Neuroblastoma
All content on Eyewiki is protected by copyright law and the Terms of Service. This content may not be reproduced, copied, or put into any artificial intelligence program, including large language and generative AI models, without permission from the Academy.
Neuroblastoma is a tumor of neural crest origin, and primarily affects children. It is the most common extra-cranial solid tumor in children. It frequently metastasizes to the orbit, and ocular signs and symptoms may be the first presentation of the tumor.
For educational videos access https://www.aao.org/disease-review/opsoclonus-myoclonus-syndrome
Disease Entity
Disease
Neuroblastoma, Malignant neoplasms adrenal gland (ICD-9 194.0), malignant neoplasm of orbit (ICD-9 190.1), secondary malignant neoplasm of other specified sites (ICD-9 198.89).
History
In 1864, German physician Rudolf Virchow was the first to describe an abdominal tumor in a child as a "glioma". Felix Marchand, a German pathologist, went on to describe characteristics of the tumors from the adrenal medulla and the sympathetic nervous system in 1891. In 1901, William Pepper described a neuroblastoma stage 4S presentation in infants that was metastatic to the liver. In 1910, James Homer Wright described circular clumps of cells in bone marrow (now named “Homer-Wright pseudorosettes” and advanced the understanding that the tumor originated from primitive neural cells and could metastasize to bone.[1]
Neuroblastoma is a complex tumor and varies from metastatic potential to spontaneous regression.
Etiology
The etiology and causative factors of neuroblastoma are currently not well understood. Neuroblastoma is a feature of neurofibromatosis type 1 and of Beckwith-Wiedemann syndrome. A small percentage of neuroblastomas are inherited in an autosomal dominant pattern. These familial forms, which account for about 1-2% of all cases, have been linked to germline mutations in the anaplastic lymphoma kinase gene (ALK),[2] aberrations in KIF1B and Phox2b,[3] amplification of the MYCN oncogene,[4] and copy-number variation within the NBPF10 gene, which results in deletions of chromosome arms 1p and 11q.[5] The vast majority of cases of neuroblastoma are sporadic with no apparent risk factors or genetically inherited predisposition[6] Chromosome 6p22 genetic variation is associated to sporadic neuroblastoma[7] .
Risk Factors
Neuroblastomas can occur both sporadically and through an inherited mutation. Germline mutations in the anaplastic lymphoma kinase gene (ALK),[8] aberrations in Phox2b[9] and deletions of chromosome arms 1p and 11q[10] are risk factors for familial neuroblastomas. Amplification of the MYCN oncogene is a common finding,[11] and shows a bimodal distribution of amplification: either 3- to 10-fold, or 100- to 300-fold. This amplification has been linked to more aggressive forms of neuroblastoma.[12] Duplicated segments of the LMO1 gene have also been identified as a risk factor for developing an aggressive form of neuroblastoma.[13] These gene mutations have been known to cause changes that affect cell regulation and promote cell growth and replication, creating what is known as a “tumor microenvironment”. Targeting of this microenvironment has been of interest as a potential therapy for neuroblastomas[14].
Since neuroblastoma is so prevalent among infants, there have been numerous retrospective case report cohorts investigating pre- and perinatal risk factors. Risk factors were stratified based on younger and older infants. Among younger infants, high birth weight, heavier maternal gestational weight gain, maternal hypertension, older maternal age, ultrasound, and respiratory distress are suggested to increase the risk of neuroblastoma. Among older infants, low birth weight was associated with increased risk while greater maternal gestational weight gain was protective. In the oldest age group, first born status, primary cesarean delivery, prolonged labor and premature rupture of the membranes were associated with increased risk.[15] Maternal ingestion of diuretics and other antihypertensives is linked with infant neuroblastoma, and is linked less strongly with maternal ingestion of vitamin, folate and iron supplementation. No associations were seen with pain relievers, antinauseants or cold medications.[16] Additionally, low (below or equal to 7) 1-min Apgar scores, maternal anemia during pregnancy,[17] maternal history of one fetal loss,[18] and use of temporary hair dye in the month before and/or during pregnancy[19] have been associated with an increased risk of neuroblastoma.
There are no known separate risk factors for orbital metastasis from neuroblastomas.
Pathophysiology
Neuroblastoma is derived from precursor cells of the sympathetic nervous system, which require a cohort of transcription factors to properly differentiate into mature sympathetic neurons. PHOX2b is one such transcription factor, and a mutation in the gene that codes for PHOX2b prevents the differentiation into mature neurons. ALK results in decreased proliferation of sympathetic neurons and is another mechanism that results in immature sympathetic neurons. Neuroblastoma is characterized by defective catecholamine synthesis, resulting in accumulation and excretion of the catecholamine metabolites homovanillic acid (HVA) and vanillylmandelic acid (VMA). [20] Pathophysiology of the paraneoplastic symptoms (opsoclonus- myoclonus and hypertension) is thought to be from abnormal antibodies directed against neuronal RNA. [21]
In the two rare cases of vision loss without direct compression of optic nerve or orbital metastases,[22] [23] the cause of the papilledema and vision loss were thought to be from a similar mechanism that causes opsoclonus: an immune response to neuroblastoma cross reacting with tissue. Other postulated theories include toxic metabolites from cancer or from chemotherapy causing axoplasmic transport stasis.[24]
Spontaneous regression mechanisms may include hypermethylation of subtelomeric DNA, apoptosis, deprivation of Nerve Growth Factor (NGF) deprivation and immune response.
As most cases of neuroblastoma are sporadic or genetic, rather than environmental, there are no primary prevention measures.
A thorough history should be obtained of both pediatric and adult patients. Patients and parents of pediatric patients should be asked when they first noticed changes around the eyelids or eyes, and if these changes occurred suddenly or gradually. Patients should be asked if they have double vision or pain with moving their eyes around. Patients should be asked systemic problems such as diarrhea, abdominal pain, confusion, musculoskeletal pain, numbness/ tingling, and fatigue.
Diagnosis
Physical examination
A thorough ocular exam should be performed any time there is suspicion of a neuroblastoma metastasis, including external examination (eyelids), slit lamp exam, ocular motility (opsoclonus), pupils, dilated fundus exam and examination of the nerve. Any ptosis should be measured and periorbital ecchymosis and anisocoria documented. If there is suspicion of neuroblastoma, the patient should be referred to an oncologist for further work up. A full physical exam including chest auscultation, liver and abdominal palpation, and lymph node palpation should be performed, in addition to orbital, chest and abdominal imaging. The most common site of the primary tumor is within the abdomen (65%), and other common sites include the neck, chest and pelvis.[25]
Signs
Ophthalmic Signs
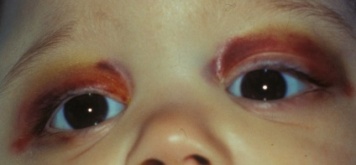
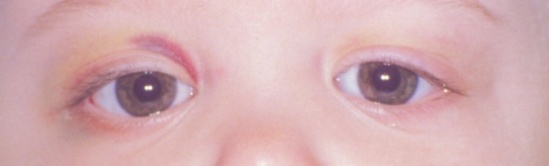
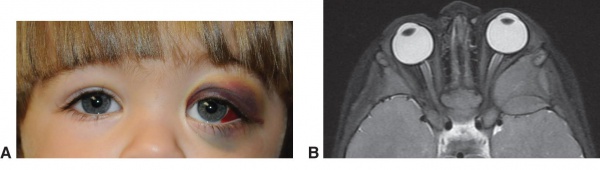
The most common clinical presentation of orbital neuroblastoma metastases in patients less than two years old is unilateral or bilateral periorbital or eyelid ecchymosis, typically called “raccoon eyes” (Figure 1).[26] This can often be confused for traumatic injury (mainly non-accidental) because of the deposition of blood in the eyelids. Less common clinical findings include proptosis,[27] periorbital swelling,[28] periorbital hemorrhage,[29] strabismus, restricted ocular motility,[30] ipsilateral Horner syndrome[31], ptosis,[32] iris heterochromia, opsoclonus/ myoclonus syndrome,[33] convergent strabismus, optic nerve atrophy or edema, and blindness.[34]
Figure 1a. "Raccoon Eyes" upon presentation
Figure 1b. After chemotherapy
A Horner’s syndrome (miotic pupil, ptotic eyelid, and anhidrosis) may result from a primary thoracic neuroblastoma involving the sympathetic chain[31]. Iris heterochromia is typically caused by congenital neuroblastoma of the cervical ganglion resulting in Horner’s. A recent study by Shabat et al, showed that from 135 patients newly diagnosed with neuroblastoma, none of those was it related with Horner's syndrome. Only one patient who had Horner syndrome the tumor had been identified prenatally and confirmed by imaging postnatally. They concluded that, Horner syndrome may not be as correlated as previously thought and that in pediatric patients, the "full investigation" could be considered in only patients with other systemic signs and symptoms.[35]
Opsoclonus (or saccadomania) is characterized by rapid, multidirectional saccadic eye movements with a high frequency (10-15 Hz) and large amplitude. In addition to neuroblastoma, other tumors of neural crest origin may cause this via a paraneoplastic etiology. Neurological deficits may remain even after treatment. Opsoclonus may exist individual or coexist with myoclonus (opsoclonus-myoclonus syndrome). Opsoclonus is certainly not specific for neuroblastoma as it may be seen as a transient finding in normal infants and may occur, rarely, in some patients without associated neurological disease. Neurological signs may persist despite treatment. It is important to note that many patients that present with Opsoclonus-Myoclonus syndrome (48%) may have an occult neuroblastoma. Additionally, some “idiopathic” cases of Opsoclonus-Myoclonus syndrome may really be related to paraneoplastic syndrome from a regressed Neuroblastoma.
*An educational video of Opsoclonus can be seen "here."
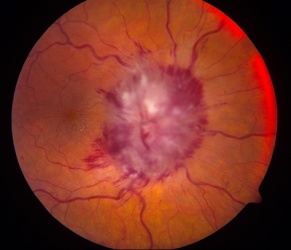
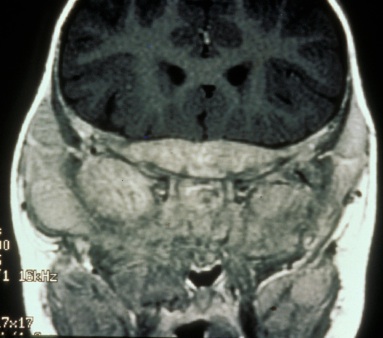
Visual loss at presentation is uncommon, and is usually due to compression or direct invasion of the optic nerves or chiasm, or orbital metastases. This may lead to florid papilledema (Figure 2). Other causes of vision loss include carcinomatous meningitis, infectious meningitis, intrusion into vitreous causing choroidal ectasia, and metastases to the iris. There have been two reports of visual loss in the setting of increased intracranial pressure in the absence of all above features, one in a child[36] and one in an adult.[37]
Figure 2. Frissen Grade V Papilledema from neuroblastoma orbital metastases
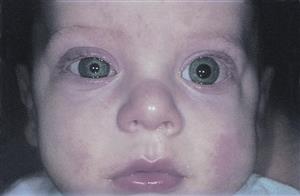
Here is an example of a young patient with unilateral metastatic orbital neuroblastoma:
Because prognosis is best with early detection of the tumor, when it is still resectable, it is important to look for the signs/symptoms of initial presentation.
A retrospective study of 405 patients with Neurobastoma[38] suggested that from all the first-presenting signs, the 3-year survival rate was best in opsoclonusmyoclonus children, followed by unilateral Horners syndrome and Proptosis/periorbital ecchymosis (3-year survival of 100%,78.6% and 11.2% respectively)[38].
Systemic Signs
Systemic manifestations include abdominal pain, abdominal distention, respiratory compromise, bone pain, paralysis, severe, watery diarrhea, confusion (from myoclonic encephalopathy), fever, and muscle weakness (from myasthenia gravis).
Symptoms
Symptoms initially can be very vague, making diagnosis difficult. These include fatigue, confusion, and weakness. Fatigue can occur if there is anemia from bone marrow involvement; confusion may occur if there is encephalopathy, weakness may occur from myasthenia gravis or from transverse myelitis if there is spinal cord compression. Abdominal pain or distention may occur from abdominal tumor and difficulty breathing may occur from respiratory tumor spread.
In a study by Graef et al., from 523 patients with neuroblastoma, 3% presented with only ocular findings which were generally associated to a poor prognosis, while female gender, young age and opsoclonus were associated to a favorable one.[39]
Clinical diagnosis
Clinical diagnosis should be made on the physical exam, and the systemic and ophthalmic signs and symptoms described above.
Diagnostic procedures
A thorough exam and clinical history is recommended in order to rule out non-accidental trauma.
Bone marrow biopsies, with bilateral posterior iliac crest aspirates and triphine (core) bone samples, should be obtained to exclude bone marrow involvement. These must be at least 1cm of marrow in order to be considered an adequate sample.[40]
In some patients with paraneoplastic- induced opsoclonus, abnormal antibodies directed against neuronal RNA may be detected in the CSF. Specifically in the case of children with neuroblastoma, serologic or CSF assay for anti-Hu antibody (also known as ANNA-1) can be present and helpful in diagnosis.[41] However, lumbar puncture (LP) is generally avoided at diagnosis unless central nervous system (CNS) metastasis is known. There have been reports that LP may be associated with an increased subsequent development of CNS metastases.[42]
Finally, palpable lymph nodes should be biopsied and histologically confirmed for staging.[43]
Imaging
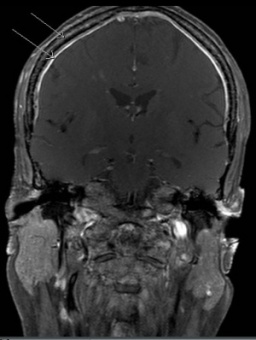
Orbital imaging must be performed on any patient who presents with ocular signs. Orbital metastases tend to occur in the posterolateral orbital wall.[44] Bone destruction may occur, specifically in the lateral orbital wall or sphenoid marrow. On Computed Tomography (CT), metastases can appear as either circumscribed or ill defined, are increased in attenuation compared to muscle, can contain small calcifications, and can invade adjacent structures, including intracranially.[45] On Magnetic Resonance Imaging (MRI), neuroblastoma metastases appear low signal on T1WI, heterogeneous on T2WI due to hemorrhage or necrosis, and heterogeneously enhance post contrast. Tumor extension intracranially and into the adjacent soft tissues may be seen more readily on MR over CT (Figure 3a and b).[46]
Positron Emission Tomography (PET) can be used for diagnosis, staging, and monitoring therapy with high sensitivity and specificity.[47]
In a child with a Horner syndrome without a history of trauma or surgery, the presence of a neuroblastoma affecting the sympathetic chain in the chest must be considered with imaging studies of the brain, neck, and chest.[48]
During neuroblastoma workup, the recommended image is head, neck, thorax, abdomen, and pelvis MRI with thin cuts. CT is not preferred due to radiation exposure but could be used. [49]
Figure 3a. MRI showing tumor along the skull base Figure 3b. Dural and calvarial enhancement from metastases
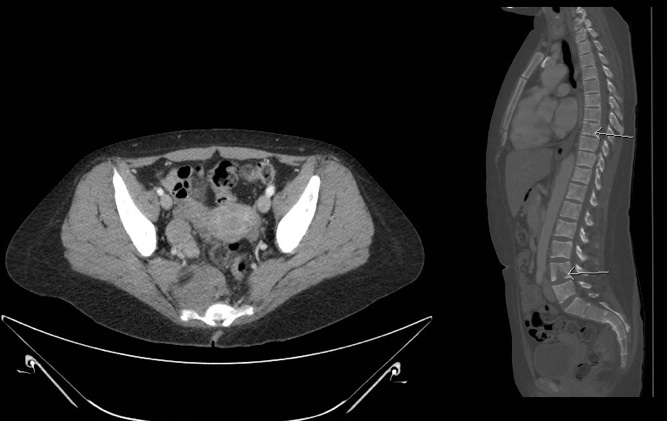
The abdomen and liver should be assessed by CT scan and/or MRI (Figure 4a). A chest CT should be performed if extension of abdominal disease or pulmonary metastasis is suspected. A MIBG scan should be performed to assess bone involvement. If the MIBG scan results are negative, a technetium 99 scan should be performed. Imaging with 123 I-MIBG is optimal for identifying soft tissue and bony metastases and is better than a PET/CT in prospective comparison.[50] If the primary tumor does not take up MIBG, additional imaging of isolated or equivocal positive lesions should be performed.[51] MRI of the spine should be performed to evaluate paraspinal tumors that may extend through neural foramina and compress the spinal cord (Figure 4b).
Figure 4a. Figure 4b.
56mm soft tissue mass in the R pelvis anterior to sacrum, to the Sagittal image of bony lytic lesions
right of the rectum. Soft tissue going into or out of the S4 neural compressing neural foramen.
foramen.
Laboratory test
Once a diagnosis of neuroblastoma is made, a thorough evaluation for metastatic involvement outside the orbit should be performed prior to therapy initiation. Blood work should be done to evaluate for pancytopenia, serum creatinine, liver function tests (alanine aminotransferase, aspartate aminotransferase, total bilirubin, alkaline phosphatase, total protein, albumin, prothrombin time/ activated prothrombin time, electrolytes, calcium, magnesium, phosphorus, uric acid, serum lactate dehydrogenase, ferritin, thyroid-stimulating hormone, T4, and immunoglobulin (Ig)G levels.
Urinary and serum catecholamines including homovanillic acid (HVA) and vanillylmandelic acid (VMA) have previously been reported to be elevated in approximately 90-95% of cases [52] [53] but newer studies have shown that VMA or HVA elevations are not always present and thus are not as sensitive as once thought. [54] [55] A urinary catecholamine level is considered to be elevated if it is 3 standard deviations higher than the age-related reference range levels. It was thought that screening infants for urinary VMA or HVA would be beneficial in detecting early cases of neuroblastoma as the catecholamine levels can be elevated in pre-clinical neuroblastoma, but trials in Canada, Japan, and Germany showed no reduction in deaths, but rather showed increase in unnecessary surgery and chemotherapy in patients whose tumors would have regressed on their own. [56]
CSF studies may show expansion of the B cell population , pleocytosis, or be completely normal. Flow cytology may be helpful.
Histopathology
Neuroblastoma is characterized by immature, only slightly differentiated nerve cells of embryonic type, that is, neuroblasts; typical cells are relatively small (10-15 mcm in diameter) with disproportionately large, darkly staining, vesicular nuclei and scant, palely acidophilic cytoplasm; they may be arranged in sheets, irregular clumps, or cordlike groups, as well as occurring individually and in pseudorosettes (with nuclei arranged peripherally about the centrally directed cytoplasmic processes); ordinarily, the stroma is sparse, and foci of necrosis and hemorrhage are not unusual. Tumors are characterized as favorable and unfavorable by the International Neuroblastoma Pathology Committee (also called the Shimada system).[57]
The mechanism by which neuroblastoma cells grow has been a subject of hypothesis and in a review by Tsubota et al, few mechanisms including precursor in sympathoadrenal lineage of neural crest and Schwann cell and initiation including upregulation by N-Myc and downregulation by polycomb repressive complex 2. [58]
Clinical features of adults vs. children
When comparing the clinical features of adults versus children, there is less of a urine catecholamine elevation (only 40-57% adults had an elevation as compared with 95% of children), N-myc amplification is rare, bone marrow involvement is less frequent, and there is a greater frequency of metastases to unusual places like lung or brain, as compared with children where bone marrow involvement is more common.
Differential diagnosis
Differential diagnosis includes esthesioneuroblastoma, ganglioneuroblastoma, pheochromocytoma, rhabdomyosarcoma, and Wilms’ tumor. Histologically, neuroblastoma appears similar to rhabdomyosarcoma, Ewing’s sarcoma, lymphoma, and Wilm’s tumor.
Management
General treatment
Treatment of metastatic neuroblastoma is stratified based on clinical features (age at presentation, staging) and specific tumor biological markers (see below). Treatment consists of a combination of chemotherapy, radiation therapy, surgery, myeloablative therapy with stem cell transplant, immunotherapy such as anti-GD2 monoclonal antibody therapy,[59] and differentiation therapy such as isotretinoin. Also, tumor cell vaccination and immunotherapy treatments are being tested in phase I/II clinical trials. Generally, chemotherapy is first given to shrink tumor size prior to surgery. Chemotherapy renders tumors more fibrous, less vascular, and more amenable to a planned excision. A multitude of chemotherapy agents used in various combinations and regimens include but are not limited to Carboplatin, Etoposide, Vincristin, Cyclophosphamide and Doxorubicin.[60]
There are current studies evaluating possible treatments to target the specific genetic mutations in neuroblastoma particularly the molecular therapies targeting ALK signaling, MDM2, PI3K/Akt/mTOR and RAS‐MAPK pathways, and some epigenetic regulators. Few examples include:
- Small molecule inhibitors of TrkB, VEGF, LIN28B, survivin, and Phox2b
- Inhibition of MYCN by means of inhibiting:
- MYCN/MAX heterodimerization
- Aurora A kinase
- Bromodomain and extra‐terminal domain (BET) protein
- Ornithine decarboxylase 1 (ODC1)
These therapies however, are pre-clinical models.[61]
General treatment for neurological symptoms, such as opsoclonus, should be directed to symptomatology, usually with immunosuppression(corticostarois) or ACTH-corticotropin, regardless of tumor removal, since there are patients that may have no-tumor and still present with neurological signs and symptoms.
Therapy for vision threatening tumors
Traditionally, therapy for impending neuroblastoma-associated visual loss includes radiation and/or high dose steroids to decompress the optic nerve(s) from an optic neuropathy. High dose steroids have been used empirically to decrease inflammation, lower the perineural pressure, and prevent neuronal injury[62]. It has been shown that cortisol receptors can be downregulated by a specific microRNA cluster which is upregulated by the MYCN gene mutation found in neuroblastoma[63]. This presents another possible mechanism of just how high-dose corticosteroids can be effective in treatment of vision threatening tumors.
Surgery
Surgery is undertaken when removal of an orbital metastases can be performed safely without destruction of surrounding tissues. Surgery of the primary site is undertaken similarly when adjacent structures and tissues will not be damaged, and plays an important part of management. Presurgical chemotherapy is frequently undertaken to shrink the tumor. Surgery is typically reserved for low and intermediate risk patients in whom a complete resection is possible. Neurosurgical decompression is recommended for patients with intraspinal neuroblastoma and rapid neurological deterioration during chemotherapy. General surgical complications include bleeding, infection, damage to adjacent structures, and need for further surgery. Specific surgical complications include nephrectomies (intraoperatively from organ extension), Horner syndrome, pleural effusions, abscess, hematuria, ileus, and tumor rupture. [64]
Follow up
Urine catecholamines are used as sensitive and specific tumor markers to assess treatment response and post-treatment surveillance.[65] The biologic dissimilarity between neuroblastomas in children as compared with adults has also been found to be useful for monitoring disease status. Additionally, MIBG, when radio-ionated with I-131 or I-121, is a very good radiopharmaceutical for diagnosis and monitoring of the disease. Routine imaging of primary tumors is helpful to evaluate for recurrence. Patients who undergo any form of eye sparing treatment (external beam radiation, chemoreduction) need frequent follow-up examinations. Patients who have been treated with radiation are at higher risk for secondary tumors in the field of treatment. Long term follow-up of all neuroblastoma patients is mandatory. Interest has developed recently regarding the utility of peripheral blood tests and “liquid biopsies” to aid in the prognosis and diagnosis of neuroblastoma, allowing for real-time non-invasive monitoring of disease progression and response to treatment[66]. Specific microRNAs have been identified in peripheral blood of patients with neuroblastoma, as well as mutations in the ALK gene in cell-free DNA of patients[67].
Prognosis
Neuroblastoma is distinguished for its varied and broad spectrum of clinical behavior, with some tumors regressing and some metastasizing despite aggressive treatment.[68] Two pretreatment risk stratification systems exist - The International Neuroblastoma Risk Group (INRG) classification system and the Children’s Oncology Group (COG) Neuroblastoma Risk Grouping. COG risk stratifies based on age, INSS stage, and tumor biology, which includes histopathology classification, MYCN amplification and tumor DNA index.[69] [70]
Age is a major prognostic factor. Survival rate is directly related to age at diagnosed. The prognosis is better in infants (3-year: 86%, 5-year 84.6%) as compared with children over one year of age (45% 5% year survival) and adults over 20 years of age (3-year survival rate 45.9%, 5-year survival rate 36.3%). There exists a worse long-term prognosis in adults regardless of stage or site. This may be due to tumor biology, more virulent clinical course, or possibly due to the fact that adults are less sensitive or have poor tolerance to pediatric chemotherapy regimens.
Low risk tumors are best defined by the absence of MYCN gene amplification and any structural genetic abnormalities (such as either 11q and/or 1p aberrations and/or 17q gain).[71] [72] Opsoclonus is generally a good prognostic factor.[73] MYCN amplification is considered an unfavorable prognostic factor by all groups.[74] Stage I, II, or IVa disease, younger age, increase of TRK- protein (product of proto-oncogne TRK) are good prognostic indicators. 11q aberration was associated with worse outcome in patients with L2 or MS tumors that lack MYCN amplification.[75] Tumor cell DNA hyperploidy was found to be a have a favorable prognosis in children younger than 18 months of age with stage 4 disease and no amplification of MYCN[76] [77]. Recent studies aimed to determine the significance of metastatic sites and their effects on prognosis. A study by Harreld, et al. showed that orbital metastatic disease is associated with decreased 5-year survival (p=0.0323) and decreased overall survival (p-0.0288) in patients with stage M neuroblastoma[78].
Acknowledgements
Most Images are Courtesy of Vanderbilt Eye Institute, Yuna Rapoport, MD and Louise Mawn, MD
Additional Resources
- http://www.cancer.gov/cancertopics/pdq/treatment/neuroblastoma/HealthProfessional/page3
- International Neuroblastoma Staging System
- Children’s Oncology Group Neuroblastoma Risk Grouping
- SEER Database
References
- ↑ Rothernberg AB, et al. Neuroblastoma- remembering the three physicians who described it a century ago: James Horner Wright, William Pepper, and Robert Hutchinson. Pediatr Radiol. 2009; 39(2):155-60.
- ↑ Mosse, et al. Identification of ALK as the Major Familial Neuroblastoma Predisposition Gene. Nature. 2008; 455 (7215): 930-935.
- ↑ Reiff T, et al. Neuroblastoma phox2b variants stimulate proliferation and dedifferentiation of immature sympathetic neurons. J Neurosci. 2010; 30:905–915.
- ↑ Schwab M, et al. Chromosome localization in normal human cells and neuroblastomas of a gene related to c-myc. Nature. 1984;308:288–91.
- ↑ Vandesompele J, et al. Unequivocal delineation of clinicogenetic subgroups and development of a new model for improved outcome prediction in neuroblastoma. J Clin Oncol. 2005; 23:2280-2299.
- ↑ Swift CC, Eklund MJ, Kraveka JM, Alazraki AL. Updates in Diagnosis, Management, and Treatment of Neuroblastoma. Radiographics. 2018;38(2):566-580
- ↑ Aygun N. Biological and Genetic Features of Neuroblastoma and Their Clinical Importance. Curr Pediatr Rev. 2018;14(2):73-90. doi:10.2174/1573396314666180129101627
- ↑ Mosse, et al. Identification of ALK as the Major Familial Neuroblastoma Predisposition Gene. Nature. 2008; 455 (7215): 930-935.
- ↑ Reiff T, et al. Neuroblastoma phox2b variants stimulate proliferation and dedifferentiation of immature sympathetic neurons. J Neurosci. 2010;30:905–915.
- ↑ Vandesompele J, Baudis M, De Preter K, et al: Unequivocal delineation of clinicogenetic subgroups and development of a new model for improved outcome prediction in neuroblastoma. J Clin Oncol 2005; 23:2280-2299.
- ↑ Schwab M, et al. Chromosome localization in normal human cells and neuroblastomas of a gene related to c-myc. Nature. 1984;308:288–91.
- ↑ Brodeur, G, et al. Amplification of N-myc in untreated human neuroblastomas correlates with advanced disease stage. Science. 1984; 224 (4653): 1121–1124.
- ↑ Brodeur, G, et al. Amplification of N-myc in untreated human neuroblastomas correlates with advanced disease stage. Science. 1984; 224 (4653): 1121–1124.
- ↑ Borriello L, Seeger RC, Asgharzadeh S, Declerck YA. More than the genes, the tumor microenvironment in neuroblastoma. Cancer Lett. 2016;380(1):304-14
- ↑ McLaughlin CC, et al. Perinatal risk factors for neuroblastoma. Cancer Causes Control. 2009;20(3):289-301.
- ↑ Schüz J, et al. Medication use during pregnancy and the risk of childhood cancer in the offspring. Eur J Pediatr. 2007;(5):433-41.
- ↑ Bluhm E, et al. Int J Cancer. Prenatal and perinatal risk factors for neuroblastoma. 2008;123(12):2885-90.
- ↑ Johnson KJ, et al. Int J Cancer. Perinatal characteristics and risk of neuroblastoma. 2008;123(5):1166-72.
- ↑ McCall, EE. Maternal hair dye use and risk of neuroblastoma in offspring. Cancer Causes Control. 2005; 16(6):743-8.
- ↑ Castleberry RP. Biology and Treatment of Neuroblastoma. Pediatr Clin North Am. 1997; 44:919–937.
- ↑ Pelosof, LC et al. Paraneoplastic Syndromes: An Approach to Diagnosis and Treatment. Mayo Clin Proc. 2010; 85(9): 838-854.
- ↑ Scott JX, et al. Paraneoplastic Papilloedema in a Child with Neuroblastoma. Indian J Cancer. 2005; 42(2):102-3.
- ↑ Kennedy MJ, et al. Paraneoplastic papilloedema in neuroblastoma. Postgrad Med J. 1987; 63:873:6.
- ↑ Kennedy MJ, et al. Paraneoplastic papilloedema in neuroblastoma. Postgrad Med J. 1987; 63:873:6.
- ↑ De Bernardi B, et al. Disseminated neuroblastoma in children older than one year at diagnosis: comparable results with three consecutive high-dose protocols adopted by the Italian Co-Operative Group for Neuroblastoma. J Clin Oncol. 2003; 21:1592–601.
- ↑ E. M. Chung, et al. From the archives of the AFIP pediatric orbit tumors and tumorlike lesions: osseous lesions of the orbit. Radiographics. 2008; 28 (4):1193–1214.
- ↑ Smith S, et al. Ocular Manifestations, and Survival in Children with Neuroblastoma: A Population-Based Study. Am J Ophthalmol. 2010;149(4):677–682.
- ↑ Ahmed S, et al. Neuroblastoma with orbital metastasis: Ophthalmic presentation and role of ophthalmologists. Eye. 2006; 20:466–470.
- ↑ Belgaumi AF, et al. Blindness in Children with Neuroblastoma. Cancer. 1997;80(10):1997–2004.
- ↑ Alfano JE. Ophthalmological Aspects of Neuroblastomatosis: a Study of 53 Verified Cases. Trans Am Acad Ophthalmol Otalaryngol. 1968;72:830–848.
- ↑ Jump up to: 31.0 31.1 Spors B, Seemann J, Homer N, Fay A. Lymphatic malformation with acquired Horner syndrome in an infant. Journal of NeuroInterventional Surgery. 2018;10:e2.
- ↑ Alfano JE. Ophthalmological Aspects of Neuroblastomatosis: a Study of 53 Verified Cases. Trans Am Acad Ophthalmol Otalaryngol. 1968;72:830–848.
- ↑ Farrelly C, et a. Occult Neuroblastoma Presenting with Opsomyoclonus: Utility of Computed Comography. Am J Roentgenology. 1984;142:807–810.
- ↑ Shubert EE, et al. Metastatic neuroblastoma causing bilateral blindness. Canad J Ophthal. 1969;4:100–103.
- ↑ Ben Shabat A, Ash S, Luckman J, Toledano H, Goldenberg-Cohen N. Likelihood of Diagnosing Neuroblastoma in Isolated Horner Syndrome. J Neuroophthalmol. 2019;39(3):308-312. doi:10.1097/WNO.0000000000000764
- ↑ Scott JX, et al. Paraneoplastic Papilloedema in a Child with Neuroblastoma. Indian J Cancer. 2005; 42(2):102-3.
- ↑ De Bernardi B, et al. Disseminated neuroblastoma in children older than one year at diagnosis: comparable results with three consecutive high-dose protocols adopted by the Italian Co-Operative Group for Neuroblastoma. J Clin Oncol. 2003;21:1592–601.
- ↑ Jump up to: 38.0 38.1 Musarella MA, Chan HS, DeBoer G, Gallie BL. Ocular involvement in neuroblastoma: prognostic implications. Ophthalmology. 1984;91(8):936-940. doi:10.1016/s0161-6420(84)34211-7
- ↑ Graef S, Irwin MS, Wan MJ. Incidence and Prognostic Role of the Ocular Manifestations of Neuroblastoma in Children. Am J Ophthalmol. 2020 May;213:145-152. doi: 10.1016/j.ajo.2020.01.023. Epub 2020 Mar 11. PMID: 32006485.
- ↑ Russell HV, et al.: The role of bone marrow evaluation in the staging of patients with otherwise localized, low-risk neuroblastoma. Pediatr Blood Cancer. 2005; 45 (7): 916-9.
- ↑ Lennon VA. Paraneoplastic autoantibodies: the case for a descriptive generic nomenclature. Neurology. 1994;44(12);2236-2240.
- ↑ Kramer K, et al. Neuroblastoma metastatic to the central nervous system. The Memorial Sloan-kettering Cancer Center Experience and A Literature Review. Cancer. 2001; 91 (8): 1510-9.
- ↑ Brodeur GM, et al. Revisions of the international criteria for neuroblastoma diagnosis, staging, and response to treatment. J Clin Oncol. 1993; 11 (8): 1466-77.
- ↑ Rao, et al. A Clinical Update and Radiologic Review of Pediatric Orbital and Ocular Tumors. Journal of Oncology. 2013; vol. 2013, Article ID 975908, 22 pages.
- ↑ E. M. Chung, at al, From the archives of the AFIP pediatric orbit tumors and tumorlike lesions: osseous lesions of the orbit. Radiographics. 2008; 28 (4) pp. 1193–1214.
- ↑ E. M. Chung, at al, From the archives of the AFIP pediatric orbit tumors and tumorlike lesions: osseous lesions of the orbit. Radiographics. 2008; 28 (4) pp. 1193–1214.
- ↑ Pflüger, T, et al. Modern nuclear medicine evaluation of neuroblastoma. The Quarterly Journal of Nuclear Medicine and Molecular Imaging. 2010; 54(4) p. 389–400.
- ↑ Mahoney NR, e al. Pediatric Horner syndrome: etiologies and roles of imaging and urine studies to detect neuroblastoma and other responsible mass lesions. Am J Ophthalmol. 2006;142(4):651-659.
- ↑ James O’Brien J and Siatkowski RM. Opsoclonus-Myoclonus Syndrome. RM. Knights Templar Eye Foundation: Pediatric Ophthalmology Education Center. AAO Feb, 2016. Accessed Aug 26th, 2020 https://www.aao.org/disease-review/opsoclonus-myoclonus-syndrome
- ↑ Papathanasiou ND, et al. 18F-FDG PET/CT and 123I-metaiodobenzylguanidine imaging in high-risk neuroblastoma: diagnostic comparison and survival analysis. J Nucl Med. 2011; 52 (4): 519-25.
- ↑ Taggart DR, et al. Prognostic value of the stage 4S metastatic pattern and tumor biology in patients with metastatic neuroblastoma diagnosed between birth and 18 months of age. J Clin Oncol. 2011; 29 (33): 4358-64.
- ↑ E. M. Chung, et al. From the archives of the AFIP pediatric orbit tumors and tumorlike lesions: osseous lesions of the orbit. Radiographics. 2008; 28 (4):1193–1214.
- ↑ Smith S, et al. Ocular Manifestations, and Survival in Children with Neuroblastoma: A Population-Based Study. Am J Ophthalmol. 2010;149(4):677–682.
- ↑ Ahmed S, et al. Neuroblastoma with orbital metastasis: Ophthalmic presentation and role of ophthalmologists. Eye. 2006; 20:466–470.
- ↑ Woodruff G, et al. Horner’s Syndrome in Children. J Pediatr Ophthalmol Strab. 1988;25:40–44.
- ↑ Tsubono Y, et al. A halt to neuroblastoma screening in Japan. N Engl J Med. 2004; 350 (19): 2010-1.
- ↑ Peuchmaur M, et al. Revision of the International neuroblastoma Pathology Classification: confirmation of favorable and unfavorable prognostic subsets in ganglioneuroblastoma, nodular. Cancer. 2003; 98 (10): 2274-81.
- ↑ Tsubota S, Kadomatsu K. Origin and initiation mechanisms of neuroblastoma. Cell Tissue Res. 2018 May;372(2):211-221. doi: 10.1007/s00441-018-2796-z. Epub 2018 Feb 14. PMID: 29445860.
- ↑ Johnson, et al. Antibody-based immunotherapy in high-risk neuroblastoma. Expert Rev Mol Med. 2007; 9(34): 1-21.
- ↑ Kohler JA, et al. Treatment of children over the age of one year with unresectable localised neuroblastoma without MYCN amplification: Results of the SIOPEN study. Eur J Cancer. 2013 S0959-8049(13)00546-7
- ↑ Zafar A, Wang W, Liu G, et al. Molecular targeting therapies for neuroblastoma: Progress and challenges [published correction appears in Med Res Rev. 2022 Jan;42(1):641]. Med Res Rev. 2021;41(2):961-1021. doi:10.1002/med.21750
- ↑ Belgaumi AE, et al. Blindness in children with neuroblastoma. Cancer. 2004; 80(10): 1997-2004.
- ↑ Ribeiro D, Klarqvist MDR, Westermark UK, et al. Regulation of Nuclear Hormone Receptors by MYCN-Driven miRNAs Impacts Neural Differentiation and Survival in Neuroblastoma Patients. Cell Rep. 2016;16(4):979-993
- ↑ Cañete A, et al. Surgical treatment for neuroblastoma: complications during 15 years' experience. J Pediatr Surg. 1998, 33(10):1526-30.
- ↑ Owens C, et al. Neuroblastoma: the impact of biology and cooperation leading to personalized treatments. Crit Rev Clin Lab Sci. 2012;49(3):85-115.
- ↑ Trigg RM, Shaw JA, Turner SD. Opportunities and challenges of circulating biomarkers in neuroblastoma. Open Biol. 2019;9(5):190056
- ↑ Combaret V, Iacono I, Bellini A, et al. Detection of tumor ALK status in neuroblastoma patients using peripheral blood. Cancer Med. 2015;4(4):540-50
- ↑ Maris JM, et al. Neuroblastoma. Lancet. 2007; 369:2106-2120.
- ↑ Shimada H, et al. Histopathologic prognostic factors in neuroblastic tumors: Definition of subtypes of ganglioneuroblastoma and an age-linked classification of neuroblastomas. J Natl Cancer Inst. 1984; 73:405-416.
- ↑ Shimada H, Ambros IM, Dehner LP, et al: The International Neuroblastoma Pathology Classification (the Shimada System). Cancer 1999; 86:364-372.
- ↑ Vandesompele J, et al. Unequivocal delineation of clinicogenetic subgroups and development of a new model for improved outcome prediction in neuroblastoma. J Clin Oncol. 2005; 23:2280-2299.
- ↑ Schleiermacher G, et al. Chromosomal CGH identifies patients with a higher risk of relapse in neuroblastoma without MYCN amplification. Br J Cancer. 2007; 97:238-246.
- ↑ Rao, et al. A Clinical Update and Radiologic Review of Pediatric Orbital and Ocular Tumors. Journal of Oncology. 2013; 975908; 22 pages.
- ↑ Seeger RC, Brodeur GM, Sather H, et al: Association of multiple copies of the N-myc oncogene with rapid progression of neuroblastomas. N Engl J Med. 1985; 313:1111-1116.
- ↑ Cohn SL, et al. The international Nerubolastoma Risk Group (INRG) Classifciation System: An INRG task Force Report. J of Clin Onc. 2009; 27 (2) 289-297.
- ↑ George RE, et al: Hyperdiploidy plus nonamplified MYCN confers a favorable prognosis in children 12 to 18 months of age with disseminated neuroblastoma: A Pediatric Oncology Group Study. J Clin Oncol. 2005; 23:6466-6473.
- ↑ Look AT, et al. Clinical relevance of tumor cell ploidy and N-myc gene amplification in childhood neuroblastoma: A Pediatric Oncology Group study. J Clin Oncol. 1991; 9:581-591.
- ↑ Morgenstern DA, London WB, Stephens D, et al. Prognostic significance of pattern and burden of metastatic disease in patients with stage 4 neuroblastoma: A study from the International Neuroblastoma Risk Group database. Eur J Cancer. 2016;65:1-10