin Med Genet. 2018;178(2):229-237. doi:10.1002/ajmg.c.31620
- ↑ Zeng Y, Lin W, Zhuang W. Safety concerns of paternal drug exposure on fertility, pregnancy and offspring: An analysis based on the FDA adverse event reporting system. Andrology. 2024 Oct 26. doi: 10.1111/andr.13790.
- ↑ Jump up to: 19.0 19.1 19.2 19.3 Cogan DG, Kuwabara T. Ocular Pathology of the 13-15 Trisomy Syndrome. Arch Ophthalmol. 1964;72:246-253. doi:10.1001/archopht.1964.00970020246021
- ↑ Jump up to: 20.0 20.1 20.2 20.3 20.4 Kepple JW, Fishler KP, Peeples ES. Surveillance guidelines for children with trisomy 13. Am J Med Genet A. 2021 May;185(5):1631-1637.
- ↑ Jump up to: 21.0 21.1 21.2 Petry P, Polli JB, Mattos VF, et al. Clinical features and prognosis of a sample of patients with trisomy 13 (Patau syndrome) from Brazil. Am J Med Genet A. 2013;161A(6):1278-1283. doi:10.1002/ajmg.a.35863
- ↑ Jump up to: 22.0 22.1 22.2 Bunting R, Leitch J. Buphthalmos in trisomy 13. Eye (Lond). 2005;19(4):487-488. doi:10.1038/sj.eye.6701454
- ↑ Jump up to: 23.0 23.1 23.2 23.3 Chan A, Lakshminrusimha S, Heffner R, Gonzalez-Fernandez F. Histogenesis of retinal dysplasia in trisomy 13. Diagn Pathol. 2007;2:48. doi:10.1186/1746-1596-2-48
- ↑ Jump up to: 24.0 24.1 Allen JC, Venecia G, Opitz JM. Eye findings in the 13 trisomy syndrome. Eur J Pediatr. 1977;124(3):179-183. doi:10.1007/BF00452109
- ↑ Jump up to: 25.0 25.1 Vislisel JM. Persistent fetal vasculature (PFV). EyeRounds. Published May 26, 2014. Accessed December 9, 2020. https://webeye.ophth.uiowa.edu/eyeforum/atlas/pages/PFV/index.htm
- ↑ Jaru-Ampornpan P, Kuchtey J, Dev VG, Kuchtey R. Primary congenital glaucoma associated with Patau syndrome with long survival. J Pediatr Ophthalmol Strabismus. 2010;47 Online:e1-4. doi:10.3928/01913913-20100618-09
- ↑ Keith CG. The ocular manifestations of trisomy 13-15. Trans Ophthalmol Soc U K. 1966;86:435-454.
- ↑ Hoepner J, Yanoff M. Ocular anomalies in trisomy 13-15: an analysis of 13 eyes with two new findings. Am J Ophthalmol. 1972;74(4):729-737. doi:10.1016/0002-9394(72)90836-7
- ↑ Mirmohammadsadeghi, A., Akbari, M. R., & Malekpoor, A. (2017). Ocular manifestations in edward's syndrome, a case report and literature review. Journal of Current Ophthalmology, 29(4), 329-331. doi:http://dx.doi.org.proxy.lib.duke.edu/10.1016/j.joco.2017.06.005
- ↑ Jump up to: 30.0 30.1 30.2 Jain SF, Saoirse Y, Conahan B, Suh D. Ocular Findings in Trisomy 13: Nasolacrimal Duct Stenosis Case Series. OJOph. 2019;09(04):161-164. doi:10.4236/ojoph.2019.94017
- ↑ Jump up to: 31.0 31.1 Magni R, Pierro L, Brancato R. Microphthalmos with colobomatous orbital cyst in trisomy 13. Ophthalmic Paediatr Genet. 1991;12(1):39-42. doi:10.3109/13816819109023083
- ↑ Ernst J, Eldib A, Scanga HL, Nischal KK. Keratoconus in a child with partial trisomy 13. Ophthalmic Genet. 2021 Jun;42(3):360-363. doi: 10.1080/13816810.2021.1897849. Epub 2021 Apr 16. PMID: 33858272.
- ↑ Copel JA, Kohari K, Merriam AA. Prenatal testing. In: García-Velasco JA, Seli E, eds. Human Reproductive Genetics. Academic Press; 2020:201-221. doi:10.1016/B978-0-12-816561-4.00012-0
- ↑ Shiefa, S., Amargandhi, M., Bhupendra, J., Moulali, S. & Kristine, T. First Trimester Maternal Serum Screening Using Biochemical Markers PAPP-A and Free β-hCG for Down Syndrome, Patau Syndrome and Edward Syndrome. Indian Journal of Clinical Biochemistry 28, 3–12 (2012).
- ↑ Boddupally K, Rani Thuraka E. Artificial intelligence for prenatal chromosome analysis. Clin Chim Acta. 2024 Jan 1;552:117669.
- ↑ Kanigowska K, Grałek M, Seroczyńska M. [Clinical ocular manifestation of Patau’s syndrom (trisomy 13)--own observations]. Klin Oczna. 2011;113(7-9):263-265.
- ↑ Nelson, K. E., Rosella, L. C., Mahant, S. & Guttmann, A. Survival and Surgical Interventions for Children With Trisomy 13 and 18. JAMA 316, 420 (2016).
- ↑ St Louis JD, Bhat A, Carey JC, Lin AE, Mann PC, Smith LM, Wilfond BS, Kosiv KA, Sorabella RA, Alsoufi B. The American Association for Thoracic Surgery 2023 Expert Consensus Document: Recommendation for the care of children with trisomy 13 or trisomy 18 and a congenital heart defect. J Thorac Cardiovasc Surg. 2024 Jan 27:S0022-5223(23)01205-9.
- ↑ Morris JK, Savva GM. The risk of fetal loss following a prenatal diagnosis of trisomy 13 or trisomy 18. Am J Med Genet A. 2008;146A(7):827-832. doi:10.1002/ajmg.a.32220
- ↑ Jump up to: 40.0 40.1 Baty BJ, Jorde LB, Blackburn BL, Carey JC. Natural history of trisomy 18 and trisomy 13: II. Psychomotor development. Am J Med Genet. 1994;49(2):189-194. doi:10.1002/ajmg.1320490205
- ↑ Jump up to: 41.0 41.1 Meyer RE, Liu G, Gilboa SM, et al. Survival of children with trisomy 13 and trisomy 18: A multi-state population-based study. Am J Med Genet A. 2016;170A(4):825-837. doi:10.1002/ajmg.a.37495



