Toxoplasmosis
All content on Eyewiki is protected by copyright law and the Terms of Service. This content may not be reproduced, copied, or put into any artificial intelligence program, including large language and generative AI models, without permission from the Academy.
Disease Entity
Disease
Toxoplasmosis is the most common cause of infectious retinochroiditis in humans.
Etiology
The causative organism, Toxoplasma gondii, is a single-cell, obligate, intracellular protozoan parasite. Cats are the definitive host for T. gondii, however, humans and a wide range of mammals, birds, and reptiles, may also serve as intermediate hosts. T. gondii has three forms: (1) the oocyst (soil form), (2) the tachyzoite (active infectious form), and (3) the tissue cyst (latent form).
Epidemiology
T. gondii’s presence in nature is widespread, but human infections are not equally distributed around the world. It has been estimated that 1 billion people are infected worldwide. Rates of infection are highest in tropical areas and relatively low in dry, arid areas and cold areas. This likely reflects the environment which are most amenable to the organism's proliferation in the environment. In the United States approximately 22.5% are IgG positive for toxoplasmosis and up to 70% in some studies. Ocular disease secondary to toxoplasmosis is estimated to be around 2% in the United States, 17.7% in Brazil, and up to 43% in Africa.
Risk Factors
Exposure to environments where the infectious organism is found, especially those frequented by felines. Increased risk of infection in males, if you own more than 3 kittens, and eating raw or undercooked meat (lamb, ground beef, shell fish, game).
General Pathology
A necrotizing retinitis occurs with vasculitis and destruction of the retina.
Pathophysiology
Infection with Toxoplasma gondii.
Toxoplasma Serotypes:
Type 1: Latin America more severe disease
Type 2: Europe/North America less aggressive
Diagnosis
Physical Examination
List of classic and atypical findings which have been reported.
Classic findings
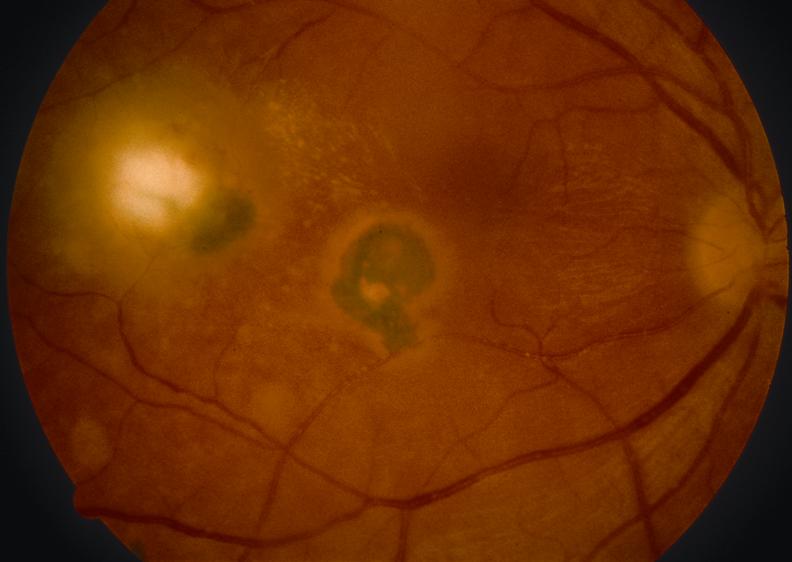
- White focal retinitis with overlying vitreous inflammation (“headlight in the fog”)
- Accompanying nearby or adjacent pigmented retinochoroidal scar
- Vitreous inflammation (mild, moderate, or severe )
- Secondary nongranulomatous iridocyclitis
- Granulomatous and stellate keratic precipitates possible
- Inflammatory ocular hypertension 10% to 15% of cases
- Retinal vasculitis (typically near the focus of Retinochoroiditis)
Atypical findings which can accompany retinochoroiditis
- Papillitis
- Neuroretinitis
- Retrobulbar neuritis
- Scleritis
- Retinal detachment
- Punctate outer retinitis
- Branch retinal artery occlusion
- Frosted branch angiitis
- Coats’-type response
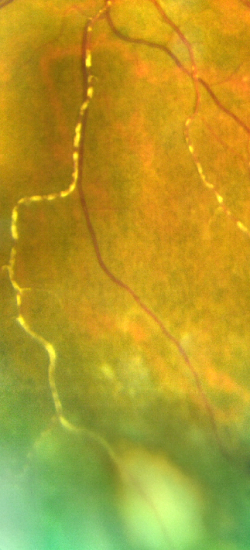
- Kyrieleis arteritis or segmental retinal arteritis (SRA)[2]
- Fuchs’-like anterior uveitis
- Multifocal diffuse necrotizing retinitis
Atypical findings which can exist in absence of retinochoroiditis
- Retinal vasculitis
- Unilateral neuroretinitis (optic disk edema, macular star)
- Inflammation in absence of overt necrotizing retinitis
- Unilateral pigmentary retinopathy mimicking retinitis pigmentosa
Recurrence
Recurrent lesions tend to occur at the margins of old scars, but they also can occur elsewhere in the fundus. The risk of recurrence is highest within the first year of the initial episode. Older age also predisposes to higher risk of recurrence. The American Academy of Ophthalmology published a report entitled "Interventions for Toxoplasma Retinochoroiditis" and agreed that there was level 2 evidence that supports the long-term prophylactic treatment with combined trimethoprim and sulfamethoxazole might reduce recurrences.
Symptoms
Typical symptoms of active disease are floaters and blurred vision. Eye pain and redness if the patient has a secondary iritis.
Clinical diagnosis
Clinical evaluation and diagnosis is the most important part of diagnosing toxoplasmosis retinitis.
Diagnostic Evaluations
Because of the high prevalence of positive toxoplasma titers in many populations, the use of serology in the diagnosis is mainly limited to reassuring the clinician that toxoplasmosis should remain in the differential. If the IgG titers for toxoplasmosis are completely negative, down to a 1:1 dilution, then toxoplasmosis is completely ruled out in an immunocompetent person. It is possible that an immunosuppressed patient could have a completely negative immunoglobulin titers and have active ocular toxoplasmosis infection.
The development of polymerase chain reaction (PCR) has been very helpful for the diagnosis of atypical or difficult cases. Detection of T gondii DNA by PCR in both aqueous humor and vitreous fluid is both sensitive and specific.
Differential diagnosis
Infectious
- Tuberculosis
- Viral Retinitis (CMV, HSV, VZV)
- Toxocariasis
- Bartonella
- Syphilis
- Endophthalmitis
Autoimmune
- Behcet's disease
- Systemic autoimmune diseases
- Retinal vasculitis
Management
Not all lesions warrant treatment. Indications for treatment include:
- Macular threatening or optic nerve or papillomacular lesions
- Close proximity of lesions to major retinal vessels
- Dense vitritis
- Marked visual impairment
- Larger lesions
- Pregnancy
- Monocular status
- The immunocompromised host
Medical therapy
Systemic Pyrimethamine, sulfadiazine, and corticosteroids
This is the classic triple therapy used in the treatment of toxoplasmosis. In a survey of the American Uveitis Society this combination triple therapy was cited as the treatment of choice by 32% of respondents. Of note, Pyrimethamine is a folic acid antagonist and can cause dose-related suppression of the bone marrow, which is mitigated by concurrent administration of folinic acid (leucovorin)
Systemic Clindamycin
An additional 27% of American Uveitis society respondents added clindamycin to the previously mentioned therapy. In general, treatment is given until the inflammatory reaction begins to decrease and the retinal lesion shows signs of healing, which is usually 4 to 6 weeks.
Systemic Azithromycin
Rothova et al reported successful treatment of ocular toxoplasmosis in immunocompetent patients with azythromicin alone
Systemic Bactrim
Intravitreal Clindamycin (+/- steroids)
Surgery
Surgical treatment is not indicated for treatment of toxoplasmosis infection.
Surgical treatment may be required for retinal detachments which occur as a complication of toxoplasmosis retinochoroiditis.
Another important consideration when considering surgery in a patient who has had toxoplasmosis, is the use of prophylactic antibiotic treatment to decrease the risk of recurrence of toxoplasmosis at the time of surgery.
Prognosis
In general final visual acuity depends on the location of the active infection, degree of inflammation, and development of complications related to ocular inflammation. Recurrences can occur in patients therefore long term follow up is recommended.
Patient Education
Clinicians should be aware and should educate parents and older children with congenital infection that late-onset retinal lesions and relapse can occur many years after birth. They should also be reassured that risk of bilateral visual impairment appears to be low. Identifying activation or reactivation early and getting treated will give the best prognosis.
Additional Resources
- Porter D, Vemulakonda GA. Toxoplasmosis. American Academy of Ophthalmology. EyeSmart/Eye health. https://www.aao.org/eye-health/diseases/toxoplasmosis-8. Accessed March 25, 2019.
References
- ↑ American Academy of Ophthalmology. Toxoplasmosis. https://www.aao.org/image/toxoplasmosis-4 Accessed August 24, 2022.
- ↑ Khadamy J (October 15, 2023) Atypical Ocular Toxoplasmosis: Multifocal Segmental Retinal Arteritis (Kyrieleis Arteritis) and Peripheral Choroidal Leision. Cureus 15(10): e47060. doi:10.7759/cureus.47060
- Jones JL, Dargelas V, Roberts J, Press C, Remington JS, Montoya JG. Risk factors for Toxoplasma gondii infection in the United States. Clin Infect Dis. 2009 Sep 15;49(6):878-84. doi: 10.1086/605433. PubMed PMID: 19663709.
- Bosch-Driessen LH, Plaisier MB, Stilma JS, Van der Lelij A, Rothova A. Reactivations of ocular toxoplasmosis after cataract extraction. Ophthalmology. 2002 Jan;109(1):41-5. PubMed PMID: 11772577.
- Kim SJ, Scott IU, Brown GC, Brown MM, Ho AC, Ip MS, Recchia FM. Interventions for toxoplasma retinochoroiditis: a report by the American Academy of Ophthalmology.Ophthalmology. 2013 Feb;120(2):371-8. doi: 10.1016/j.ophtha.2012.07.061. Epub 2012 Oct 11. Review. PubMed PMID: 23062648.
- Conrady CD, Besirli CG, Baumal CR, et al. Ocular Toxoplasmosis after Exposure to Wild Game [published online ahead of print, 2021 Feb 9]. Ocul Immunol Inflamm. 2021;1-6. doi:10.1080/09273948.2020.1854316
- Rothova A, Meenken C, Buitenhuis HJ, et al. Therapy for ocular toxoplasmosis. Am J Ophthalmol.1993; 115:517-23. [PubMed: 8470726]