Intermediate Uveitis
All content on Eyewiki is protected by copyright law and the Terms of Service. This content may not be reproduced, copied, or put into any artificial intelligence program, including large language and generative AI models, without permission from the Academy.
Disease Entity
Intermediate uveitis (IU) is a chronic, relapsing disease of insidious onset. According to the Standardization of Uveitis Nomenclature (SUN) working group criteria, IU is defined as an intraocular inflammation mainly focused on the vitreous and peripheral retina. It incorporates pars planitis, posterior cyclitis, and hyalitis.
IU accounts for 1.4 to 31% of all uveitis patients, and up to 20% of cases of pediatric uveitis.
Epidemiology
IU is a rare disease, with an estimated incidence of 1.4-2.0 /100.000. The prevalence is estimated to be 5.9/100.000. However, the epidemiology of IU seems to have geographical differences, possibly due to different etiologies. Whereas in Europe, the United States and China IU is usually idiopathic, in other parts of the world, such as India, infectious causes, particularly tuberculosis, are more frequent. No gender predominance has been noted.
Etiology
IU may be due to a number of causes:
Infectious causes
- Tuberculosis
- Leprosy
- Lyme’s disease
- Syphilis
- Toxocariasis
- Whipple’s disease.
Non-infectious causes
- Sarcoidosis – about 25% of patients with sarcoidosis develop IU, and 2 to 10% of patients with IU develop sarcoid disease
- Blau syndrome
- Multiple sclerosis (MS) – the proportion of MS in IU patients varies from 7 to 30.4%, and 3-27% of patients with MS develop IU. IU in children is rarely related to MS, but children with IU may develop MS later in life. Retinal periphlebitis at the time of diagnosis increased the risk of MS and/or optic neuritis. HLA-DR2 and HLA-DR15, -DR51 and -DQ6 were closely associated with both pars planitis and MS.
- Inflammatory Bowel Disease
- Lymphoma – 66% of intraocular lymphomas are a manifestation of a primary central nervous system lymphomas (PCNSL), and 10-20% of them may present as vitreous or retinal infiltrates mimicking uveitis.
- Tubulointerstitial nephritis and uveitis (TINU) syndrome
- Sjogren syndrome.
Idiopathic IU
IU without apparent cause is the most frequent type of IU, accounting for more than 70% of cases. Idiopathic IU where snowbanking and/or snowball formation occur has been termed pars planitis.
General Pathology
Histologic studies show condensed vitreous, fibroblasts, lymphocytes, and lymphocyte cuffing of the peripheral retinal veins. Snowbanks and snowballs consist of a fibrovascular layer containing mononuclear leukocytes and fibrocyte-like cells, as well as vitreous collagen, Muller cells and fibrous astrocytes.
Pathophysiology
IU seems to be a T-cell mediated disease, as these cells account for up to 95% of all cell types in the vitreous of patients with IU; most of these cells are CD4+. IU may be initiated by an unknown antigen, leading to a clinical picture of vasculitis and vitreous inflammation.
HLA associations have been reported in IU. HLA-DR is the most significant occurring in 67-72% of patients; others include HLA-B8 and HLA-B51.
Clinical Manifestations
History
IU classically affects younger patients, aged 15 to 40 years. Presentation is typically insidious and bilateral but asymmetrical in both eyes.
Symptoms
- Painless floaters
- Decreased/blurred vision
- Pain, photophobia and red eye are rare symptoms
Physical examination
- Visual acuity is variably affected depending on inflammatory activity and complications,
- Mild to moderate anterior segment inflammation may occur in 28-50% of patients, characterized by few cells and scattered keratic precipitates in the anterior segment. However, there may be more severe findings of anterior uveitis such as posterior synechia, particularly involving the inferior iris, and especially in IU associated with MS and in children,
- Vitreous cells with anterior predominance are a universal finding,
- Vitreous condensation and haze may occur in severe cases
- Snowballs are whitish focal collections of inflammatory cells and exudate, usually most numerous in the inferior vitreous,
- Snowbanking is characterized by a grey-white fibrovascular and/or exudative plaque, most frequently found inferiorly; it can often be seen only with indirect ophthalmoscopy and scleral depression,
- Peripheral periphlebitis is a common finding (16-36%), particularly in patients with MS,
- Younger patients may present with vitreous hemorrhage.
Diagnosis
IU is typically a clinical diagnosis. Investigation is usually warranted in cases of IU to determine potentially treatable causes and to exclude infection.
Notably, malignancy should be considered and investigated in elderly patients, as primary intraocular lymphoma may present with diffuse vitritis mimicking IU. Along with severe vitritis, chorioretinal lesions and poor or partial response to therapy should raise suspicion for lymphoma.
Laboratory studies
- Complete blood count (CBC)
- Erythrocyte sedimentation rate (ESR) and or C-reactive protein (CRP)
- Angiotensin-converting enzyme (ACE)
- Lysozyme
- Non-treponemal and treponemal testing (RPR or VDRL and FTA-ABS)
- Interferon gamma release assay (IGRA)
- Tuberculin skin test
Imaging
- Chest radiograph and/or chest computerized tomography (CT)
- Brain, orbit, and spinal magnetic resonance imaging (MRI)
Ocular imaging
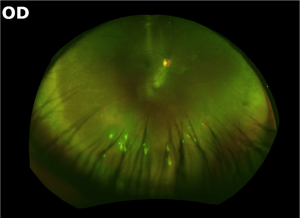
Digital color fundus photography
In patients with IU, fundus photographs allow documentation of baseline appearance of retinal lesions, and helps to assess progression of the condition and response to treatment during follow up. Peripheral retinal pathologies such as snowbanking, retinoschisis, peripheral tractional membranes and macular complications such as macular edema, epiretinal membranes, atrophy and scars can be documented by colour fundus photography.
Optical coherence tomography (OCT)
OCT imaging is very useful in evaluating the macula for CMO, ERM, macular hole ad atrophy. It can be a useful tool for the follow-up of these patients, and allows monitoring the response to treatment. Visual potential and prognostic factors can also be determined by OCT: correlations have been shown between:
- vision and foveal thickness
- IS/OS junction abnormalities and poorer visual prognosis
- CMO with healthy IS/OS junction and better visual prognosis with aggressive treatment.
Intravenous Fluorescein Angiography (IVFA)
IVFA is beneficial to show the activity of retinal vascular inflammation in IU. It allows the detection of CMO, and it helps detecting areas of perivasculitis, areas of non-perfusion, and areas of retinal neovascularization.
Indocyanine green angiography (ICGA)
ICGA has not demonstrated any clinical usefulness in cases of IU
Ultrasonography (US)
US imaging may be helpful in cases where media opacities (band keratopathy, cataract, or significant vitreous inflammation) preclude adequate visualization of the fundus and do not allow for OCT imaging. US at 20- and 50-Hz frequencies can detect snowbanking in IU, and 20-Hz probe may be more useful to detect the presence of CMO.
Ultrasound biomicroscopy
Ultrasound biomicroscopy (UBM) can be used to evaluate patients with IU. It employs higher frequencies than US (35-100 MHz), providing high-resolution images of the pars plana region. It can detect snowbanking, cyclitic membranes, vitreous membranes, and peripheral vitreoretinal tractions.
Diagnostic procedures
Diagnostic vitrectomy
Diagnostic vitrectomy may be necessary in:
- Suspicion of malignancy,
- Cases where retinitis or endophthalmitis cannot be excluded,
- Cases refractory to medical therapy.
Samples can be obtained by vitreous aspiration, vitreous biopsy, or chorioretinal biopsy.
- Vitreous aspiration (0.2-0.5 cc are aspirated using a 23—gauge needle) is associated with more traction at the vitreous base, and increased risk of hypotony and retinal detachment.
- Vitreous biopsy (1 cc of vitreous is collected directly from the vitrectomy bag) is associated with lesser vitreous traction and can be combined with therapeutic vitrectomy.
- Chorioretinal biopsies are highly invasive, and are only indicated in cases where other techniques have failed and disease is bilateral and rapidly progressive. It is usually performed in the worst-seeing eye, and can be done via a scleral flap or by pars plana vitrectomy.
Management
Treatment of IU should be directed at the cause, if one is detected. Malignancy and infection must be ruled out before commencing nonspecific anti-inflammatory therapy.
- Mild vitreous cells in the absence of symptoms or vision loss may be observed. However, little consensus exists as to what threshold to adopt in these cases
- Treat vision-threatening complications (CMO, vitritis) in symptomatic patients with active disease.
Some 25% to 35% of patients with pars planitis have mild disease, no macular edema or other complications, and good vision, and do not need treatment; these patients maintain good vision with up to 10 years of follow-up.
However, when treatment is needed, the goal is complete suppression of the inflammation (i.e., to “grade 0” inflammation).
A stepladder approach has been the most widely adopted strategy in the treatment of IU, with CS therapy being the mainstay of treatment:
Corticosteroids
Periocular CS
Periocular CS (e.g., subtenon injection of 0.5-1.0 mL of triamcinolone acetonide 40 mg/mL)are beneficial in patients with unilateral or asymmetric involvement and in the presence of macular edema. Injections can be repeated every 6 to 8 weeks until the vision and CMO have stabilized; at least 2 or 3 injections are suggested before considering this modality ineffective.
Intravitreal steroids (IVS)
Triamcinolone acetonide (IVTA)
IVTA is associated with high rates of complications, and thus many authors consider it an emergency procedure when essential structures such as the macular have to be rescued immediately, to allow time to organize the long term management.
Slow-release intravitreal implants
Slow-release dexamethasone 0.7 mg intravitreal implant (Ozurdex®; Allergan, Irvine, CA, USA)
Ozurdex has been approved for treatment of intermediate and posterior uveitis, after having shown in case series to significantly improve retinal thickness and visual acuity and to delay and reduce recurrence of uveitic CMO.
Sustained-release fluocinolone acetonide 0.19 mg (Iluvien®, Alimera) or 0.59 mg intravitreal implant Retisert®, Bausch and Lomb)
The MUST trial: compared systemic anti- inflammatory therapy vs. fluocinolone acetonide intraocular implant for intermediate uveitis, posterior uveitis, and panuveitis. No difference was observed in the rates of improvement in macular edema; however, subjects treated with fluocinolone implant had greater quantitative improvement in macular thickness over the follow-up period. A few studies have shown implanted eyes have improved CMO (Pavesio et al, Callanan et al). The use of fluocinolone implants has been limited by the high rate of complications, including cataract formation, ocular hypertension, and hypotony.
Systemic CS
Patients with bilateral disease, severe ocular inflammation, or unilateral disease unresponsive to periocular steroids should be treated systemically, e.g., prednisone 1.0-1.5 mg/ kq body weight/day per os for 4-6 weeks, with gradual tapering according to patient’s response.
Steroid-sparing immunosuppressive therapy
Immunosuppressants are considered a second step in patients for long-term treatment. Indications for commencing systemic immunomodulatory therapy are:
Ocular and anatomical
- Acute, vision-threatening disease
- Chronic, persistent disease
- Exudative RD
- Macular involvement
- Bilateral, vision-threatening disease
Therapeutic failure
Failure of local steroid therapy
Failure of systemic steroid therapy
- Active uveitis with doses of prednisone 30 mg or 0.5 mg/kg/day
- Recurrence of uveitis after tapering oral CS to less than 7-10 mg/day
- Intolerance to oral CS
- Need for steroid-sparing.
The choice of immunosuppressive agent is dependent on the patient’s clinical findings, patient comorbidities, and on the ophthalmologist’s preference and experience.
Methotrexate is the most widely used first‐line immunosuppressive agent in children with chronic non-infectious uveitis because of its long-term safety record and well tolerance. Cyclosporine has also been reported effective. Other drugs used include , mycophenolate mofetil, azathioprine, leflunomide, tacrolimus and rituximab.
Anti-tumor necrosis factor- α (Anti-TNF-α) agents
These class of agents include adalimumab and infliximab. Anti-TNF-α agents may be used successfully as the third step in patients not responding to conventional immunosuppressive agents, although no study exists directly addressing the use of these drugs in non-infectious IU.
However, since pars planitis is associated with increased risk for MS development, Anti-TNF-α agents should be employed with extreme caution, as they may potentiate demyelinating disease.
Interferon α may help improve macular oedema in non-infectious IU, and interferon β has showed success in the treatment of IU-related CMO. However, to date no study has directly compared Interferon α versus interferon β for this indication.
Pars plana vitrectomy (PPV)
PPV is the fourth step of therapy, and has been used in the following clinical scenarios:
- Vitreous condensations
- Vitreous hemorrhage
- Retinal detachment
- ERM causing retinal traction
- Ciliary traction causing hypotony
- Active inflammation and CMO refractory to medical treatment.
Vitrectomy promotes mechanical clearance of inflammatory mediators and debris, and anatomical correction of vitreoretinal traction. In addition, it may be used for diagnostic purposes.
Cryotherapy and laser photocoagulation
Although cryotherapy was used to induce regression of vitreous base neovascularization and to stabilize inflammation, it has now fallen out of favor, as it might aggravate blood-ocular-barrier disruption and possibly accelerate the rate of retinal detachment.
Laser photocoagulation is currently considered as an adjunctive approach to other treatment modalities, especially in cases associated with peripheral neovascularization, retinal traction, or retinoschisis.
Medical follow up
In the acute phase, patients are reevaluated every 1 to 4 weeks, depending on disease severity.
In the chronic phase, reexamination is performed every 3 to 6 months.
Complications
IU can lead to blindness due to complications and permanent damage to ocular structures, especially if the diagnosis is delayed. Children are at high risk of amblyopia.
- Neovascularization in the peripheral retina (6.5%), usually associated with snowbanking
- Neovascularization in the optic nerve head
- Band keratopathy (in up to 45% of eyes)
- Optic disc swelling (3 to 7.4%)
- Cystoid macular oedema (CMO) is a common complication of IU, occurring in up to 50% of patients, and is the major cause of impaired vsion in these patients. The risk of CMO increases with disease severity and duration
- Epiretinal membrane (ERM) formation is common, occurring in approximately 35% of eyes
- Cataract formation in 15-50% of eyes, usually PSC or anterior subcapsular
Cataract surgery in patients with IU should only be performed after inflammation has cleared, ideally with no inflammatory for at least 3 months.
Some authors advocate starting oral prednisone 1 mg/kg body weight/day for 5 days before surgery, with tapering over the following month.
An adequate preoperative control of inflammation, a meticulous surgical technique, a foldable hydrophobic acrylic intraocular lens implanted in the capsular bag and good postoperative inflammation control are crucial for successful cataract surgery in pars planitis patients. A combined pars plana vitrectomy may be considered in cases with significant vitreous opacity.
Studies have shown good clinical outcomes after cataract surgery in patients with IU, with 90% of patients obtaining at least some improvement in vision, and 88% of patients achieving visual acuity above 20/40.
- Uveitic glaucoma occurs in 7.6% of acute uveitis cases and in 11.1% of patients with chronic IU at 5 years
- Retinal detachment (RD, 2.2-51% of eyes): all forms of RD may occur, including exudative RD secondary to inflammation, tractional due to vitreous traction, and rhegmatogenous secondary to vitreous traction leading to peripheral hole formation
- Inferior peripheral retinoschisis is a complication which occurs almost exclusively in children.
Prognosis
The natural history of IU is variable. Although generally considered a benign disease, many cases show a prolonged course with exacerbations, and only 10% of patients may have a self-limited course of disease. In addition, many patients develop complications that may lead to visual loss. Adequate control of inflammation and prompt detection and treatment of associated complications are thus essential in improving the overall prognosis of the patient.
Younger age has been shown to be a significant prognostic factor in IU. Children have worse prognosis than adults, and children under the age of 7 years are more prone to complications and worse visual outcomes than older children.
Despite the disease course and the many complications, IU’s overall prognosis is encouraging, with most patients retaining at least 20/40 visual acuity.
References
1. Brad Bowling. Kanski's Clinical Ophthalmology - A Systematic Approach, 8th edition. Elsevier , 2016.
2. Myron Yanoff, Jay S. Duker. Ophthalmology, 4th edition. Elsevier, 2014.
3. Nika Bagheri, Brynn N. Wajda. Will's Eye Manual - Office and Emergency Room Diagnosis and Treatment of Eye Disease, 7th edition Wolters Kluwer. 2017.
4. Pinar Cakar Ozdal, Nilufer Berker, Ilknur Tugal-Tutkun. Pars Planitis: Epidemiology, Clinical Characteristics, Management and Visual Prognosis. J Ophthalmic Vis Res. 2015 Oct-Dec; 10(4): 469–480. doi: 10.4103/2008-322X.176897
5. Thomas Ness, Daniel Boehringer, Sonja Heinzelmann. Intermediate uveitis: pattern of etiology, complications, treatment and outcome in a tertiary academic center. Orphanet J Rare Dis. 2017; 12: 81. doi: 10.1186/s13023-017-0638-9.
6. B Manohar Babu, S R Rathinam. Intermediate uveitis. Indian J Ophthalmol. 2010 Jan-Feb; 58(1): 21–27. doi: 10.4103/0301-4738.58469.
7. Douglas A Jabs. Immunosuppression for the Uveitides. Ophthalmology. 2018 Feb;125(2):193-202. doi: 10.1016/j.ophtha.2017.08.00.