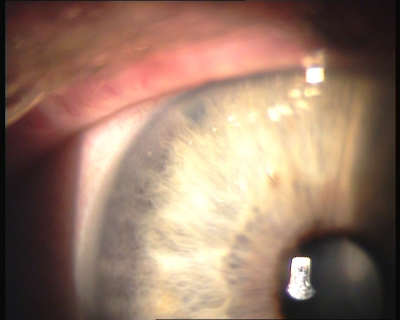
Laser Peripheral Iridotomy
All content on Eyewiki is protected by copyright law and the Terms of Service. This content may not be reproduced, copied, or put into any artificial intelligence program, including large language and generative AI models, without permission from the Academy.
Introduction
Laser peripheral iridotomy (also described as ‘laser iridotomy’ or simply termed 'iridotomy') is a medical procedure which uses a laser device to create a hole in the iris, thereby allowing aqueous humor to traverse directly from the posterior to the anterior chamber and, consequently, relieve a pupillary block.[1][2][3] It is commonly used to treat a wide range of clinical conditions, encompassing primary angle‐closure glaucoma, primary angle closure (narrow angles and no signs of glaucomatous optic neuropathy), patients who are primary angle‐closure suspects (patients with reversible obstruction) and even eyes with secondary causes of iridocorneal angle-closure.[1]
The first report of an effective surgical treatment for glaucoma was made in 1857 by the German ophthalmologist Albrecht von Graefe, who described the execution of a broad-sector surgical iridectomy through a corneal wound.[4]
However, since the emergence of laser technology in the field of ophthalmology, laser peripheral iridotomy (LPI) has largely surpassed the former technique. In the mid-1970’s, argon laser became the first to be routinely used to perform a non-invasive iridotomy.[5][6][7] Although it is well absorbed by iris pigment, argon laser (and Nd:YAG-KTP laser, which is often referred to as "argon laser") iridotomy alone was associated with some complications and relatively high failure and subsequent closure rates.[5][8][9] Q-switched Nd:YAG laser (YAG laser 1064 nm) devices surfaced in the 1980’s and demonstrated to be effective in light-coloured irides because its mechanism of action was independent of the melanin content of the iris. Furthermore, it required considerably less total energy than pure argon/Nd:YAG-KTP 532 nm laser iridotomy, achieved a superior rate of single treatment success with lower risk of subsequent closure and was less likely to cause damage to the cornea, lens and retina.[3][5][10][11][12]
Indications
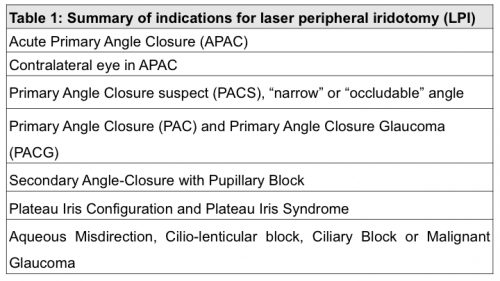
Laser peripheral iridotomy (LPI) is indicated to prevent or overcome a suspected relative pupillary block by creating an alternative pathway for aqueous flow. Mainly used for patients in the primary angle closure spectrum, it can also be useful in secondary angle closure glaucoma and in the management of other types of glaucoma with associated pupillary block. The iridocorneal angle should be, in all cases, carefully examined after LPI to rule out other mechanisms of a closed angle requiring treatment.[13] The indications for LPI are summarized in table 1.
1. Acute Primary Angle Closure (APAC)
The benefit of LPI is well established to treat and prevent an acute angle closure (AAC) crisis. The technique is usually performed soon after the diagnosis is made in order to prevent irreversible vision loss and prevent a recurrent AAC episode. It is essential to perform LPI in the fellow eye when indicated.[2][3] Before performing an LPI, however, patients experiencing APAC should receive aqueous suppressants to lower the IOP, relieve symptoms, improve cooperation and visualization of the ocular media in order to safely and effectively apply the laser beam.[2][3][5] In conclusion, LPI associated with medical treatment is the preferred definitive treatment of acute angle-closure glaucoma with a pupillary block mechanism.[1]
2. Contralateral Eye in Acute Primary Angle Closure
The fellow eye of a patient with AAC crisis should be evaluated because it is at high risk for a similar event and can occur within days of the first episode.[2] LPI is indicated in gonioscopically-proven occludable angles after an AAC episode as 50% of patients will suffer a second attack in the fellow eye over a 5-year period.[1][2][14][15]The risk is significantly reduced if an iridotomy or iridectomy is performed in this eye.[2][16]
3. Primary-Angle Closure suspect (PACS), “narrow” or “occludable” angle
These eyes feature at least 2 quadrants of iridotrabecular contact (ITC) but normal IOP, no peripheral anterior synechiae (PAS) and no evidence of glaucomatous optic neuropathy or visual field defects. Observational studies suggest that the majority of these eyes will not develop either PAC or PACG.[2][17] All cases of PACS must be assessed individually. In general, the treatment complications are to be balanced against the perceived risk of angle-closure.[1][2][5]The ZAP trial[18] demonstrated a small, but significant, prophylactic effect of LPI in these cases, however, the widespread use of prophylactic LPI for PACS or “occludable angles” is not recommended. Therefore, some authors defend that following such patients without treatment is a safe option.[2][19]
4. Primary Angle Closure (PAC) and Primary Angle-Closure Glaucoma (PACG)
Iridotomy is strongly recommended in eyes with PAC or PACG.[1][2][20] PAC patients may have elevated IOP from chronic compromise of aqueous outflow. Chronic angle-closure glaucoma features permanent synechial closure of any extent as confirmed by indentation gonioscopy.[1] Medical treatment alone is contraindicated as all patients require relief of pupil block by iridotomy, iridectomy or lens extraction. If the peripheral anterior synechiae occupy less than 50% of the circumference, iridectomy or iridotomy may suffice. Since complications of laser iridotomy are uncommon, its use as the initial procedure is justified in practically every case, as soon as the corneal transparency allows the LPI to be performed. If IOP remains uncontrolled after LPI, a filtering surgery should be considered as the next step.[1] However, In asymptomatic (“chronic”) angle-closure, a high presenting pressure (>35 mmHg), more than 6-clock hours of peripheral anterior synechiae and/or established glaucomatous optic neuropathy are signs that the angle-closure will not respond fully to an LPI, and that surgical treatment may be needed to reach the IOP goal.[1]
5. Secondary Angle-Closure Mechanisms
Uveitis is a common cause of secondary pupillary block and laser iridotomy may be required to prevent recurrence of pupillary block. Pupillary block can also develop slowly with a senile cataract or rapidly with dislocated or subluxated lenses, or with swollen post-trauma lenses. Iridotomy may not be curative, but helps by relieving pupillary block component, allowing for safer lensectomy. Pupillary block can occur in pseudophakic eyes, more often with anterior chamber lenses if an iridectomy is absent or occluded.[1][3][5]
6. Other Indications
1. Pigmentary Dispersion Syndrome (PDS), Pigmentary Ocular Hypertension (POH) and Pigmentary Glaucoma (PG)
In PDS, the principle supporting LPI is to equalize the pressure between the anterior and posterior chamber so as to reduce the posterior concavity of the iris. This would allow for reduced friction between the iris and zonular fibers and reduce or eliminate pigment dispersion.[1][2][21][22] ND:YAG LPI has been proposed as a means for eliminating a reverse pupillary block, although the benefit from an LPI in patients with PDS, POH and PG is not yet established. However, LPI has demonstrated to decrease the biomechanical factor of iris-zonule contact, which may prove to be beneficial to patients before the onset of trabecular dysfunction. There is insufficient evidence of high quality on the effectiveness of peripheral iridotomy for pigmentary glaucoma or pigment dispersion syndrome and, therefore, the long-term effects on visual function and other patient-important outcomes have not been established. In pigmentary glaucoma, this procedure is usually not recommended, since there is already an optic neuropathy which can progress with IOP spikes after LPI.[21][22]
2. Plateau Iris Configuration and Plateau Iris Syndrome
Plateau Iris configuration refers to a specific condition in which eyes to have ITC after iridotomy, with close apposition of the peripheral iris to the angle on gonioscopy and a deep central anterior chamber.[1][2] On pupil dilation, these eyes do not experience a significant increase of IOP as opposed to eyes with Plateau Iris Syndrome, which experience an IOP spike in this scenario and may require more extensive treatments. About one third of eyes with PAC treated with LPI have an angle that retains significant ITC.[2][23] LPI should be performed in every eye suspected of having a Plateau Iris configuration, since it eliminates any pupillary block component.[1][2] These eyes present a thicker iris and an anteversion of the ciliary body and these anatomical factors predict failure of a laser iridotomy to open an appositionally closed angle.[2][5] The long-term risk of PACG in eyes with Plateau Iris configuration does not appear to be higher than in eyes whose angles widen more after LPI.[2]
3. Aqueous Misdirection, Cilio-lenticular block, Ciliary Block or Malignant Glaucoma
Aqueous misdirection is a challenging form of secondary angle closure that presents with elevated intraocular pressure (IOP) and shallowing of the central and peripheral anterior chamber caused by anterior rotation of the iris and ciliary body, occurring classically in the post-operative period. Since this clinical entity is a diagnosis of exclusion, LPI is important since it eliminates the possibility of a pupillary block mechanism in ciliary block glaucoma.[1]
4. Congenital anomalies that can be associated with secondary glaucoma
Familial iris hypoplasia, anomalous superficial iris vessels, aniridia, Sturge-Weber syndrome, neurofibromatosis, Marfan’s syndrome, Pierre Robin syndrome, homocystinuria, goniodysgenesis, Lowe’s syndrome, microcornea, microspherophakia, rubella, broad thumb syndrome, persistent hyperplastic primary vitreous. Laser iridotomy and surgical iridectomy are not effective.[1]
5. Systemic drugs with effects on the iridocorneal angle
A wide range of systemic drugs (e.g. topiramate, methylphenidate, selective serotonin re-uptake inhibitors, tricyclic antidepressants) have been implicated in case reports with angle-closure, however, these eyes pose added difficulties in the decision-making process since the pre-treatment gonioscopy findings alone are not enough to rule out such risk. It is sensible to repeat the gonioscopy in eyes with narrow angles whenever these drugs are initiated. The role of prophylactic LPI in this patient population should weighed against the risk of angle-closure or of withdrawal of these drugs.[1][2]
Contraindications
The following contraindications should be managed prior to performing an LPI.[2][3][5]
- Ocular media opacities (e.g. corneal edema, anterior chamber inflammation)
- High risk eyes (e.g. very shallow or flat anterior chambers, uveitis)
- Uncooperative patients.
Preoperative Management
The following recommendations should be followed in order to safely and effectively perform an LPI:[1][2][5][20]
- Apply pilocarpine 2%-4% to cause miosis, reduce iris thickness and facilitate perforation.
- In the presence of corneal edema, use topical glycerin 10% if available, systemic acetazolamide, intravenous mannitol or oral hyperosmotic agents.
- IOP-lowering drugs: to prevent IOP spikes after the procedure use topical alpha-2 agonist (brimonidine or apraclonidine) 1 hour prior to the procedure and immediately afterwards. In the setting of AAC, IOP should be controlled beforehand with medical treatment.
- Apply topical anaesthesia (e.g. oxybuprocaine).
- Apply iridotomy lens (e.g.Abraham (+66 diopters), Wise (+103 diopters) or CGI©LASAG CH lens) to the eye with coupling gel. The use of the lens keeps the eyelids open, minimizes eye movement, provides magnification and improves visualization, reducing the risk of corneal burns and simultaneously increases the power density by concentrating the laser energy.
- Focus: Set defocus to zero, focus first on the iris surface and then offset the YAG beam so that it converges slightly posteriorly in the stroma. A bubble of plasma travels towards the surgeon so it is safer to have the focal point within the iris stroma.
- Position of iridotomy: The hole should be created in the iris periphery, aiming for an iris crypt or area of iris thinning, at a site in the superior quadrants between 11 and 1 o’clock, which allows the lesion to be covered by the upper eyelid, avoiding the 12 o'clock position as air bubbles may occlude the iridotomy. Avoid iris vessels. If silicone oil (except high MW silicone) or other tamponading fluids or gases present, perform an inferior LPI.
- Parameters: Consult tables 2 and 3.
- Endpoint: A small (150 to 200 micron), peripheral and completely patent is the ideal result. Whole thickness perforation of the iris is assumed when pigment, mixed with aqueous, flows from the posterior into the anterior chamber. Once a full thickness hole has been made, it should be enlarged horizontally to achieve an adequate size. Iridotomy size should be sufficient for patency in spite of iris oedema, pigment epithelial proliferation and pupil dilation. Transillumination through the iridotomy is not a reliable indicator of patency
- Post-operative medication: Prescribe a topical corticosteroid 3 to 4 times a day for 4-7 days as well as a IOP-lowering drug.
- Evaluate IOP in 30 minutes to 2 hours following LPI and anterior chamber depth one week later to confirm opening of the AC angle. If the patency is uncertain check with gonioscopy, reconsider the mechanism, perform ultrasound biomicroscopy (UBM) / anterior segment-optical coherence tomography (AS-OCT) if available and/or repeat the iridotomy.
Types of Lasers for LPI
Iridotomy can be accomplished using the photodisruptive Q-switched Nd:YAG laser (Nd:YAG) or the photo-thermal Nd:YAG-KTP 532 nm (which is still often called Argon laser) and solid-state lasers. The two laser types may also be used in sequential combination. Laser parameters should be individualized to each patient and adjusted appropriately during the procedure. Independently of the device used, some authors aim for an iridotomy of at least 200 microns in size and ideally 500 micron in diameter.[3][5]
Argon/Nd:YAG-KTP 532 nm or solid-state Laser
Nd:YAG-KTP 532 nm and solid-state lasers produce thermal (coagulative) effects with lower energies at higher exposures or explosive effects (vaporization) if higher energies are used. Unlike Nd:YAG, these lasers require pigment for light energy absorption and therefore need more adjustments and greater variety in techniques and laser parameters to iris pigment density posterior to the stroma. Sequential Nd:YAG-KTP 532nm/Nd:YAG LPI can be useful in patients with thick brown irides, particularly in East Asian and African populations. Nd:YAG-KTP 532 nm laser pre-treatment to thin the iris results in lower energy needed with the Nd:YAG laser, which is then used to penetrate the iris and create an iridotomy. By using only a fraction of the power required for pure YAG laser iridotomy, it is more efficient and carries a lower risk of complications, namely anterior chamber hemorrhage.[3][5]
Nd:YAG laser iridotomy
The Q-switched Nd:YAG lasers rely on tissue photodisruption to create a rapidly expanding ionic plasma wave and thus is efficient in all iris colours. The subsequent shock-wave and mainly cavitation bubble will in turn mechanically burst through the stromal tissue. The Nd:YAG laser is preferred by many surgeons because it perforates the iris easily, requiring lower energy than Nd:YAG-KTP 532 nm lasers. It is easier to penetrate dark brown irides with these devices because the former have a tendency to absorb too much energy during treatment, and pale irides absorb laser energy poorly. Moreover, Nd: YAG iridotomies may be less likely to close over time.[1][3][5][9][24]
Complications
1. Transient Increase in IOP
Acute onset IOP elevation after LPI is the most common postoperative complication following LPI. It is usually transient, occurring most frequently in the first 4 hours after treatment. At least an 8 mmHg increase from baseline has been reported in 6% to 10% of patients and an IOP superior to 21 mmHg in 2% to 72%.[1][25] Its occurrence as a primary event is thought to be caused by obstruction of the trabecular meshwork by released blood, iris pigment and other debris.[5] Plateau iris syndrome or other non-pupillary block angle closure mechanism, inflammation, extensive peripheral anterior synechiae (PAS) and prolonged corticosteroid therapy are possible secondary causes of IOP elevation post-procedure. Prophylaxis with pressure-lowering agents is an effective strategy to prevent IOP rise after the procedure.[1][12]
2. Anterior Chamber Bleeding (Hyphema)
Ii is a common complication post-LPI. It refers to bleeding from the iridotomy site, is minor in most cases and can usually be stopped by light pressure applied to the eye with the contact lens. The incidence and severity of anterior chamber bleeding has been reported to be similar whether the patient was on or off antithrombotic therapy (i.e. aspirin, clopidogrel, warfarin) and between groups taking different antithrombotic medications. Therefore, antiaggregants and anticoagulants need not be discontinued before LPI, and specific patients may have a bleeding diathesis regardless of the medications taken.[25][26][27]
3. Visual Dysphotopsias
Visual symptoms are uncommon but known to occur after the procedure, which include blurring, glares, halos, lines, spots and shadows at a reported rate of 2% to 16 %. [25] Symptom rates according to LPI location are mixed across large studies. The practice of placing laser peripheral iridotomy (LPI) at the 10-2 o’clock position to prevent dysphotopsia illustrates how anatomical reasoning can lead us astray. While it seems logical that hiding the iridotomy behind the upper lid would reduce visual symptoms, systematic study of patient outcomes fails to support this assumption. A systematic review of 1,756 eyes from 878 patients compared the incidence of dysphotopsia based on LPI location (superior, inferior, or nasal/temporal). The analysis found no significant difference in the incidence of dysphotopsia among the location groups. Overall, the incidence of linear dysphotopsia was 2-3% regardless of LPI placement. Interestingly, the review also noted that LPI could resolve pre-existing halos and glare, suggesting that the procedure may have some beneficial effects on visual symptoms. [28]Although Spaeth et al.[29] report dysphotopsia symptoms after LPI in 9% of eyes with completely covered, 26% with partially covered and 17.5% with fully exposed iridotomies, a large trial by Srinivasan et al. reported no statistical difference of new visual symptoms according to LPI location (superior versus nasal or temporal) 2 weeks post-LPI.[30] The same group also found that the majority of visual symptoms were likely to resolve after 6 months of follow-up which may indicate that patients adapt or learn to ignore them.[31] Finally, temporal LPI have been reported to cause less visual symptoms versus superior LPIs.[25][32] Therefore, patients complaining of newly-onset visual symptoms post-LPI can be reassured that improvement of symptoms may occur over time without active intervention. Tinted lenses have been used to relieve visual symptoms.[3]
4. Cataract Progression
The risk of a traumatic cataract has been reported to be increased in post-LPI subjects at follow-up periods ranging from 12 months to 6 years[11][25][33][34], and case reports of posterior subcapsular cataracts after LPI may be found in the literature.[3]Postulated mechanisms include direct damage to the crystalline lens, altered aqueous fluid dynamics and mild inflammatory changes to the iris which may induce metabolic changes that accelerate cataract formation.[5]
5. Iridotomy closure and necessity for repeat LPI
Closure of the iridotomy has been reported to occur at a rate between 1% at 2 weeks to 20% within the first 6 months after the procedure, but the vast majority of iridotomies may remain patent years after the procedure.[25][35][36] This may occur due to accumulation of debris and pigment granules, although complete closure of the iridotomy is unusual with ND:Yag lasers. Repeat LPI can be performed at the same site immediately after the first procedure, a few hours later if debris impede the observation of the iridotomy or creating an iridotomy at another site.
6. Aqueous misdirection syndrome
In rare cases, aqueous misdirection can complicate the management of primary angle-closure. This may occur following laser iridotomy and other surgical procedures. Forward movement of the lens iris diaphragm causes secondary angle-closure resulting in IOP elevation. These cases, typically have very small eyes (axial length <21 mm) and higher hypermetropic refraction (> +6D). It is believed that the ciliary processes come into contact with the lens equator, and/or a firm zonule/posterior capsule diaphragm, causing misdirection of aqueous into the vitreous.[1] Malignant glaucoma is a rare complication of laser iridotomy and should be considered in eyes with progressive anterior chamber shallowing and myopia despite normal IOP.
Other complications
A reduction in endothelial cell count after LPI has been reported in the literature.[12][25] Postoperative inflammation is usually transient and mild, rarely resulting in posterior synechiae. Rare complications include sterile hypopyon, macular hole formation, cystoid macular oedema and retinal haemorrhages.[10][26][37]
Outcomes
1. IOP control
The effectiveness of the procedure in controlling the IOP varies according to the staging of the disease, with earlier stages of the angle-closure spectrum reporting the highest success rates, while the prognosis is less favorable after an AAC crisis an even more guarded if glaucomatous optic nerve damage can be demonstrated. In fact, several studies regarding the spectrum of PAC report that 7.1%-28.0% of PACS eyes, 42.4%-80.0% PAC eyes and 83.3%-100% of PACG eyes required additional medical and/or surgical intervention after LPI.[5][38][39][40] On the other hand, the EAGLE trial, which included PAC and PACG patients, showed that initial treatment with clear-lens extraction was superior to LPI with topical medical treatment for participants with an IOP over 30 mmHg and primary angle closure or primary angle-closure glaucoma, with significantly fewer participants in the clear-lens extraction group needing additional treatment to control intraocular pressure and fewer glaucoma medications than patients who received standard care. The benefit of CLE in patients with PAC and an IOP inferior to 30 mmHg is unclear. Intraocular pressure was better with clear-lens extraction than with standard care, with the mean pressure being around 1 mm Hg lower in the clear-lens extraction group at 3 years.[25][41]
2. Anatomical improvement
The effect of LPI on angle width has been evaluated across studies using a variety of quantitative and qualitative parameters. A large number of studies have demonstrated short-term increases in angle width across the PAC spectrum expressed as an increase in the Shaffer grade and an increase in quantitative angle-width parameters using UBM and AS-OCT.[25] However, two prospective studies have demonstrated a gradual and significant decrease in angle width after LPI was performed.[18][42] Baseline anatomical factors associated with persistent angle closure after LPI include the presence of PAS, increased IOP, UBM parameters such as shallower peripheral angle width, thicker iris, and more anteriorly positioned ciliary body; and AS-OCT parameters such as greater mean lens vault and thicker iris. These factors represent a narrower angle before LPI and non-pupillary block mechanisms of angle closure.[25] Indeed, LPI induces significant changes in the 3-D anterior segment morphology in primary angle closure suspect patients with an increase in central anterior chamber depth, anterior chamber volume and widening of the iridocorneal angle with no change in central corneal thickness, intraocular pressure or pupil diameter.[43] LPI is more effective than pilocarpine in widening the iridocorneal angle without significant shallowing the anterior chamber in eyes with anatomically narrow angles.[44]
3. Prevention of AAC crisis
LPI has been shown in trials to prevent recurrent attacks of acute angle closure, with an effectiveness and safety equal to that of surgical iridectomy, and with better acceptability to patients.[45] Good effectiveness in preventing acute attacks in fellow eyes has also been reported.[16] Nonetheless, isolated reports do exist of eyes experiencing a recurrent acute attack after LPI.[3][46] In AAC, laser iridotomy relieves the attack in most cases. Superior outcomes have been reported in Caucasian eyes than in Asian eyes[3]. In AAC attacks, significant amount of PAS, a higher presenting IOP, longer attack duration, poor initial response to therapy (<30% IOP) and a larger C/D ratio, have been associated with inadequate long-term IOP control after LPI. These findings are consistent with a more severe disease affecting the ITC and the trabecular meshwork.[2][3][25]
4. Disease Progression
Progression from PACS to PAC was based on the development of PAS or elevated IOP. Progression from PACS or PAC to PACG was defined using both structural (disc appearance) and functional (visual field) criteria in most studies.[2] In the EAGLE cohort, visual acuity outcomes in the clear-lens extraction (CLE) group were similar to the LPI group.[47] In the EAGLE trial, CLE was superior to LPI in improving visual function. This improvement (eg, contrast sensitivity) may have resulted from eliminating mild age-related changes in lens transparency, and by correction of refractive error, which caused an improvement in good visual acuity without the need for spectacles. While this magnitude of change is unlikely to be clinically important, it points to the overall improvement in visual function associated with CLE. A recent systematic review found insufficient evidence based on randomized controlled trials as to whether iridotomy slows progression of visual field loss at one year compared to no iridotomy.[48]
Summary
LPI is an integral component in the management of PAC. Although LPI has been available since the 1970s, its role in the treatment algorithm for PAC is still under debate; questions such as who should be treated with an iridotomy and whether iridotomy prevents disease progression continue to be relevant today.[2] After LPI, most PACS subjects can expect to receive no further treatment. On the other hand, many PAC and APAC eyes, and most PACG eyes, need additional treatment to control IOP. LPI has a favorable effect on IOP, especially when extensive synechial closure or glaucomatous nerve damage have not occurred. Factors predictive for IOP elevation, or the need for further treatment after LPI, were those reflective of worse disease at baseline, such as higher IOP, greater cup-to-disc ratio, and greater synechial closure at presentation.[2] The safety and effectiveness of LPI for PAC/PACG has promoted a gradual widening of the criteria for iridotomy during the past couple of decades. However, more inclusive criteria spurred the debate over treatment algorithms, as we still do not know which eyes will develop glaucomatous damage, despite early manifestations of narrow angles, and offering iridotomy too soon would expose healthier patients to complications without clear evidence of benefit.[5] More recently, the role of cataract extraction in the PAC disease spectrum has continued to fuel debate around LPI timing and its indications. In fact, the EAGLE recommends clear-lens extraction as first-line treatment for the subset of PAC patients with an IOP over 30 mmHg. Newer and more objective angle imaging devices are expected to contribute towards predicting the success of laser iridotomy in the future.[2][5][41] In conclusion, LPI increases angle width in all stages of PAC and has a favorable effect on IOP in eyes without extensive angle or disc damage. Although there is a gradual decrease in angle width in PACS eyes after LPI, further treatment after LPI in this subgroup is infrequent. In contrast, additional treatment after LPI is more likely in PAC, APAC, and PACG eyes, and they should be monitored for IOP elevation and progression to glaucoma.[1][2][13]
References
- ↑ Jump up to: 1.00 1.01 1.02 1.03 1.04 1.05 1.06 1.07 1.08 1.09 1.10 1.11 1.12 1.13 1.14 1.15 1.16 1.17 1.18 1.19 1.20 1.21 European Glaucoma Society terminology and guidelines for glaucoma, 4th edition---part 1: supported by the EGS Foundation. Br J Ophthalmol. 2017;101(4):1-72.
- ↑ Jump up to: 2.00 2.01 2.02 2.03 2.04 2.05 2.06 2.07 2.08 2.09 2.10 2.11 2.12 2.13 2.14 2.15 2.16 2.17 2.18 2.19 2.20 2.21 2.22 2.23 2.24 Prum BE Jr, Herndon LW Jr, Moroi SE, et al. Primary Angle Closure Preferred Practice Pattern guidelines. Ophthalmology. 2016;123(1):P1-P40.
- ↑ Jump up to: 3.00 3.01 3.02 3.03 3.04 3.05 3.06 3.07 3.08 3.09 3.10 3.11 3.12 3.13 Lam DSC, Tham CCY, Congdon NG, Baig N. Peripheral iridotomy for angle-closure glaucoma. In: Shaarawy TM, Sherwood MB, Hitchings RA, Crowston JC, eds. Glaucoma. Elsevier; 2015;708-715. doi:10.1016/B978-0-7020-5193-7.00072-8
- ↑ Gräfe A. Ueber die Iridectomie bei Glaucom und über den glaucomatösen Process [Internet]. Albrecht von Graefes Archiv für Ophthalmologie. 1857;3:456-555. doi:10.1007/bf02720732
- ↑ Jump up to: 5.00 5.01 5.02 5.03 5.04 5.05 5.06 5.07 5.08 5.09 5.10 5.11 5.12 5.13 5.14 5.15 5.16 Cruz M. Laser iridotomy. In: Henriques J, Duarte A, Quintão T, eds. Laser Manual in Ophthalmology--Fundamentals and Laser Clinical Practice. Medical Laser Interdisciplinary Portuguese Society; 2017:115-122.
- ↑ Khuri CH. Argon laser iridectomies. Am J Ophthalmol. 1973;76:490-493.
- ↑ Palanker DV. Fifty Years of Ophthalmic Laser Therapy [Internet]. Archives of Ophthalmology. 2011. p. 1613. doi:10.1001/archophthalmol.2011.293
- ↑ Quigley HA. Long-term follow-up of laser iridotomy. Ophthalmology. 1981;88: 218–224.
- ↑ Jump up to: 9.0 9.1 Robin AL, Pollack IP. A Comparison of Neodymium:YAG and Argon Laser Iridotomies. Ophthalmology. 1984;91: 1011–1016.
- ↑ Jump up to: 10.0 10.1 Berger BB. Foveal photocoagulation from laser iridotomy. Ophthalmology. 1984;91: 1029–1033.
- ↑ Jump up to: 11.0 11.1 Vijaya L, Asokan R, Panday M, George R. Is prophylactic laser peripheral iridotomy for primary angle closure suspects a risk factor for cataract progression? The Chennai Eye Disease Incidence Study. Br J Ophthalmol. 2017;101: 665–670.
- ↑ Jump up to: 12.0 12.1 12.2 Kumar RS, Baskaran M, Friedman DS, Xu Y, Wong H-T, Lavanya R, et al. Effect of prophylactic laser iridotomy on corneal endothelial cell density over 3 years in primary angle closure suspects. Br J Ophthalmol. 2013;97: 258–261.
- ↑ Jump up to: 13.0 13.1 International Council of Ophthalmology : Enhancing Eye Care : Glaucoma [Internet]. [cited 1 Jun 2019]. Available: http://www.icoph.org/enhancing_eyecare/glaucoma.html
- ↑ Lowe RF. Acute Angle-Closure Glaucoma: The second eye: An analysis of 200 cases. Br J Ophthalmol. 1962;46: 641–650.
- ↑ Bain WE. The fellow eye in acute closed-angle glaucoma. Br J Ophthalmol. 1957;41: 193–199.
- ↑ Jump up to: 16.0 16.1 Ang LP, Aung T, Chew PT. Acute primary angle closure in an Asian population: long-term outcome of the fellow eye after prophylactic laser peripheral iridotomy. Ophthalmology. 2000;107: 2092–2096.
- ↑ Wilensky JT, Kaufman PL, Frohlichstein D, Gieser DK, Kass MA, Ritch R, et al. Follow-up of angle-closure glaucoma suspects. Am J Ophthalmol. 1993;115: 338–346.
- ↑ Jump up to: 18.0 18.1 Jiang Y, Chang DS, Zhu H, Khawaja AP, Aung T, Huang S, et al. Longitudinal changes of angle configuration in primary angle-closure suspects: the Zhongshan Angle-Closure Prevention Trial. Ophthalmology. 2014;121: 1699–1705.
- ↑ Weinreb RN, Moghimi S. Prophylactic laser iridotomy in primary angle-closure suspects. Lancet. 2019;393: 1572–1574.
- ↑ Jump up to: 20.0 20.1 Weinreb RN, Friedman DS. Angle Closure and Angle Closure Glaucoma: Reports and Consensus Statements of the 3rd Global AIGS Consensus Meeting on Angle Closure Glaucoma. Kugler Publications; 2006.
- ↑ Jump up to: 21.0 21.1 Michelessi M, Lindsley K. Peripheral iridotomy for pigmentary glaucoma. Cochrane Database Syst Rev. 2016;2: CD005655.
- ↑ Jump up to: 22.0 22.1 Buffault J, Leray B, Bouillot A, Baudouin C, Labbé A. Role of laser peripheral iridotomy in pigmentary glaucoma and pigment dispersion syndrome: A review of the literature [Internet]. Journal Français d’Ophtalmologie. 2017. pp. e315–e321. doi:10.1016/j.jfo.2017.09.002
- ↑ Kumar RS, Tantisevi V, Wong MH, Laohapojanart K, Chansanti O, Quek DT, et al. Plateau iris in Asian subjects with primary angle closure glaucoma. Arch Ophthalmol. 2009;127: 1269–1272.
- ↑ Pollack IP, Robin AL, Dragon DM, Green WR, Quigley HA, Murray TG, et al. Use of the neodymium:YAG laser to create iridotomies in monkeys and humans. Trans Am Ophthalmol Soc. 1984;82: 307–328.
- ↑ Jump up to: 25.00 25.01 25.02 25.03 25.04 25.05 25.06 25.07 25.08 25.09 25.10 Radhakrishnan S, Chen PP, Junk AK, Nouri-Mahdavi K, Chen TC. Laser Peripheral Iridotomy in Primary Angle Closure: A Report by the American Academy of Ophthalmology. Ophthalmology. 2018;125: 1110–1120.
- ↑ Jump up to: 26.0 26.1 Arunmongkol S, Suwan Y, Supakontanasan W, Nilphatanakorn S, Teekhasaenee C. Eight-ball hyphema after laser iridotomy in a patient with undiagnosed hypocellular myelodysplastic syndrome. Int Med Case Rep J. 2018;11: 133–137.
- ↑ Golan S, Levkovitch-Verbin H, Shemesh G, Kurtz S. Anterior chamber bleeding after laser peripheral iridotomy. JAMA Ophthalmol. 2013;131: 626–629.
- ↑ Balas M, Mathew DJ. Dysphotopsia and location of laser iridotomy: a systematic review. Eye (Lond). 2024;38(7):1240-1245. doi:10.1038/s41433-023-02913-1
- ↑ Spaeth GL, Idowu O, Seligsohn A, Henderer J, Fonatanarosa J, Modi A, et al. The effects of iridotomy size and position on symptoms following laser peripheral iridotomy [Internet]. American Journal of Ophthalmology. 2006. pp. 427–428. doi:10.1016/j.ajo.2006.01.006
- ↑ Srinivasan K, Zebardast N, Krishnamurthy P, Abdul Kader M, Raman GV, Rajendrababu S, et al. Comparison of New Visual Disturbances after Superior versus Nasal/Temporal Laser Peripheral Iridotomy: A Prospective Randomized Trial. Ophthalmology. 2018;125: 345–351.
- ↑ Kavitha S, Ramulu PY, Venkatesh R, Palaniswamy K, Kader MA, Raman GV, et al. Resolution of Visual Dysphotopsias after Laser Iridotomy: Six-Month Follow-up. Ophthalmology. 2019;126: 469–471.e1.
- ↑ Vera V, Naqi A, Belovay GW, Varma DK, Ahmed IIK. Dysphotopsia after temporal versus superior laser peripheral iridotomy: a prospective randomized paired eye trial. Am J Ophthalmol. 2014;157: 929–935.
- ↑ Tan AM, Loon SC, Chew PTK. Outcomes following acute primary angle closure in an Asian population. Clin Experiment Ophthalmol. 2009;37: 467–472.
- ↑ Lim LS, Husain R, Gazzard G, Seah SKL, Aung T. Cataract progression after prophylactic laser peripheral iridotomy: potential implications for the prevention of glaucoma blindness. Ophthalmology. 2005;112: 1355–1359.
- ↑ Waisbourd M, Shafa A, Delvadia R, Sembhi H, Molineaux J, Henderer J, et al. Bilateral Same-day Laser Peripheral Iridotomy in the Philadelphia Glaucoma Detection and Treatment Project. J Glaucoma. 2016;25: e821–e825.
- ↑ Jiang Y, Chang DS, Foster PJ, He M, Huang S, Aung T, et al. Immediate changes in intraocular pressure after laser peripheral iridotomy in primary angle-closure suspects. Ophthalmology. 2012;119: 283–288.
- ↑ Tsui JC, Marks SJ. Unilateral Stage 1A Macular Hole Secondary to Low-Energy Nd:YAG Peripheral Iridotomy. Cureus. 2021;13(1):e12603. Published 2021 Jan 10. doi:10.7759/cureus.12603
- ↑ Peng P-H, Nguyen H, Lin H-S, Nguyen N, Lin S. Long-term outcomes of laser iridotomy in Vietnamese patients with primary angle closure. Br J Ophthalmol. 2011;95: 1207–1211.
- ↑ Rosman M, Aung T, Ang LPK, Chew PTK, Liebmann JM, Ritch R. Chronic angle-closure with glaucomatous damage: long-term clinical course in a North American population and comparison with an Asian population. Ophthalmology. 2002;109: 2227–2231.
- ↑ Chen M-J, Cheng C-Y, Chou C-K, Liu CJL, Hsu W-M. The long-term effect of Nd:YAG laser iridotomy on intraocular pressure in Taiwanese eyes with primary angle-closure glaucoma. J Chin Med Assoc. 2008;71: 300–304.
- ↑ Jump up to: 41.0 41.1 Azuara-Blanco A, Burr J, Ramsay C, Cooper D, Foster PJ, Friedman DS, et al. Effectiveness of early lens extraction for the treatment of primary angle-closure glaucoma (EAGLE): a randomised controlled trial. Lancet. 2016;388: 1389–1397.
- ↑ Lee KS, Sung KR, Shon K, Sun JH, Lee JR. Longitudinal changes in anterior segment parameters after laser peripheral iridotomy assessed by anterior segment optical coherence tomography. Invest Ophthalmol Vis Sci. 2013;54: 3166–3170.
- ↑ Mansoori T, Balakrishna N. Anterior segment morphology after laser iridotomy in primary angle closure suspects. Clin Exp Optom. 2018;101: 333–338.
- ↑ Mogil RS, Khezri N, Ren R, Adleyba O, Abumasmah R, Ghassibi MP, et al. Changes in Iridocorneal Angle and Anterior Chamber Structure in Eyes With Anatomically Narrow Angles: Laser Iridotomy Versus Pilocarpine. J Glaucoma. 2018;27: 1073–1078.
- ↑ Schwenn O, Sell F, Pfeiffer N, Grehn F. Prophylactic Nd:YAG-laser iridotomy versus surgical iridectomy: a randomized, prospective study. Ger J Ophthalmol. 1995;4: 374–379.
- ↑ Tanasescu I, Grehn F. Acute angle-closure glaucoma despite previous Nd: YAG laser iridotomy: a report on 13 cases. Ophthalmologe. 2003;100: 832–835.
- ↑ Day AC, Cooper D, Burr J, Foster PJ, Friedman DS, Gazzard G, et al. Clear lens extraction for the management of primary angle closure glaucoma: surgical technique and refractive outcomes in the EAGLE cohort. Br J Ophthalmol. 2018;102: 1658–1662.
- ↑ Le JT, Rouse B, Gazzard G. Iridotomy to slow progression of visual field loss in angle-closure glaucoma. Cochrane Database Syst Rev. 2018;6: CD012270.