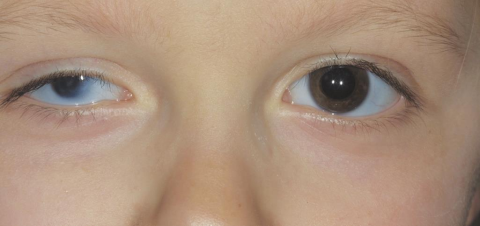
Microphthalmos
All content on Eyewiki is protected by copyright law and the Terms of Service. This content may not be reproduced, copied, or put into any artificial intelligence program, including large language and generative AI models, without permission from the Academy.
Disease Entity
Microphthalmos also called microphthalmia, is a rare developmental disorder of the eye in which one or both eyes are abnormally small. It may occur as an isolated entity but is commonly associated with other ocular or systemic findings as part of a syndrome.[1][2] Although both microphthalmos and nanophthalmos (also called pure or simple microphthalmos) are characterized by a smaller than normal eye, nanophthalmos does not present with anatomical malformations.[2]
Epidemiology
Due to the rarity of this condition, few population studies exist. The estimated birth prevalence of microphthalmia varies greatly and is calculated based off of national congenital anomaly registry studies.[3][4][5][6][7][8][9]Also, some studies describe the prevalence of microphthalmia with other related conditions such as anophthalmia and coloboma since these conditions exist on a phenotypic spectrum. A prevalence as high as 1.5-19/10,000 has been reported.[10] More recently, an estimated range of 0.2-1.7/10,000 has been reported.[9] This suggests there may be geographic heterogenicity in prevalence.
Microphthalmia may be unilateral but is more commonly bilateral. Patients with bilateral disease are more likely to have systemic involvement.[11] It does not present differently according to sex and race and affects 3-12% of blind children.[2][3] [4][5][9]
Etiology
The precise pathogenesis of microphthalmia remains largely unknown. It has been hypothesized that this malformation occurs due to improper development of the optic vesicles, anterior neural tube, or optic pits during early embryonic development or closure of the fetal fissure.[9] It has also been hypothesized that abnormalities of the posterior segment including decreased size of the optic cup, altered proteoglycans in the vitreous, low intraocular pressure, abnormal growth factor production and inadequate production of secondary vitreous may contribute to microphthalmia.[2] Some cases of microphthalmia associated with a cyst may result from failure of invagination of optic vesicle.[8][12][13]
Genetic, chromosomal and environmental factors can contribute to microphthalmia. Most of cases of microphthalmos are sporadic, but autosomal dominant, autosomal recessive and X-linked modes of inheritance have been described. One study cited that there are more than 100 genetic traits associated with microphthalmos and coloboma. Mutations in genes SOX2, OTX2, BMP4, CHD7, GDF6, RARB and SHH present an autosomal dominant inheritance pattern, while mutations in genes PAX6, STRA6, FOXE3, RAX, SMOC1, VSX2 are associated to autosomal recessive mode of inheritance and BCOR, HCCS and NAA10 mutations are associated to X-linked mode of inheritance.[14] Of these, SOX2 and PAX6 have been identified as a major causative genes.[2][4][9][15] It is also commonly seen among patients with chromosomal disorders including trisomy 13 or trisomy 18.[9][14][16]
Environmental risk factors include: maternal age over 40, elevated maternal pre-pregnancy body mass index (BMI), maternal smoking, multiple births, infants of low birth weight, low gestational age, gestational-acquired infections (rubella, toxoplasmosis, varicella, cytomegalovirus, parvovirus B19, influenza virus, and coxsackie A9), maternal vitamin A deficiency, fever, hyperthermia, exposure to X-rays, solvent misuse and exposure to drugs like thalidomide, warfarin and alcohol.[6][12][13][16][17][18][19][20]
Clinical Diagnosis and Evaluation
Ocular Evaluation
Microphthalmia is defined as a small eye with anatomical malformation and whose axial length is two standard deviations below the mean for age, corresponding to an axial length below 21 mm in adult eyes.[10] It is typically diagnosed clinically by inspection and palpation of the eye through the lids. Other useful ocular exam findings include measurements of corneal diameter.
Despite the rarity of this condition, if present, it causes substantial visual impairment. Ocular disorders may affect the anterior segment and/or the posterior segment. These abnormalities include microcornea, corneal opacification, corectopia, ectopia lentis, aniridia, cataract, persistent fetal vasculature and/or retinal dysplasia persistent hyperplastic primary vitreous and retinal dysplasia).[2] It’s also commonly associated with uveal colobomas. Microphthalmia may be associated to an orbital cyst (typically located in the inferior orbit) originated from the optic nerve where it usually communicates with the subarachnoid space and can grow throughout the patient’s life.[2]
Visual acuity will depend on the type of ocular malformations and vision loss is especially seen in patients with posterior microphthalmos. A good visual acuity can be present in eyes with small iris or choroidal colobomas. However, eyes with macular and optic nerve head involvement have poor vision
Microphthalmic eyes are usually highly hypermetropic, but sometimes can be highly myopic if staphyloma formation is present.
Systemic Evaluation
Microphthalmia can be associated with intellectual disability, craniofacial malformations (as cleft lip/palate or microcephaly) and malformations of hands and feet (polydactyly). This ocular disorder can occur in isolation or be syndromic (33-50%). Syndromes associated to Microphthalmia are CHARGE syndrome, Aicardi syndrome, Duker syndrome, Lenz microphthalmia syndrome, Oculo-Dento-Osseous Dysplasia, Fraser Syndrome, Cryptophthalmos syndrome, Cerebro-Oculo-Facial Syndrome, Goltz syndrome, Lowe syndrome, Meckel-Gruber syndrome, Basal cell nevus syndrome of Gorlin-Goltz, Cross syndrome and Microphthalmia with linear skin defects.[21][22]
Since Microphthalmia can be associated to these non-ocular anomalies, physical examination (including dysmorphology examination) is mandatory to determine the presence of distinguishing clinical features. Patients should be evaluated by multidisciplinary teams composed by ophthalmologists, pediatricians, and clinical geneticists. If no syndrome is identified in childhood, further examination after three or four years should be performed as many syndromes become more apparent at this age. [2]
Family history
Microphthalmia can exhibit a hereditary pattern. Therefore, it is crucial to complete eye examination of both parents and to obtain a three-generation family history of eye anomalies, including microphthalmia and coloboma.
Differential Diagnosis
Includes cryptophthalmos, cyclopia, and synophthalmia
Imaging
Fetal orbits are usually detectable by 11-12 weeks of gestation.[22] Although the diagnosis of microphthalmia is often a subjective one and the diagnosis is often made because of concurrent syndromic findings. Prenatally, ultrasound is used to determine the length of the globe in microphthalmic eyes and to make examination of the orbits.[10][22] If clinical suspicion genetic evaluation through amniocentesis or chorionic villus sampling with chromosomal microarray analysis should be offered.
Magnetic resonance imaging (MRI) is extremely useful because there is higher resolution of the structures of interest and no radiation exposure. It can also reveal remnant of ocular tissue, an optic nerve, and extraocular muscles which can help distinguish anophthalmia from severe microphthalmia.[14] If there is an orbital cyst, it produces a homogeneous signal that varies from isointense to hypointense on MRI T1-weighted image, while on T2-weighted image the cyst appears hyperintense and there is no enhancement with gadolinium. [10]
Electrophysiological tests are critical for assessing the severity of visual impairment and help to determine at which level the abnormality is. In cases of severe microphthalmia, a flash visual evoked potential (VEP) will establish if any visual function is present. A pattern VEP will determine the severity of the disease and detect any optic nerve dysfunction while an electroretinogram will identify if there is retinal dysfunction.[10]
Management
Early examination identification is crucial to develop early management plans. As mentioned, a multi-disciplinary approach is essential to proper diagnosis and therapeutic interventions. If no syndrome is identified in childhood, further examination after three or four years should be performed as many syndromes become more apparent at this age.[2]
Medical therapy
If retinal function is detectable, refraction and treatment of any underlying amblyopia is critical.
After initial socket expansion (see below), patients must still be followed to monitor for angle closure glaucoma, retinal detachment (if chorioretinal coloboma), and need for correction of refractive error.
Surgery
The reduction of ocular volume caused by microphthalmia affects normal facial and orbital development. Therefore, early socket expansion, usually started within weeks of life, is imperative to minimize facial deformity, particularly in cases of severe microphthalmia. Therapeutic strategies aim simultaneous management of both soft tissue hypoplasia and asymmetric bone growth. [23][24][25]
Management and timing of initiating socket expansion therapy can be guided by the axial length. If the axial length of the eye is over 16 mm, orbital growth is more likely to be normal and the timing of expansion therapy is variable depending on social/aesthetic needs. However, if the axial length is less than 16 mm, it is unlikely to promote normal orbital growth alone and it is necessary to increase the socket volume early on to prevent pronouncing asymmetry as the child grows.[10]
Mild/moderate microphthalmia is generally managed conservatively with the insertion of progressively enlarging conformers. The length of time between changing conformers is not uniform and averages between a week to month.[10][26]
In severe mircophthalmia, socket expansion through conformers is often not enough and these patients require additional interventions including endo-orbital volume replacement (implants, expanders, dermis fat grafting). Orbital osteotomies are indicated in more severe cases. One study even described an expanding bony orbitotomy with the implantation of an inflatable balloon device to expand the soft tissues.[26]
If an orbital cyst is present, depending on the size and location, it may be retained or removed. In some cases, large cysts may provide natural expansion of the socket, therefore surgery is postponed until it reaches 90% of the orbital volume, allowing removal for cosmetic reasons at about the time the child starts school.[2][10][27]
Prognosis
The potential for visual development depends on the ocular structures affected and the severity of the malformations. The treatment aims to maximize the existing vision and provide improvement at the aesthetic level.
References
- ↑ American Academy of Ophthalmology. Microphthalmos. https://www.aao.org/image/microphthalmos Accessed July 15, 2019
- ↑ 2.0 2.1 2.2 2.3 2.4 2.5 2.6 2.7 2.8 2.9 Verma AS, Fitzpatrick DR. Anophthalmia and microphthalmia. Orphanet J Rare Dis. 2007;2:47. Published 2007 Nov 26. doi:10.1186/1750-1172-2-47
- ↑ 3.0 3.1 Dharmasena, Aruna; Keenan, Tiarnan; Goldacre, Raph; Hall, Nick; Goldacre, Michael J (2017). "Trends over time in the incidence of congenital anophthalmia, microphthalmia and orbital malformation in England: Database study". British Journal of Ophthalmology. 101 (6): 735–739.
- ↑ 4.0 4.1 4.2 Morrison D, FitzPatrick D, Hanson I, Williamson K, van Heyningen V, Fleck B, Jones I, Chalmers J, Campbell H. National study of microphthalmia, anophthalmia, and coloboma (MAC) in Scotland: investigation of genetic aetiology. J Med Genet. 2002;39:16–22.
- ↑ 5.0 5.1 Campbell H, Holmes E, MacDonald S, Morrison D, Jones I. A capture-recapture model to estimate prevalence of children born in Scotland with developmental eye defects. J Cancer Epidemiol Prev. 2002;7:21–28.
- ↑ 6.0 6.1 Shaw GM, Carmichael SL, Yang W, Harris JA, Finnell RH, Lammer EJ. Epidemiologic characteristics of anophthalmia and bilateral microphthalmia among 2.5 million births in California, 1989–1997. Am J Med Genet A. 2005;137:36–40.
- ↑ Kallen B, Robert E, Harris J. The descriptive epidemiology of anophthalmia and microphthalmia. Int J Epidemiol. 1996;25:1009–1016.
- ↑ 8.0 8.1 Forrester MB, Merz RD. Descriptive epidemiology of anophthalmia and microphthalmia, Hawaii, 1986–2001. Birth Defects Res A Clin Mol Teratol. 2006;76:187–92.
- ↑ 9.0 9.1 9.2 9.3 9.4 9.5 PH, Danysh HE, Weber KA, Shaw GM, Mitchell LE, Lupo PJ. Epidemiology of anophthalmia and microphthalmia: Prevalence and patterns in Texas, 1999-2009. Am J Med Genet A. 2018 Sep;176(9):1810-1818.
- ↑ 10.0 10.1 10.2 10.3 10.4 10.5 10.6 10.7 N K Ragge, I D Subak-Sharpe, J R O Collin, A practical guide to the management of anophthalmia and microphthalmia. Eye. 2007 21:1290–1300
- ↑ Galindo-Ferreiro A, Elkhamary SM, Alhammad F, AlGhafri L, AlWehaib M, Alessa D, Aldossari S, Akaishi P, Khadekar R, AlShaikh O, Schellini SA. Characteristics and management of congenital anophthalmos and microphthalmos at a tertiary eye hospital. Orbit. 2019 Jun;38(3):192-198.
- ↑ 12.0 12.1 Weiss AH, Kousseff BG, Ross EA, Longbottom J. Simple microphthalmos. Arch Ophthalmol. 1989;107:1625–1630
- ↑ 13.0 13.1 Weiss AH, Kousseff BG, Ross EA, Longbottom J. Complex microphthalmos. Arch Ophthalmol. 1989;107:1619–1624.
- ↑ 14.0 14.1 14.2 Bardakjian T, Weiss A, Schneider A. Microphthalmia/Anophthalmia/Coloboma Spectrum. 2004 Jan 29 [Updated 2015 Jul 9]. Available from: https://www.ncbi.nlm.nih.gov/books/NBK1378/
- ↑ Taha Najim R, Topa A, Jugård Y, Casslén B, Odersjö M, Andersson Grönlund M. Children and young adults with anophthalmia and microphthalmia: Diagnosis and Management. Acta Ophthalmol. 2020 Dec;98(8):848-858.
- ↑ 16.0 16.1 Warburg M. An update on microphthalmos and coloboma. A brief survey of genetic disorders with microphthalmos and coloboma. Ophthalmic Paediatr Genet. 1991;12:57–63. doi: 10.3109/13816819109023675.
- ↑ Dolk H, Busby A, Armstrong BG, Walls PH. Geographical variation in anophthalmia and microphthalmia in England, 1988–94. BMJ. 1998;317:905–909.
- ↑ Weiland HT, Vermey-Keers C, Salimans MM, Fleuren GJ, Verwey RA, Anderson MJ. Parvovirus B19 associated with fetal abnormality. Lancet. 1987;1:682–683. doi: 10.1016/S0140-6736(87)90442-9.
- ↑ Knox EG, Lancashire RJ. Epidemiology of congenital malformations. London: HMSO; 1991.
- ↑ O'Keefe M, Webb M, Pashby RC, Wagman RD. Clinical anophthalmos. Br J Ophthalmol. 1987;71:635–638.
- ↑ Traboulsi EI. Compendium of Inherited Disorders and the Eye. New York: Oxford University Press; 2005
- ↑ 22.0 22.1 22.2 Society for Maternal-Fetal Medicine, Benacerraf BR, Bromley B, Jelin AC. Anophthalmia and Microphthalmia. Am J Obstet Gynecol. 2019 Nov;221(5):B20-B21.
- ↑ Kennedy RE. Growth retardation and volume determinations of the anophthalmic orbit. Trans Am Ophthalmol Soc. 1972;70:277–297.
- ↑ Kennedy RE. The Effect of Early Enucleation on the Orbit in Animals and Humans. Trans Am Ophthalmol Soc. 1964;62:459–510.
- ↑ Hintschich C, Zonneveld F, Baldeschi L, Bunce C, Koornneef L. Bony orbital development after early enucleation in humans. Br J Ophthalmol. 2001;85:205–208.
- ↑ 26.0 26.1 Wavreille O, François Fiquet C, Abdelwahab O, Laumonier E, Wolber A, Guerreschi P, Pellerin P. Surgical and prosthetic treatment for microphthalmia syndromes. Br J Oral Maxillofac Surg. 2013 Mar;51(2):e17-21.
- ↑ Cui Y, Li Y, Hou Z, Wang Y, Chang Q, Xian J, Li D. Management of congenital microphthalmos and anophthalmos with orbital cyst. J AAPOS. 2019 Apr;23(2):92.e1-92.e6. doi: 10.1016/j.jaapos.2018.10.015. Epub 2019 Mar 27.