Nocardia and the Eye
All content on Eyewiki is protected by copyright law and the Terms of Service. This content may not be reproduced, copied, or put into any artificial intelligence program, including large language and generative AI models, without permission from the Academy.
Disease Entity
ICD10:H16.
Disease
Nocardia is a rare cause of ocular infections. Ocular infections caused by Nocardia include keratitis, scleritis, conjunctivitis, canaliculitis, dacrocystitis, orbital cellulitis, and endophthalmitis.[1] Corneal infections being the most common.[1] The diagnosis is often missed or delayed as the clinical picture may resemble other causative organisms. Patients often suffer from significant ocular morbidity for a prolonged period before an accurate diagnosis is made. Nocardia infections do not respond to commonly used first-line medications for bacterial keratitis, such as fluoroquinolones. With the initiation of appropriate therapy, Nocardia infections respond promptly and carries a good prognosis.
Etiology
Nocardia belong to the bacterial order Actinomycetales that comprises three families and more than 40 genera; 14 of which seem to be relevant to human and veterinary medicine.[1] Originally classified as a fungus, Nocardia are now recognized as a true bacteria. Nocardia are aerobic, gram-positive, non-motile, branching, filamentous bacteria (less than 1.5 micrometers in diameter).[1] There are at least 12 accepted species within the genus Nocardia. These include N. asteroides, N. amarae, N. brasilinesis, N. brevicatea, N. carnea, N. farcinica, N. nova, N. otitidis-caviarum (N. caviae), N. pinensis, N. seriolae, N. transvalensis and N. vaccinni.[1]
Nocardia represent indigenous microflora of soil, mud, dust and decaying vegetation.[1] Pathogenic species of Nocardia have been detected in house dust, beach sand, garden soil, and swimming pools.[1] Nocardia species do not present as normal flora either in the eye or in the respiratory tract. Actinomycetes are found around the word with no racial or occupational predilection noted.[2] Nocardia asteroides is prominent in temperate regions and Nocardia brasiliensis is found in tropical and subtropical areas.[1]
Risk Factors
Predisposing factors are trauma, surgery, corticosteroid use, and contact lens wear.[2] Traumatizing agents reported include vegetative material, dirt, stone, gravel, flying insects; as well as injuries from nails and fish line sinkers.[2] Nocardia has been isolated from eyes with contact lens associated keratitis.[2] Inappropriate lens hygiene and extended wear can predispose to infectious keratitis.
N.asteroides keratitis has been found following surgical trauma. Nascimento et al reported a case following an uncomplicated myopic keratomileusis.[3] Perez-San-tonja et al found Nocardia following uncomplicated LASIK treatment for myopia.[4] One case has been reported following penetrating keratoplasty.[5] The use of topical corticosteroids may also be a predisposing factor. A rabbit model of topical corticosteroid use in Nocardia keratitis observed the development of large granulomatous lesions with extension into the anterior chamber in those treated with topical steroids, whereas no extension was noted in those not treated with steroids.[6] The Steroids for Corneal Ulcers Trial (SCUT) showed that the use of topical steroids was associated with worse clinical outcomes in Norcardia keratitis at 3 months.[7]
Pathophysiology
The rate of growth and multiplication of Nocardia is slow and does not cause a fulminant infection immediately post invasion. It is presumed that parasite-related virulence is involved, as the organism can infect healthy individuals. The virulence is partly due to structural and biochemical cellular changes that occur in the growth cycle. A combination of mycolic acids within the cell wall are altered throughout nocardial growth, that contributes to its toxicity and virulence throughout the growth cycle.[8][9]A toxic cell wall component known as trehalose 6,6’-dimycolate is associated with the virulence of Nocardia through the inhibition of phagosome-lysosome fusion in macrophages and the escape of lysosome fusion in macrophages.[10][11][12] There is a probably a role of nocardial surface membrane-bound catalase and superoxide dismutase (SOD) in its resistance to being killed by polymorphonuclear leukocytes.[13] There is also a possible role of Nocardia exotoxins in causing host-tissue damage.[14][15]
Primary Prevention
Primary prevention of Nocardia eye infections involves avoiding the above risk factors. This includes proper contact lens use, avoidance of potential insults by wearing protective eyewear, minimizing topical steroid use, and employing proper aseptic technique during surgical procedures.
Diagnosis
History
A detailed history should be taken from patients presenting with suspected microbial ocular infections. This should begin with the characteristics and onset of the symptoms, including any pain, photophobia, or vision changes. It is important to inquire about any predisposing factors, such as contact lens wear including the type of lens, wearing time and lens hygiene, recent trauma including surgical procedures, use of immunosuppressive agents and pre-existing eye conditions such as dry eye and neurotrophic keratopathy. Other relevant medical history such as decreased immunologic defenses or systemic immunosuppression medications is useful in assessing the risk of microbial ocular infections.
Physical Examination
A comprehensive ocular examination should be conducted. This includes vision, pupil assessment, extraocular motility and alignment, intraocular pressures, confrontation, and external examination. This is followed by a slit-lamp examination. This involves the systematic assessment of the lids, lashes and lacrimal system, conjunctiva and sclera, cornea, anterior chamber, iris, lens and anterior vitreous. A dilated examination should be performed to assess the optic nerve, macula, vessels, and periphery to rule out posterior pole involvement. Clinical features of Nocardia infections of the eye are found below.
Signs
Nocardia Keratitis
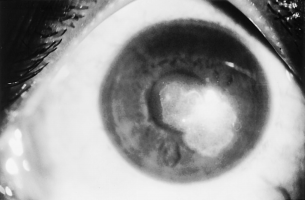
The conjunctiva may show a fine papillary reaction. Keratitis can present as a nonspecific punctate epitheliopathy or an ulcer with margins studded with yellow white pinhead sized superficial infiltration (Figure. 1).[1][16] Patchy infiltrations are predominantly anterior stromal with involvement of the epithelial and subepithelial tissues.[16] These infiltrates often present in a classical wreath like pattern with satellite lesions (Figure. 2).[1][16] Infiltrates are usually situated in the midperiphery of the cornea adjacent to the sites of minor corneal trauma or abrasion.[16]
Usually, there is an overlying epithelial defect and the surrounding stroma is clear.[16] Anterior chamber reaction and hypopyon are usually associated.[16] Intraocular pressure is usually normal. [16] Keratic precipitates and endothelial ring deposits on the endothelium have also been reported.[17] Peripheral deep neovascularization may be seen. The cornea may be hypesthetic. Superficial granular infiltrates can coalesce into white plaque, resulting in corneal ulceration. [18] A summary of the typical and atypical features of Nocardia keratitis is detailed in Table 1.[1]
Nocardia Endophthalmitis
Intraocular nocardiosis is rare. Reported Nocardia endophthalmitis are commonly from haematogenous (endogenous) spread of the organism and occasionally exogenously. Hematogenous spread occurs to the eye, resulting in choroidal abscess or endophthalmitis.[19][20] The clinical picture consists of a large, yellow choroidal lesion with hemorrhage in the overlying retina. Chorioretinal lesions are usually located in the central and paracentral regions. Retinal thickening and exudative retinal detachment can occur, as well as optic nerve head elevation. Nocardia endophthalmitis can present with a hypopyon and no posterior findings.[21] Endogenous Nocardia endophthalmitis is most commonly unilateral. Bilateral nocardial infections have been reported in cases of pulmonary Nocardia asteroides infection and in those with hypogammaglobulemia.[22] Exogenous Nocardia can occur after cataract surgery, penetrating injury and trabeculectomy. There is anterior segment inflammation and hypopyon. Yellowish-white nodules can be seen over the iris (Figure 3) and puffball opacities may be visible in the anterior vitreous.[23]
Nocardia Scleritis
Isolated Nocardia scleritis is rare and results as an extension of corneal infection. Predisposing factors including cataract surgery, exposed scleral buckle following cataract surgery, explantation of scleral buckle and trauma.[24] Signs include conjunctival and scleral inflammation and areas of necrosis, thinning, hemorrhage, and discharge. Scleral abscesses and engorged episcleral vessels may be seen. Anterior chamber reaction is associated.[24]
Nocardia Other
Other ocular manifestations of Nocardia include conjunctivitis, infection of the lacrimal system, preseptal cellulitis, and dacryoadenitis.
Symptoms
Nocardia eye infections usually run a protracted course.[1] Presenting symptoms consist of pain, photophobia, blepharospasm, and lid swelling. The amount of pain may be out of proportion to clinical findings.[1]
Clinical diagnosis
A diagnosis of Nocardia eye infection is made based on the history, symptoms and clinical signs found during the systematic ocular examination.
Diagnostic procedures
The gold standard for the diagnosis of Nocardia keratitis is a scraping from the corneal lesion using a spatula or blade.[25] Usually, multiple scrapings are collected for three smears and to inoculate several culture media allowing for the growth of all types of bacteria (aerobic, facultatively anaerobic, and anaerobic), fungi and Acanthameoba.[25]
Laboratory test
Smears are collected and stained by gram stain, Giemsa stain, and potassium hydroxide with calcoflour white stain. The presence of gram-positive, branching, beaded filaments that stain with 1% acid-fast stain (using 1% sulfuric acid, modified Kinyoun’s method) is suggestive of a Nocardia infection.[26]
Nocardia organisms in culture are not fastidious and grow aerobically on media as tiny, white, dry colonies. They usually grow within 48-72 hours on blood agar, chocolate agar, and Sabouraud’s dextrose agar (without antibiotics). Growth on one medium is considered significant with corresponding smear results and a clinical picture suggestive of Nocardia. Nocardia isolation is considered significant, given that it is not a common contaminant.[26]
Differential diagnosis
Differentials for Nocardia infections include Moraxella, nontuberculous Mycobacteria and filamentous fungi. Moraxella is a gram-positive diplobacillus that can produce corneal ulcers in select patient groups including diabetics, alcoholics and the malnourished.[27][28][29] Moraxella ulcers are oval and located inferiorly on the cornea. Nontuberculous Mycobacteria are acid-fast bacilli that cause slowly progressive corneal infections. The infiltrate in myobacterial infections have indistinct fluffy or feather-like appearances with radiating projections, described as a “snow-flake” or “cracked windshield” appearances.[30][31][32] Filamentous fungi produce slow growing, corneal ulcers with dry, raised infiltrates with hyphate edges and satellite lesions.
Management
General treatment
General treatment consists of medical and surgical therapies. Medical therapies are inclusive of topical and systemic antibiotic treatment. In the case of advanced Nocardia infections, surgical treatment may be warranted. In addition, general measures such as the discontinuation of contact lens wear, avoidance of immunosuppressive agents and avoidance of further insults should be facilitated.
Medical therapy
Nocardia organisms show good susceptibility to amikacin and sulphonamides.[33][34] Present research indicates excellent in vitro activity of amikacin against all Nocardia species, including N. amamiensis, N. thailandica, N. levis, and N. puris.[33] Amikacin 2 to 2.5% hourly is usually initiated as first- line treatment. Aminoglycosides such as gentamicin and tobramycin are second line treatment of this infection. Antimicrobial susceptibility of Nocardia to fluoroquinolones such as ciprofloxacin and clarithromycin varies across species. Gulpczynski et al. reported that Nocardia species including Nocardia nova, N. cyriacigeorgica, N. abscessus and N.brasiliensis showed resistance to ciprofloxacin.[35] Keratitis and scleritis cases respond well to amikacin therapy, the outcome of endophthalmitis management is not encouraging.[36] Oral trimethoprim- sulfamethoxazole 160mg/800mg hourly has been reported as a treatment for persistent scleritis.[37]
Medical follow up
Nocardia infections usually require admission or daily follow up to assess for progress and the suitability of the treatment regimen.
Surgery
Nocardia keratitis is usually well treated with medical therapy. However, surgical treatment may be warranted in the case of progressive corneal thinning, extension beyond the limbus, or presentation at an advanced stage. Surgical options are therapeutic lamellar keratectomy, penetrating keratoplasty, and conjunctival flap. [17][18][38]
Surgical follow up
Close follow up is recommended with a review day one post-operatively.
Complications
Complications described in the literature include progressive thinning leading to perforation, endophthalmitis, and extension to adjoining sclera.[1]
Prognosis
With medical treatment, the ulcer heals rapidly.[39] The infiltrate resolves with minimal scarring, forming a fine superficial nebular opacity (Figure. 5). Nocardia keratitis tends to heal with peripheral vascularization or with vascularized scars. Patients usually continue to be asymptomatic with no recurrences. Visual prognosis is good and if the infection responds well to medical therapy, visual acuity improves.[40][41]
References
- ↑ Jump up to: 1.00 1.01 1.02 1.03 1.04 1.05 1.06 1.07 1.08 1.09 1.10 1.11 1.12 1.13 Sridhar MS et al. Ocular nocardia infections with special emphasis on the cornea. Surv Ophthalmol. 2001. Mar-Apr;45(5):361-78.
- ↑ Jump up to: 2.0 2.1 2.2 2.3 Sridhar MS et al. Clinicomicrobiological review of Nocardia Keratitis. Cornea. 1998 17:17-22.
- ↑ Nascimento EG et al. Nocardia keratitis following myopic keratomileusis. J Refract Surg. 1995. 11:510-1.
- ↑ Perez-Santonja JJ et al. Nocardial keratitis after laser in situ keratomileusis. J Refract Surg. 1997. 13:314–7.
- ↑ Colomina J et al. Corneal ulcer caused by Nocardia asteroides after penetrating keratoplasty. Med Clin (Barc). 1997. 108:424–5.
- ↑ Newmark E et al. Report of a case of Nocardia asteroides keratitis. Am J Ophthalmol. 1971.72:813–5.
- ↑ Lalitha, P., et al. Nocardia keratitis: clinical course and effect of corticosteroids. American journal of ophthalmology 154, 934-939 e931 (2012).
- ↑ Beaman BL, Moring SE. Relationship among cell wall composition, stage of growth, and virulence of Nocardia asteroides GUH-2. Infect Immun. 1988. 56:557–63.
- ↑ Beaman BL, Moring SE, Ioneda T. Effect of growth stage on mycolic acid structure in cell walls of Nocardia asteroides GUH-2. J Bacteriol. 1988. 170:1137–42.
- ↑ Ioneda T et al. Occurence of trehalose mycolate as the major lipidic component carrying toxic property in clinical Nocardia isolates. Rev Microbiol. 1982. 20:345–8.
- ↑ Spargo BJ et al. Cord factor (alpha, alpha-trehalose 6,6-dimycolate) inhibits fusion between phospholipid vesicles. Proc Natl Acad Sci USA. 1991. 88:737–40.
- ↑ Yano I et al. Isolation of mycolic acid-containing glycolipids in Nocardia rubra and their granuloma forming activity in mice. J Pharmacobiodyn. 1987. 10: 113–23.
- ↑ Beaman BL et al. Role of superoxide dismutase and catalase as determinants of pathogenicity of Nocardia asteroides: importance in resistance to microbicidal activities of human polymorphonuclear neutrophils. Infect Immun. 1985. 47:135–41.
- ↑ Michel G, Bordet C. Cell walls of Nocardiae, in Goodfellow M, Brownell GH, Serrano JA (eds): The Biology of Nocardiae. London, Academic Press, 1976, pp 141–59.
- ↑ Oarada M, Ito E, Milkami Y. Fatty degeneration of the liver induced by HS-6 toxin produced by Nocardia Otitidiscaviarum from human cutaneous nocardiosis. Agric Biol Chem 1990. 54:2737–9.
- ↑ Jump up to: 16.0 16.1 16.2 16.3 16.4 16.5 16.6 Srinivasan M, Sharma S: Nocardia keratitis as a cause of corneal ulcer. Arch Ophthalmol. 1987.105:464.
- ↑ Jump up to: 17.0 17.1 Hirst LW, Merz WG, Green WR. Nocardia asteroides corneal ulcer.. Am J Ophthalmol. 1982. 94:123–4.
- ↑ Jump up to: 18.0 18.1 Tseng SH, Chen JJ, Hu FR. Nocardia brasiliensis keratitis successfully treated with therapeutic lamellar keratectomy. Cornea. 1999. 15:165–7.
- ↑ Yap EY et al. Nocardia choroidal abscess in a patient with systemic lupus erythematosus. Aust NZ J Ophthalmol. 1998. 26:337–8.
- ↑ Jampol LM, Strauch BS, Albert DM. Intraocular nocardiosis. Am J Ophthalmol. 1973. 76:568–73.
- ↑ Davitt B, Gehrs K, Bowers T. Endogenous Nocardia endophthalmitis. Retina. 1998. 18:71–3.
- ↑ Sher NA, Hill CW, Eifrig DE. Bilateral intraocular Nocardia asteroides infection. Arch Ophthalmol. 1977. 95:1415–8.
- ↑ Meyer SL, Font RL, Shaver RP: Intraocular nocardiosis. Report of three cases. Arch Ophthalmol.1970. 83:536–41.
- ↑ Jump up to: 24.0 24.1 Parsons MR, Holland EJ, Agapitos PJ. Nocardia asteroides keratitis associated with extended-wear soft contact lenses. Can J Ophthalmol. 1989. 24:120–2.
- ↑ Jump up to: 25.0 25.1 Jones DB, Liesegang TJ, Robinson NM. Laboratory diagnosis of ocular infections. Cumitech-13, American Society for Microbiology, Washington DC, 1981.
- ↑ Jump up to: 26.0 26.1 Berd D. Laboratory identification of clinically important aerobic actinomycetes. Appl Microbiol 1973.25:665–81.
- ↑ Baum J, Fedukowicz HB, Jordan A. A survey of Moraxella corneal ulcers in a derelict population. Am J Ophthalmol. 1980. 90:476–80.
- ↑ Heidemann DG et al. Branhamella catarrhalis keratitis. Am J Ophthalmol. 1987. 103:576–81.
- ↑ Marioneaux et al. Moraxella keratitis. Cornea. 1991. 10:21–4.
- ↑ Broadway DC et al. Mycobacterium chelonei keratitis: a case report and review of previously reported cases. Eye.1994. 8:134–42.
- ↑ Mirate DJ et al. Mycobacterium chelonei keratitis: a case report. Br J Ophthalmol. 1983. 67:324–6.
- ↑ Robin JB et al. Mycobacterium chelonei keratitis after radial keratotomy. Am J Ophthalmol. 1986. 102:72–9.
- ↑ Jump up to: 33.0 33.1 Reddy AK, Garg P,Kaur I. Speciation and susceptibility of Nocardia isolated from ocular Infections. Clin Microbiol Infect 2010; 16: 1168–1171.
- ↑ Lalitha P et al. Nocardia keratitis: species, drug sensitivities, and clinical correlation.Cornea 2007; 26: 255–259.
- ↑ Gulpczynski Y et al. Determination of antimicrobial susceptibility patterns of Nocardia species from clinical specimens by Etest. Clin Microbiol Infect 2006; 12: 905–912.
- ↑ Garg P. Fungal, Mycobacterial, and Nocardia infections and the eye:an update. Eye (Lond). 2012. Feb; 26(2): 245-251.
- ↑ Johansson B, Fagerholm P, Petranyi G, Claesson Armitage M, Lagali N. Diagnostic and therapeutic challenges in a case of amikacin-resistant Nocardia keratitis. Acta Ophthalmol. Feb 2017;95(1):103-105. doi:10.1111/aos.13182
- ↑ Chen CJ: Nocardia asteroides endophthalmitis. Ophthalmic Surg 14:502–5, 1983
- ↑ Donnenfeld ED et al.Treatment of Nocardia keratitis with topical trimethoprim-sulfamethoxazole. Am J Ophthalmol. 1985. 99:601–2.
- ↑ Huang AJW, Plugfelder SC: Nocardial and actinomycotic keratitis, in Pepose JS, Holland GN, Wilhelmus KR (eds): Ocular Infection and Immunity. St.Louis, Mosby, 1996, pp 1043–7.
- ↑ Schardt WM, Unsworth AC, Hayes CV. Corneal ulcer due to Nocardia asteroides. Am J Ophthalmol. 1956.42:303–6.