Teleophthalmology
All content on Eyewiki is protected by copyright law and the Terms of Service. This content may not be reproduced, copied, or put into any artificial intelligence program, including large language and generative AI models, without permission from the Academy.
Significance
Teleophthalmology is evolving into an integral clinical tool in the American healthcare system. It allows clinicians to provide quality health care outside of clinics, thus improving access to medical care, patient outcomes, and overall patient satisfaction. Eye screenings are conducted within the community, often in schools, health fairs, and places of worship.[1] Bringing the clinic directly to the patients allows health professionals to capture more vision threatening diseases (VTD) such as diabetic retinopathy, glaucoma, age-related macular degeneration, and cataract.[2] By detecting VTD’s early on, teleophthalmology allows for timely referral to appropriate experts and consequently, earlier treatment.[1][3] This tool is actively being used in the US army to support the accessibility of ophthalmic care for military personnel, and allied forces worldwide.[4]
Screening sites may not have the comprehensive eye examination equipment that is readily available in a permanent clinical facility, such as slit lamps, phoropters and other imaging equipment.[3] On-site screenings may utilize smaller footprint equipment including an automated refractor, non-contact tonometer, non-mydriatic retinal camera, and optical coherence tomography. Often, screenings are advertised to the local community via email, newsletters, or by word of mouth. A qualified ophthalmologist, known as the reader, is available either in person or remotely to interpret the data collected and develop an assessment and treatment plan for the screened patient.
Teleophthalmology may potentially provide health services to underserved and remote rural populations who otherwise may not have access to specialized eye care.[3][5] Tele-ophthalmology has the potential to improve ophthalmic care and diagnose VTDs.[1][6] Ultimately, the goal of tele-ophthalmology is to decrease the global burden of vision threatening disease and improve the quality of life for patients with eye diseases worldwide.
Screening Methods
Patient History
A focused patient history is acquired prior to screening using a standardized intake form. Demographic data including age, gender, and ethnicity, as well as medication use and a brief medical ocular and surgical history is obtained. Additional data may be collected particularly if the ophthalmic evaluation is conducted as a part of a health fair where other medical specialties are conducting health screening (for example: Body Mass Index, blood pressure, pulse oximetry, smoking status, dietary habits). Additionally, patients are asked if they have a family history of pertinent medical conditions like hypertension, hyperlipidemia, diabetes, cardiovascular disease, and eye conditions.
Visual Acuity
Visual acuity is measured using a standard Snellen chart with patients 20 feet away (or 6 meters) or more often, using a modified eye chart with corrected distance from the chart. This test is central to any ophthalmic evaluation and can help screeners identify subjects with blurred vision (refractive errors as seen in myopia and hyperopia as well as amblyopia). The visual acuity outcome then has to be placed in the context of the eye evaluation to help identify the cause of blurred vision.[7]
Automated Refractor
The automated refractor measures the estimated correction needed for a patient’s eyes to focus properly an image at distance. Automated refractors are key in measuring the eye’s refractive state in sphere, cylinder, and axis. These measurements are then fine tuned in order to be translated into an eyeglasses corrective prescription for the patient. Because these measurements are automated, this measurement requires minimal communication. Autorefractors are particularly useful when there is a language barrier between provider and subject as well as with younger children and those with disabilities.[8] [9] Automated refractors generate an estimate of the refractive error of the eye and are then fine tuned with an actual refraction by a health care provider prior to being dispensed.
Non-contact Tonometry
Non-contact tonometry, as in "air-puff" tonometry, is used to measure intraocular pressure in a noninvasive manner. These instruments are easy to handle and do not require anesthetic eye drops or fluorescein administration, making it especially useful in a field setting and with less cooperative patient groups such as young children and disabled persons.[10] This is a critical test in screening for eye diseases like glaucoma, a leading cause of blindness in people over 60 and one of the leading causes of blindness globally.[11] Non-contact tonometry correlates well with other contact measurements (as in Goldmann applanation tonometry) yet may over or under-estimate intraocular pressure in some individuals and may need to be interpreted in the context of a screening test and be rechecked in the office by an ophthalmologist for confirmation.
Fundus Photography
Digital non-mydriatic retinal cameras allow for imaging of the retina, retinal vasculature, optic disc, and macula. It is a vital tool in evaluation as it allows diagnosing disease processes like diabetic retinopathy, age-related macular degeneration, glaucoma, and hypertensive retinopathy.[12] Cataracts may also be suspected when fundus image quality is limited by media opacities. Low light conditions are used to induce physiological pupillary dilation, foregoing the need for dilating (mydriatic) eye drops and thus making fundus photography a more accessible tool to be used within the community.[5] Additionally, fundus autofluorescence (FAF) has been successfully integrated in community based ocular screening programs. Some retinal camera models can perform nonmydriatic 45 degree color imaging and FAF imaging, allowing the convenience of a single instrument needing to be transported. FAF significantly improves the screeners’ abilities to characterize retinal changes and highlights certain ocular pathology more predominantly than color fundus photography alone.[13]
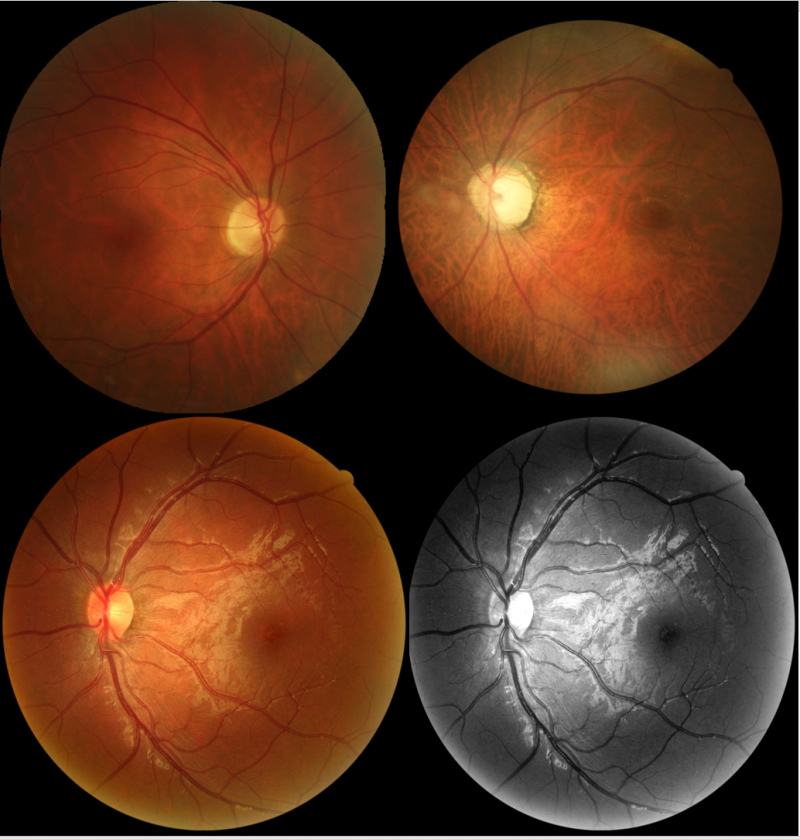
Optical Coherence Tomography
Optical Coherence Tomography (OCT) is a noninvasive imaging test that allows screeners to obtain high resolution, cross-sectional images of the retina, retinal nerve fiber layer, optic nerve head, and anterior segment. The OCT is an invaluable tool in ocular screenings, especially in the cases of age-related macular degeneration, glaucoma, and diabetic retinopathy.[14][15] Anterior segment OCT can also be used to identify narrow angle at risk for potential glaucoma.
Future Directions and Challenges
The major challenges of tele-ophthalmology revolve around patient retention and pursuing follow up appointments after ocular disease detection. A number of patients, even if given means of transportation to travel to their appointments, do not keep their follow up appointments.[16] Additionally, regulatory challenges restrict the wide use of tele-ophthalmology as a viable option for health care throughout the United States. Future initiatives need to focus on engaging policies to regulate telemedicine applications in ophthalmology. Challenges also remain regarding cost and reimbursement for tele-ophthalmology services, thus limiting a more widespread adoption of tele-ophthalmology. The American Academy of Ophthalmology and other societies have recognized the value of tele-ophthalmology in providing valuable ocular services especially to subjects in remote and under-served communities. The Veteran Administration has also initiated landmark initiatives in tele-ophthalmology to enhance the care of veterans. In India, a tele-ophthalmology model, KIDROP has been successful in detecting retinopathy of prematurity and delivering care to underserved areas. In the settings epidemics and pandemic like COVID19 infection (2020), tele-ophthalmology can become a preferred mode for delivering initial contact with the patient. Tele-ophthalmology can reduce healthcare costs, increase access in underserved areas, and improve DR screening and detection; however, reimbursements by insurance companies and failure to implement the technology and much needed areas remain barriers to the use of this technology.
Artificial intelligence (AI)-Human hybrid based Teleophthalmology is being evaluated as an alternative improved means of DR screening. [17]
Additional Resources
- American Academy of Ophthalmology. Teleophthalmology: How to Get Started https://www.aao.org/practice-management/article/teleophthalmology-how-to-get-started San Francisco: American Academy of Ophthalmology, 2020. Accessed April 10, 2020.
References
- ↑ Jump up to: 1.0 1.1 1.2 Sreelatha, O. K. & Ramesh, S. V. Teleophthalmology: improving patient outcomes? Clin. Ophthalmol. Auckl. NZ 10, 285–295 (2016).
- ↑ Szirth, B., Khouri, A., Bhagat, N. & Shahid, K. New Concepts in Screening for Vision Threatening Disease. Invest. Ophthalmol. Vis. Sci. 48, 1578–1578 (2007).
- ↑ Jump up to: 3.0 3.1 3.2 Rural and Remote Health. Available at: https://rrh.org.au/. (Accessed: 27th June 2018)
- ↑ Anthony CM, Altman AH, Otte B, Mines MJ, Mazzoli RA, Lappan CM, Legault GL. Teleophthalmology in the United States Army: A Review From 2004 Through 2018. Mil Med. 2023 Jan 4;188(1-2):e182-e189.
- ↑ Jump up to: 5.0 5.1 Shahid, K. et al. Ocular Telehealth Screenings in an Urban Community. Telemed. E-Health 18, 95–100 (2012).
- ↑ Sangani R, Henry RK, Shah MK, Szirth B, Bhagat N. Use of Resident Tele-Ophthalmic Diagnosis to Aid in Management of Vision-Compromising Retinal Pathology. ARVO . May 2022.
- ↑ Holladay, J. T. Proper Method for Calculating Average Visual Acuity. J. Refract. Surg. 13, 388–391 (1997).
- ↑ Pesudovs, K. & Weisinger, H. S. A Comparison of Autorefractor Performance. Optom. Vis. Sci. 81, 554 (2004).
- ↑ Saw, S. M. et al. Near-Work Activity and Myopia in Rural and Urban Schoolchildren in China. J. Pediatr. Ophthalmol. Strabismus 38, 149–155 (2001).
- ↑ Raina, U. K., Rathie, N., Gupta, A., Gupta, S. K. & Thakar, M. Comparison of Goldmann applanation tonometer, Tono-Pen and noncontact tonometer in children. Oman J. Ophthalmol. 9, 22–26 (2016).
- ↑ Parihar, J. K. S. Glaucoma: The ‘Black hole’ of irreversible blindness. Med. J. Armed Forces India 72, 3–4 (2016).
- ↑ Kolomeyer, A. M., Baumrind, B. R., Szirth, B. C., Shahid, K. & Khouri, A. S. Fundus Autofluorescence and Colour Fundus Imaging Compared During Telemedicine Screening in Patients with Diabetes. J. Telemed. Telecare 19, 209–212 (2013).
- ↑ Kolomeyer, A. M., Nayak, N. V., Szirth, B. C. & Khouri, A. S. Fundus Autofluorescence Imaging in an Ocular Screening Program. International Journal of Telemedicine and Applications (2012). doi:10.1155/2012/806464
- ↑ Adhi, M. & Duker, J. S. Optical coherence tomography – current and future applications. Curr. Opin. Ophthalmol. 24, 213–221 (2013).
- ↑ Shah MK, Henry RK, Szirth B, Bhagat N. Utility of Remote Point-Of-Care Tele-Retinal Imaging for Screening and Diagnosis of Diabetic Retinopathy: A Pilot Study. ARVO. May 2022
- ↑ Kolomeyer, A. M. et al. Software-Assisted Analysis During Ocular Health Screening. Telemed. E-Health 19, 2–6 (2013).
- ↑ Dow ER, Khan NC, Chen KM, Mishra K, Perera C, Narala R, Basina M, Dang J, Kim M, Levine M, Phadke A, Tan M, Weng K, Do DV, Moshfeghi DM, Mahajan VB, Mruthyunjaya P, Leng T, Myung D. AI-Human Hybrid Workflow Enhances Teleophthalmology for the Detection of Diabetic Retinopathy. Ophthalmol Sci. 2023 May 12;3(4):100330.
- Xia, T., Patel, S. N., Szirth, B. C., Kolomeyer, A. M. & Khouri, A. S. Software-Assisted Depth Analysis of Optic Nerve Stereoscopic Images in Telemedicine. Int. J. Telemed. Appl. 2016, 7603507 (2016).