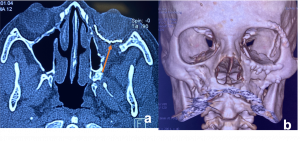
Imaging in Orbit and Oculoplasty
All content on Eyewiki is protected by copyright law and the Terms of Service. This content may not be reproduced, copied, or put into any artificial intelligence program, including large language and generative AI models, without permission from the Academy.
Orbital Anatomy
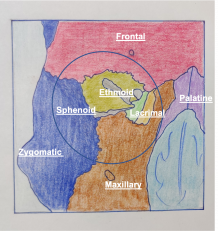
The bony orbit is a quadrangular truncated pyramid situated between the anterior cranial fossa above and maxillary sinus below. Each orbit is formed by seven bones: frontal, ethmoid, lacrimal, palatine, maxillary, zygomatic and sphenoid.[1]
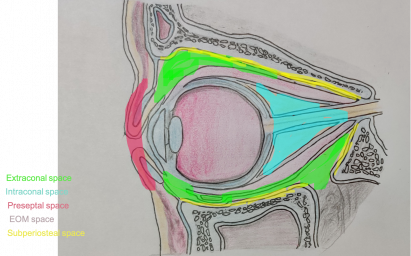
The orbit is divided into various spaces and knowledge of these compartments is required for proper diagnosis of pathologies on imaging and for choosing the best approach to accessing tumors. Due to the characteristics of the types of tissue in each compartment, they have specifications within them. However, sometimes they may extend into more than one compartment. Here, we describe the various spaces and lesions specific to them[2]:
a) Extraconal Space: It is bound peripherally by periorbita, internally by four recti and anteriorly by septum. Common tumors in this space are lymphoma, capillary hemangioma, neoplasms of the lacrimal gland, neurofibromas and metastases. Tumors in this space produce eccentric proptosis, can usually be palpated and best visualized on coronal section.
b) Intraconal Space: It is the space bound by four rectus muscles (muscular cone) and their septum and anteriorly by tenons capsule lining the back of the eye. The optic nerve with sheath lies within it. Many circumscribed orbital tumors such as capillary hemangioma, solitary neurofibroma, varix, and lymphangioma occur in this space and are best studied in coronal planes. Parasagittal planes are used to study the optic nerve but diameter of optic nerve and subarachnoid space are best described in coronal planes. Various optic nerve lesions in this space are optic nerve sheath meningioma, glioma, and optic neuritis and perineuritis.
c) Extraocular muscle (EOM): Enlargement of extraocular muscles should be assessed by evaluating both sides in coronal section. Extraocular muscles are affected in various conditions like thyroid orbitopathy, myositis and tumors such as metastases. To differentiate between them, axial or parasagittal cuts are required to know whether only muscle belly is enlarged or its tendon as well.
d) Lacrimal gland: Best assessed in coronal or axial plane.
e) Subperiosteal space: It is a potential space between orbital bones and periorbita, limited anteriorly by the strong adhesions of periorbita to the orbital rim. Common lesions in this space are dermoid cyst, mucocoele, subperiosteal abscess, myeloma, osteoma, sphenoid wing meningioma and fibrous dysplasia. Based on the location of the lesion all imaging planes are important in this particular space.
Orbital Imaging Techniques
X-Ray
The use of x-ray is limited to patients with trauma or suspected intraorbital foreign body. Standard views include PA view, lateral view and optic foramen view. In a true lateral radiograph, there should be superimposition of the floor of anterior cranial on both sides. For optic foramen and superior oblique fissure, Rheese projections are used.[3]
Ultrasound
It is non-invasive, cost-effective and easy to perform. It is an important first line investigation for evaluating intraocular pathology especially in opaque media. It helps in differentiating cystic from solid lesions. It has two modes A scan and B scan. In normal eyes anterior and posterior chamber are cystic with echogenic lens present in between. Posterior to the globe, fat (echogenic) along with optic nerve sheath complex is seen as a central hypoechoic linear structure. Ultrasound has limited use in deep-seated, orbital lesions due to decreased resolution with depth. Its use is contraindicated in open globes.[4]
Contrast enhanced ultrasound is microbubble-based ultrasound with contrast agents helping in differentiating masses from pseudo masses (RD, haemorrhage).[5]
Color Doppler
Helpful in evaluating blood flow in vascular lesions like vascular malformations, arterial/venous tumours, AVM & CCF.[6]
Computed Tomography (CT scan)
CT is an important modality of orbital imaging. It provides volumetric cross-sectional image acquisitions. It is useful for orbital trauma to assess bony fracture or extraocular muscle entrapment and in intraorbital foreign body localization.[7] It has an advantage over MRI in assessing orbital osseous lesions or soft tissue lesions that cause bony erosion, presence of calcification, and to differentiate acute hemorrhage from mass lesions. It is also useful to evaluate anatomy of extraocular muscles and orbital apex in thyroid eye disease.[8]
Contrast enhanced CT is performed following intravenous administration of iodine rated contrast media like iohexol (omnipaque) & iodixanol (visipaque).[9] It is performed to evaluate different orbital lesions with different enhancement patterns. Enhancement is based on the blood flow and vascular permeability.[10] It is also useful in identifying abscesses in the orbit by portraying a "ring enhancement."
Magnetic Resonance Imaging (MRI)
MRI is based on the magnetic resonance properties of nuclear particles especially hydrogen in human body when placed in a strong external magnetic field. The probes with hydrogen atoms behave as tiny dipoles and get aligned along the direction of magnetic field, majority parallel (low energy state) and remaining few antiparallel high energy state. Following this, radio frequency is applied in which some atoms change directions from parallel to anti-parallel and reaches a state of excitation. When external radio frequency is switched off, they release energy and return to the pre-excited state, a process known as relaxation and is dependent on composition of tissue and environment in which the tissue is situated.[11]
MRI provides higher soft issue resolution and tissue characterization, with better delineation of different orbital components. It helps in the diagnosis of neoplasm, vascular malformation, inflammatory disorders, and optic nerve lesions.
Diffusion Weighted Images (DWI) help in further characterization of orbital masses. It assesses random brownian motion of water molecules in a tissue and thus high cellularity lesions show limited diffusion and low ADC, like lymphomas. It also separates abscess which is more diffusion restricted from other inflammatory processes.[12]
MRI vs CT
MRI is the preferred imaging modality as it provides excellent soft tissue resolution of orbital contents, images the optic nerve along its entire course without any bony shadow and has no exposure of radiation. However, it is costly, requires the patient to lie still for a long time and is an absolute contraindication in metallic intraorbital foreign bodies and cardiac pacemakers.[11]
CT is the ideal modality in bony pathologies, calcification, intraorbital foreign bodies and fracture. It is economical and requires a shorter duration of time. However, it has exposure of radiation specially in cases when required in repeated imaging and in children. It is also less sensitive for soft tissues, optic nerve, and cavernous sinus disorders.[8]
Orbital structure imaging requires slice thickness of 3 or 4mm and inter slice thickness of 0.5 to 1mm.
There are three type of cuts for imaging:
Axial – recognized by presence of both orbits in scan with brain tissue behind
Coronal – recognized by presence of both orbits in the scan with brain tissue above
Sagittal – is the presence of only one orbit at a time with brain tissue above and behind.
Orbital Infections[13]
| Lesion | Age/Sex Predeliction | Clinical Presentation | Imaging Findings | Image |
|---|---|---|---|---|
| Preseptal Cellulitis | More common in < 5 years of age group | Lid edema, erythema | CT- shows stranding, swelling, variable enhancement of the preseptal soft tissues anterior to the globe
|
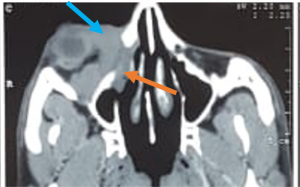
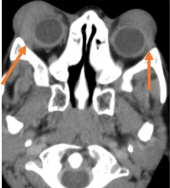
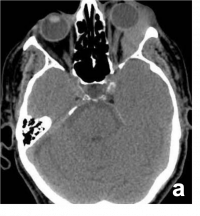
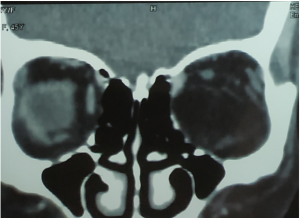
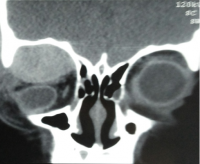
 Sub Periosteal Abscess. Coronal contrast-enhanced CT image of right orbit showing extraconal marginally enhancing fluid collections located superonasally, consistent with subperiosteal abscess. These are present in continuation with opacified right ethmoidal air cells and right maxillary antrum with soft tissue density with hyperdense content. |
| Orbital Cellulitis | More common in > 5 years of age group | Proptosis, chemosis, limitation of EOM, fever | CT- early- eyelid edema and sinusitis
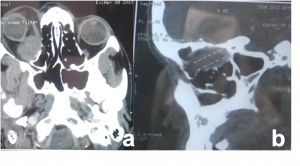
Marked postcontrast enhancement. Inflammation is seen in the medial or superomedial orbit adjacent to the opacified sinus, associated with fat stranding
| |
| Sub periosteal abscess[14] | No age/gender predilection | Collection of subperiosteal fluid causing abaxial proptosis along with other signs of cellutlitis | CT- shows hypodense, convex collection with ring enhancement present along orbital wall
| |
| Invasive Sino-orbital fungal disease[15] | Immunocompromised (mucormycosis)
Imuunocompetent (aspergillosis) |
Severe limitation of EOM with associated chemosis and blepharoptosis.
In mucormysosis, very acute progression of disease with presence of black nasal fliuid, eschar or skin discoloration (characteristic). On fundus examination, CRAO present. In aspergillosis, sub acute progression. |
CT- lobulated soft tissue msses, opacification of paranasal sinuses with high attenuation content with enhancing adjacent orbital soft tissue
T1- low to high intensity T2- marked hypointensity |
Orbital Inflammatory Disease
| Lesion | Age/Sex Predeliction | Clinical Presentation | Imaging Findings | Other features | Image |
|---|---|---|---|---|---|
| Thyroid Eye Disease[16] | Young adults; Female > Male | Axial Proptosis, Redness, watering, lateral flare, lid retraction, photophobia.
Restrictive myopathy |
CT- spindle enlargement of EOMs occurs with relative normal tendinous insertion (coca-cola bottle sign).
MRI - Bulky EOMs are isointense on T1, slightly hyperintense on T2W images and show enhancement. |
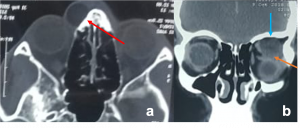
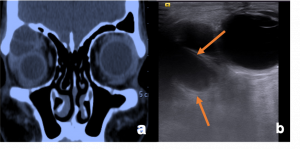
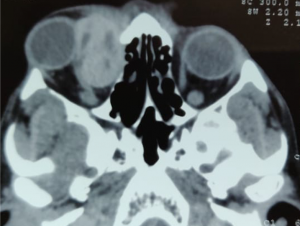
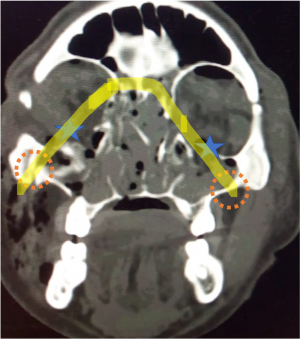
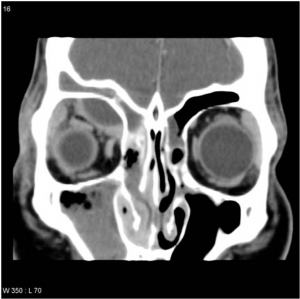
Most frequent involvement IR>MR>SR>LR. Superior Ophthalmic Vein may be enlarged due to apical compression | 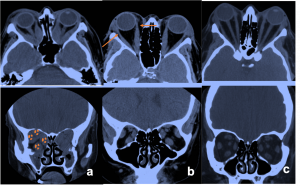 Axial and coronal section of non-contrast CT images of orbit showing three different patterns of TED. a) Bilateral fusiform enlargement of the extraocular muscles sparing the tendinous insertion with posterior crowding (orange stars) b) Bilateral fusiform enlargement of the extraocular muscles sparing the tendinous insertion along with increased orbital fat. c) Bilateral increased orbital fat (denoted by increase of hypodense areas of fat attenuation). EOM appears normal. |
| Myositis[17] | Mean age 30-40 years | Pain, ocular dysmotility, and redness.
EOMs involvment is typically unilateral, painful, and rapid in onset (hours to days). |
CT- diffuse enlargement of EOM involving tendinous insertion with inflammation in the adjacent fat. | ||
| IgG4 Orbital Disease[18] | Mean age 40 - 55 years; Male : Females = 1.3:1 | Painless unilateral or bilateral eyelid swelling, erythema, proptosis, associated dacroadenitis may be present. | CT- nonspecific mass-like inflammation may involve any of the soft tissues of the orbit.
Bilateral, tendon sparing EOM enlargement, preferentially involving the lateral rectus muscles, as well as the lacrimal gland involvement .
|
Most commonly affects lacrimal gland | |
| Wegners Polyangitis[19] | Mean age 65-75 years | Ocular pain, diplopia, decreased visual field with sign suggestive of URTI or lung infection. | Imaging findings depend on orbital location, the extent of involvement and invasion into adjoining structures.
CT - show sinusitis and bony invasion MRI - identifies granulomas and deliniates mucoid plugs. |
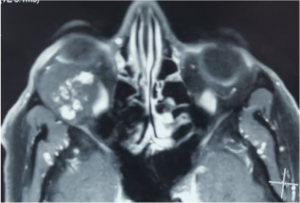
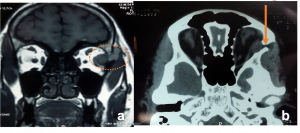
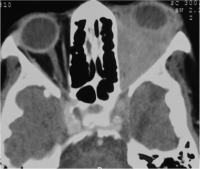 Wegners Granulomatosis Polyangitis. Non-contrast axial CT images in soft tissue window showing left orbital large infiltrative extra- and intra-conal soft tissue masses that mould to the orbital contour associated with proptosis of the globe. Ipsilateral maxillary sinus in the same case showed pansinusitis with medial wall sclerotic thickening. | |
| Sarcoidosis[20] | Bimodal presentation: 20-30 years and 50-60 years | Abaxial proptosis; dacryoadenitis; restrictive myopathy; ocular involement may be present | CT- Bilateral diffuse enhancement and enlargement of the lacrimal glands with infiltration of the adjacent fat.
|
||
| Pseudotumor | Mean age group:40-50 years. | Abrupt onset pain, abaxial proptosis, eyelids swelling, erythema | CT - preferred modality.
Lacrimal gland diffusely enlarged with overall shape preserved. EOM tubular configuration enlargement with tendon involving. (MR > SR> LR>IR). Optic Nerve - non-enhanced optic nerve surrounded by inflammation (tramline sign). Cavernous sinus - if involved best seen on MRI |
Tumors/Lesion
Extraocular
Extraocular lesions can be either transcompartmental, intraconal, or extraconal.
Transcompartmental
Such lesions occupying both extraconal and intraconal space. They can be benign or malignant.
Benign
Vascular
| Lesion | Age/Sex Predeliction | Clinical Presentation | Imaging Findings | Other Features | Image |
|---|---|---|---|---|---|
| Infantile Haemangioma[21] | Present at birth during infancy, grows with the child say up to 5–7 years age, and disappear by 10 years | Soft, non-tender, non-pulsatile ill-defined mass. Cutaneous discoloration or leash of vessels on the lids, periocular soft tissues.
Commonly located anterior to the globe in the eyelid. |
USG Doppler- Hyperechoic.
T1- intermediate signal T2- hyperintense Proliferative phase intense early enhancement with multiple flow voids (characteristic) Involuting phase - heterogeneous with fibro-fatty tissue, decreased vascularity and less prominent enhancement. |
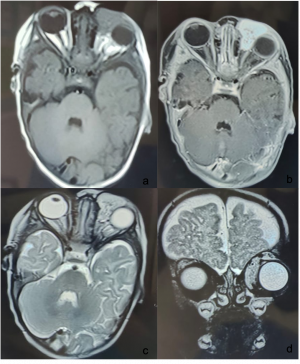 Infantile Haemangioma. a & b) CE MRI (T1 weighted) of brain and orbit- Axial section showing well-defined lesion involving the left orbit with no intraconal extension, involving the medial rectus muscle. It shows heterogeneous enhancement with contrast. c & d) MRI brain and orbit (T2 weighted)- Axial and coronal section, the lesion shows hyperintensity. | |
| Lymphangioma/ Venolymphatic Malformation[22] | Children
(>50% are diagnosed at birth). |
Painless progressive long standing proptosis in a child, often sudden increase following URTI or haemorrhage
On examination, irregular heterogeneous masses, which are poorly defined |
USG- to evaluate its vascularity, and to separate its cystic and solid composition. Macrocysts appear as anechoic area separated by septae. Microcysts appear hyperechoic.
|
Larger lesions may cause bone remodelling with extension into preseptal or infratemporal fossa through the inferior orbital fissure.
Presence of fluid-fluid levels produced by haemorrhages (chocolate cyst) of various ages is pathognomonic. |
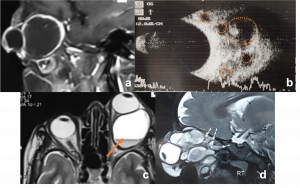 Lymphangioma. a,c Macrocystic lymphatic malformation. Sagittal T1 WI of left orbit showing large cyst with hypointense signal and Axial T2 WI MRI showing large cyst with fluid-fluid levels (haemorrhagic content hyperintense on T2 signal (orange arrow) with septa of hypointense fluid) b) USG orbit demonstrates MIXED variant lymphatic malformation, with multiple cyst (orange circles) d) Saggital T2 WI MRI of orbit showing multiple cyst with hyperintesne signal (microcyst marked with white arrow) |
| Varices[23] | 2nd and 3rd decade , M=F | Painless, progressive proptosis that increases on stooping forward/ Valsalva.
Usually located in posterior orbit. |
USG - for dynamic assessment.
It appears as intermittently anechoic lesion which on straining distends with flow. CT- irregular, segmentally dilated or smooth, variably enhancing lesion, which significantly increases in size with straining. It is useful to detect bony abnormality associated with it. Calcification may be seen. MRI- On T1 hypo to hyperintense T2 - hyperintense. |
||
| AV Malformations[24] | Post traumatic, no age/ gender predilection | Pulsatile mass in orbit with pulsatile proptosis. eyelid and conjunctival congestion present with bruit heard.
Usually found in the superior orbit. |
CT and MRI helps in diagnosis and its extent.
CT Angiography - important for treatment planning. They show enlargement and rapid filling of the proximal arterial system with early drainage into distal venous outflow. |
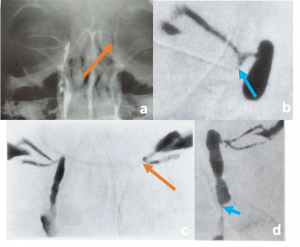
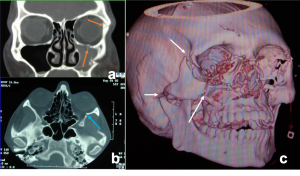
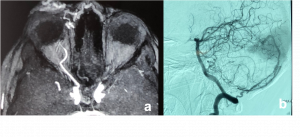 Orbital and Eyelid Arterio-Venous Malformation a) Magnetic Resonance Angiography TRICKS (Time-Resolved Imaging of Contrast KineticS) confirms mid and delayed arterial phase blushing of orbital and eyelid mass with dense contrast. b) Digital Subtraction (DSA) images of right orbit showing large dural AVM with the engorged external carotid artery and its branches and early venous drainage. | |
| Carotid-Cavernous Fistula[25] | No age/ gender predilection | Direct – sudden pulsatile exophthalmos, conjunctival chemosis, with dilated episcleral vessels and bruit.
|
USG Doppler – shows increased velocity with reversal of blood flow direction, dilated Superior Ophthalmic Vein (SOV) and arterial pulsations.
CT/MRI - proptosis, EOM enlargement, dilated and tortuous SOV, with engorgement and early enhancement of the ipsilateral cavernous sinus. CT Angiography- differentiate direct and indirect CCF but cerebral angiography is used for accurate diagnosis and intervention. |
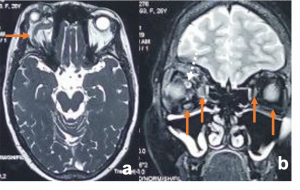 Traumatic carotid-cavernous fistula) Axial T2WI MRI of right orbit showing dilated superior ophthalmic vein along its whole course (orange arrow) b) Coronal T2WI MRI showing dilated superior ophthalmic vein (white dotted arrow) with engorgement of the extra ocular muscles compared to the contralateral normal side (orange arrows) |
Mesenchymal
| Lesion | Age/Sex Predeliction | Clinical Presentation | Imaging Findings | Other Features | Image | |
|---|---|---|---|---|---|---|
| Rhabdomyosarcoma[26] | Mean age - 8 to 10 years, M>F | Proptosis or upper lid mass with blepharoptosis
Pain only in advanced cases. Usually present in superonasal quadrant. |
CT- irregular, homogenous well-defined soft tissue extraconal masses isodense to EOM, but may be heterogenous due to focal haemorrhage. CT is important for bony involvement.
T1- isointense to muscle T2-hyperintense with areas of high T1 signal secondary to hemorrhage and necrosis. It shows mild to moderate uniform enhancement. MRI is important for aggressive tumours to know their intracranial extension and invasiveness in adjacent paranasal sinuses. |
The tumor may conform to adjacent bony walls and orbital structures such as the globe. | ||
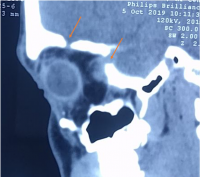
| Neuroblastoma[27] | Mean age of presentation 2 years | Proptosis (may be pulsatile) and swelling of lid (unilateral/ bilateral raccoon eyes). Abdominal features.
Can have ecchymosis, Horner’s syndrome. Usually involves Posterolateral orbital wall. |
CT - metastases appear as either circumscribed or ill defined, increased attenuation to muscle, may have small calcifications. It can invade adjacent structures.
T1- hypointesne T2- heterogeneous due to hemorrhage or necrosis, and heterogeneously enhance post contrast. |
Systemic imaging including CT/MRI abdomen and/or chest. MRI spine for paraspinal tumors | 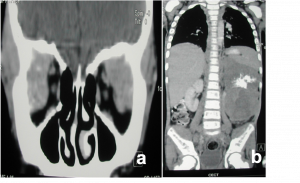 Metastatic Neuroblastoma a) Coronal contrast-enhanced CT of the right orbit demonstrates bilateral transcompartmental, heterogeneously enhancing ill-defined soft tissue masses with proptosis of both the globes. b) Coronal CT Abdomen demonstrates a heterogeneously enhancing, centrally necrotic, partially calcified mass arising from the left adrenal gland, associated with multiple adjacent enlarged retroperitoneal lymph nodes. | alt= |
| Lacrimal Gland Tumors [28](Adenocystic Carcinoma/ Mucoepidermoid carcinoma) | Middle aged adults | Ocular pain, inferonasal dystopia, abaxial proptosis | CT - heterogeneous, irregular, infiltrative mass with poorly demarcated margins. Bony erosion and intrinsic calcification is common. It shows marked and focal enhancement. | |||
| Lymphoproliferative disorders[29]
--Lymphoma (most common), lymphoid hyperplasia and atypical lymphoid hyperplasia, and ocular adnexal lymphoma. |
Elderly | Minimal or no pain and with gradual progression. Usually bilateral. | CT- homogenous hyperdense lacrimal gland mass. Bony destruction is usually absent
MRI - T1- isointense T2-hypointense to muscle. Marked enhancement post contrast. |
Intraconal
Benign
Vascular
| Lesion | Age/Sex Predeliction | Clinical Presentation | Imaging Findings | Image |
|---|---|---|---|---|
| Cavernous Malformation/ Solitary Encapsulated Venolymphatic malformation
(SEVLM)[30] |
Middle age, Female | Painless, progressive proptosis well-defined (pathologically encapsulated), oval to round | USG- well circumscribed lesion, medium to high internal reflectivity and absent internal flow.
MRI- hypointense relative to muscle on T1, hyperintense on T2. |
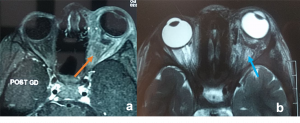
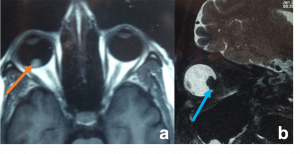
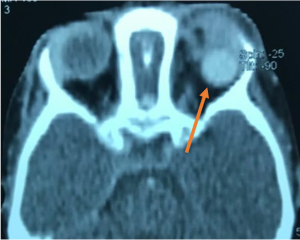
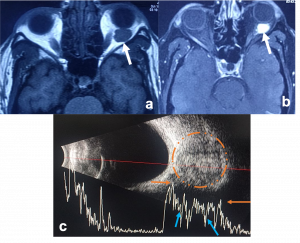 Cavernous Malformation. a) Axial T1WI MRI shows a left intraconal ovoid, well encapsulated soft tissue mass that indents the posterior aspect of the eye globe associated with mild proptosis. b)Post contrast axial T1WI with fat suppression demonstrates progressive heterogeneous bright enhancement (white arrow) of the lesion c) USG orbit (B/A) showing a well encapsulated mass on B scan (orange circle) with corresponding internal acoustic reflections on A scan showing characteristic dancing spikes demonstrating rise and fall of waves as it traverses through multiple blood-filled channels within the tumor (blue arrows) |
Neural
| Type | Age/Sex Predeliction | Clinical Presentation | Imaging Findings | Other features | Image |
|---|---|---|---|---|---|
| Optic Nerve Glioma[31] | 1st decade | Decreased visual activity, visual field defect, proptosis, and relative afferent pupillary defect | The optic nerve cannot be seen separate from the mass; hence, the nerve appears tubular, tortuous, and kinked.
NF associated Gliomas- On CT - hypodense and poorly-defined masses. Minimal or no enhancement is seen . Sporadic Gliomas- solid cystic appearance.
|
NF-1 glioma- bilateral optic nerve gliomas with intracranial involvement (diffuse enlargement of the chasm).
Sporadic- unilateral fusiform enlargement of the optic nerve. Intracranially, there is chiasmatic-hypothalamic mass with central cystic component. |
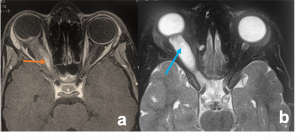 Optic Nerve Glioma. a) Axial T1WI MR images demonstrate right optic nerve globular shaped mass of hypointense T1 and b) hyperintense T2 signal with mild post-contrast enhancement (orange and blue arrows). The optic nerve cannot be made out separately. Note the presence of a small area of cystic degeneration inside the lesion (blue arrow). |
| Optic Nerve Sheath Meningioma[32] | 5th decade, F>M | Insidious vision loss over months to years.
|
CT- smooth tubular thickening of the ON - iso to hyperdense and show calcification in 20–50% of cases. There is marked and homogeneous contrast enhancement.
T2 - heterogeneous and varies from slightly hypointense to slightly hyperintense. |
Contrast-enhanced T1W images with fat suppression demonstrate a diffuse tubular enhancing lesion around the optic nerve with characteristic tram track sign on axial images and target sign/doughnut on coronal images | |
| Optic Neuritis[33] | Papilitis
(children) Retrobulbar neuritis (young adults) |
Unilateral rapid onset of vision loss. Eye pain worsen with movements. | CT has no role
MRI - increased signal and bulk of the nerve |
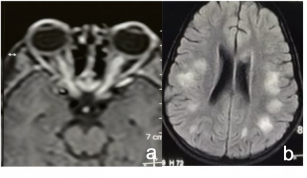
Evaluate MRI brain for periventricular plaques |
Extraconal
Benign
Vascular
Lesions in this space may include cavernous haemangioma, venolymphatic malformations, capillary haemangiomas.
Neural
| Lesion | Age/Sex Predeliction | Clinical Presentation | Imaging Findings | Other features |
|---|---|---|---|---|
| Schwannoma[34] | 20-40 years mean age group | Painless, progressive proptosis with diplopia | Schwannomas have a heterogeneous T2 signal and variable patterns of enhancement with homogeneous or ring enhancement being most common. | Orbital schwannomas can show delayed homogeneous enhancement like cavernous malformations, but do not follow the centripetal enhancement pattern of the latter. |
Mesenchymal
| Lesion | Age/Sex Predeliction | Clinical Presentation | Imaging Findings | Other features | Image | |
|---|---|---|---|---|---|---|
| Dermoid Cyst[35] | Child or young adult | Round to oval well defined slow growing mass near the frontozygomatic/frontoethmoidal suture.
Intraorbital dermoid often cause proptosis with globe dystopia. |
USG- capsulated well circumscribed lesions with variable internal reflectivity due to areas of calcification within.
CT- They have cystic center with areas of fat within and appear hypodense. A fat–fluid level and calcified rim may be present. Denser foci within the lesion represent flecks of keratin and sebum. MRI- Lesion tends to be heterogeneously hyperintense on T2WI and hypointense on T1WI. |
External – superotemporal
Internal – supero nasal Orbital - superotemporal |
||
| Lipoma[36] | Any age group | Mostly asymptomatic untill large. It arises mainly from the anterior orbit. | On CT scan, typically lipoma appears as distinctive low attenuation lesion sometimes with a finely defined border. On MRI, it is hyperintense on T1 weighted image. However, it does not enhance after contrast. | alt= | ||
| Hydatid Cyst[37] | Any age group | Abaxial Proptosis. Chemosis, lid edema, restriction of EOM, visual impairment | Well defined, thin walled, oval lesion with peripheral rim enhancement of their capsule.
T1 - hypointense T2 - Hyperintense USG - double layer sign, water lily sign. MRI spectroscopy can be performed |
Lacrimal gland tumor
| Lesion | Age/Sex Predeliction | Clinical Presentation | Imaging Findings | Other features | Image |
|---|---|---|---|---|---|
| Pleomorphic Adenoma[38][38] | 40-55 years | Indolent painless enlargement of the gland causing proptosis and characteristic S-shaped ptosis | CT- well circumscribed homogenous mass in lacrimal fossa with bony scalloping.
MRI- bright enhancement. T1- hypo/iso intense T2- hyperintense |
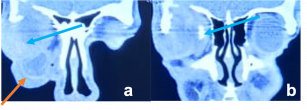
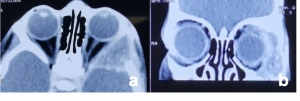
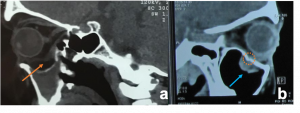
Bone remodelling may be present but no bony erosion. | 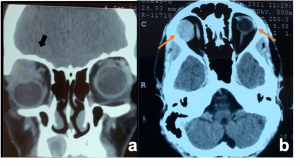 Pleomorphic Adenoma. a & b). Non-contrast CT scan demonstrates well-defined, round to oval lesions, smooth in outline, associated with prominent scalloped lacrimal fossa (orange curved line) formation due to pressure without bone erosion in comparison to left orbit (orange arrows). Long-standing large tumours lobulations and radiolucent areas of cystic degeneration. |
Bony lesions
| Lesion | Age/Sex Predeliction | Clinical Presentation | Imaging Findings | Other Features | Image |
|---|---|---|---|---|---|
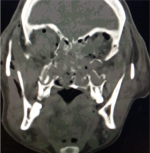
| Langerhans Cell Histiocytosis[39] | Early childhood | Abaxial proptosis, bony defect with orbital infiltration of lesion. Usually present at superotemporal orbit/sphenoid wing | CT- soft tissue homogenous mass with hyperdense margin.
MRI- T1 intermediate signal, T2- hypointense relative to bone marrow fat. |
File:LCH.jpg | |
| Sphenoid wing Menigioma[40] | 50-60 years; Caucassian Female | Depends on location of primary tumor. Orbital mass/temporal mass/sella tursica. | CT- intraosseous part appears as bone hyperostosis and expansion
MRI- there is increase in the dark signal of the bone (extremely hypointense on T2). |
Meningiomas of the greater wing of sphenoid cause intraosseous growth, which may cause narrowing of intraorbital foramina. | File:Figure 5 SWM.jpg |
| Fibrous cell dysplasia[41] | Less than 30 years age | Non-specific | CT, characteristic expansile bony lesion with ground glass density is seen.
T1 isointense T2 hypointense because of sclerosis and fibrous stroma. |
Ossifying fibroma mimics them on CT, but they have borders better defined than FD. | |
| Dumbell dermoid cyst[42] | 10-30 years | C chewing oscillops (characteristic sign) iawhi h is caused by a synkinetic movement of the temporallis muscle associated with pursing of the lips during chewing. | CT scan demonstrates a mass lesion that straddles the lateral orbital wall (mostly). A bony channel in the lateral orbital wall connects the two lobes. The deep lobe displaces many of the lateral intraorbital structures. |
Malignant
| Lesion | Age/Sex Predeliction | Clinical Presentation | Imaging Findings | Other Features | Image |
|---|---|---|---|---|---|
|
Haemangiopericytoma/ Solitary Fibrous Tumor (SFT)[43] |
No age/sex predeliction | Proptosis, painless. Extraconal location most commonly along the paranasal sinuses. Extraconal location most commonly along the paranasal sinuses | CT- homogeneous to heterogeneous, rounded or elongated masses of moderate density, smooth, well circumscribed. Calcification in 25% cases. Enhancement is moderate to marked.
MRI - round to oval tumour, well defined borders. T1 - the signal is isointense to muscle, and hypointense to fat. T2 - the lesion is hyperintense to fat. |
Dynamic CT may show prominent early enhancement with rapid washout. |
Intraocular
Ultrasound should be the first-line radiologic modality for the evaluation of suspected intraocular masses, and MRI later.
| Lesion | Age/Sex Predeliction | Clinical Presentation | Imaging Findings | Other features | Image |
|---|---|---|---|---|---|
| Retinoblastoma[44] | Children | White reflex or strabismus. Most common intraocular tumor of childhood. | USG and MRI to distinguish RB from other causes of white reflex.
USG- evaluate calcification
CT- Radiation risk |
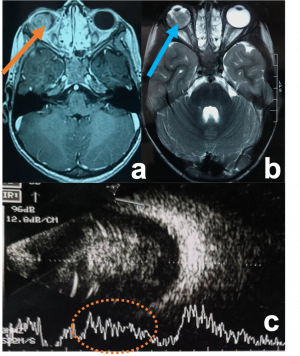 Retinoblastoma. a) Contrast-enhanced T1WI axial MRI image showing an enhancing right intraocular mass with no extraocular extension (orange arrow) b) T2WI axial MRI image showing hypointense signal (blue arrow) c) USG (B/A) showing intraocular mass lesion with specks of calcification corresponding to the spikes of A-scan. | |
| Uveal Melanoma[45] | Middle aged adults | Dimunition of vision, assoicated with exudative retinal detachment. | MRI is the investigation of choice.
T1 hyperintensity T2- hypointense It enhances brightly on contrast |
Lacrimal System
Dacrocystography (DCG)
For anatomic evaluation of the lacrimal system. It tells us about the exact level of obstruction or stenosis and the presence of diverticulum, fistula, stones, or tumours. CT-DCG show adjacent bony structures also. During the procedure, dye is injected into bilateral lacrimal systems and magnified images then are obtained.[46] There are two types :
Macro DCG
X-Ray based. An erect scan delayed by 5 minutes post injection of contrast is taken.
Micro DCG
Computed tomography (CT-DCG)/magnetic resonance imaging (MRI-DCG) scan based.
Dacryoscintigraphy (DSG)
For physiologic evaluation of the lacrimal system. It is a noninvasive test. Radiolabelled Tc99m is instilled into conjunctival cul-de-sac, while a modified gamma camera records images over 20 minutes. It is used for identification of the location of a partial or functional block, such as conjunctivochalasis or punctal occlusion.[47]
Orbital Trauma[47]
Fractures[48]
Foreign body[49]
Imaging should be done in cases with wooden foreign body to know the exact size, shape and location of the foreign body. Intra-orbital wooden foreign body is not visible on X-ray orbit due to its radiolucent property. Diagnosis of intraorbital wooden foreign body on B-scan ultrasonography needs expertise, and very deep foreign body may be missed. MRI-scan is the imaging modality of choice for the diagnosis of intraorbital wooden foreign body. Intra-orbital wooden foreign body can be seen better in MRI scan because density of protons and their relaxation time in the wood are different from the orbital soft tissue. The wooden foreign body appears hypo-intense as compared to the orbital fat on T1 and T2. The appearance of intra-orbital wooden foreign body on CT-scan varies with the time according to the degree of the hydration. In the acute stage, a wooden foreign body is seen as a continuous linear track of air because its density is similar to the air. In the sub-acute stage, because of the increased hydration, the density of wooden foreign body becomes similar to the orbital fat, making the diagnosis difficult. In chronic stages, its density is more than the orbital fat.[49]
Orbital Hemorrhage[50]
| Age/Sex Predeliction | Clinical Presentation | Location | Imaging Findings | Image |
|---|---|---|---|---|
| Non specific; M> F (more trauma in males) | Acute onset proptosis, with ecchymosis and limitaion of EOM. | Retrobular, subperiosteal | On CT, hemorrhage is identified as an ill-defined high attenuation of the soft tissues.
|
Imaging after Oculoplastic Surgery
At times, post operative imaging is also required for confirming the adequate position of implants.[51] This may be done for post orbital fracture repairs with inadequate cosmesis to look for residual enophthalmos, post enucleation for a migrated impant, post surgery for anophthalmic socket with tissue expander and post surgery for lid retraction with metallic weights.
References
- ↑ Turvey TA, Golden BA. Orbital anatomy for the surgeon. Oral Maxillofac Surg Clin North Am. 2012;24(4):525-536. doi:10.1016/j.coms.2012.08.003
- ↑ Lemke AJ, Kazi I, Felix R. Magnetic resonance imaging of orbital tumors. Eur Radiol 2006;16:2207-19.
- ↑ Bray LC, Griffiths PG . The value of plain radiography in suspected intraocular foreign body. Eye 1991; 5: 751–754.
- ↑ Bryden FM, Pyott AA, Bailey M, McGhee CN . Real time ultrasound in the assessment of intraocular bodies. Eye 1990; 4: 727–731.
- ↑ D'Arrigo JS, Mano Y. Bubble production in agarose gels subjected to different decompression schedules. Undersea Biomed Res. 1979 Mar;6(1):93-8. PMID: 462654.
- ↑ Mulligan PR, Prajapati HJ, Martin LG, Patel TH. Vascular anomalies: classification, imaging characteristics and implications for interventional radiology treatment approaches. Br J Radiol. 2014;87(1035):20130392. doi:10.1259/bjr.20130392
- ↑ Boyette JR, Pemberton JD, Bonilla-Velez J. Management of orbital fractures: challenges and solutions. Clin Ophthalmol. 2015;9:2127-2137. Published 2015 Nov 17. doi:10.2147/OPTH.S80463
- ↑ Jump up to: 8.0 8.1 Khan SN, Sepahdari AR. Orbital masses: CT and MRI of common vascular lesions, benign tumors, and malignancies. Saudi J Ophthalmol. 2012;26(4):373-383. doi:10.1016/j.sjopt.2012.08.001
- ↑ Pannu HK, Thompson RE, Phelps J, Magee CA, Fishman EK. Optimal contrast agents for vascular imaging on computed tomography: iodixanol versus iohexol. Acad Radiol. 2005 May;12(5):576-84. doi: 10.1016/j.acra.2005.01.015. PMID: 15866130.
- ↑ Tawfik HA, Abdelhalim A, Elkafrawy MH. Computed tomography of the orbit - A review and an update. Saudi J Ophthalmol. 2012;26(4):409-418. doi:10.1016/j.sjopt.2012.07.004
- ↑ Jump up to: 11.0 11.1 Ettl AR, Salomonowitz E, Koornneef L. Magnetic resonance imaging of the orbit: Basic principles and anatomy. Orbit. 2000 Dec;19(4):211-237. doi: 10.1076/orbi.19.4.211.2649. PMID: 12045939.
- ↑ Baliyan V, Das CJ, Sharma R, Gupta AK. Diffusion weighted imaging: Technique and applications. World J Radiol. 2016;8(9):785-798. doi:10.4329/wjr.v8.i9.785
- ↑ Ali R. Sepahdari, Vinay K. Aakalu, Rashmi Kapur, Edward A. Michals, Nitu Saran, Adam French, and Mahmood F. Mafee American Journal of Roentgenology 2009 193:3, W244-W250
- ↑ Tabarino F, Elmaleh-Bergès M, Quesnel S, Lorrot M, Van Den Abbeele T, Teissier N. Subperiosteal orbital abscess: volumetric criteria for surgical drainage. Int J Pediatr Otorhinolaryngol. 2015 Feb;79(2):131-5. doi: 10.1016/j.ijporl.2014.11.021. Epub 2014 Nov 25. PMID: 25529362.
- ↑ Imaging Features of Invasive and Noninvasive Fungal Sinusitis: A Review.Manohar Aribandi, Victor A. McCoy, and Carlos Bazan, III RadioGraphics. 2007 27:5, 1283-1296
- ↑ Gonçalves AC, Gebrim EM, Monteiro ML. Imaging studies for diagnosing Graves' orbitopathy and dysthyroid optic neuropathy. Clinics (Sao Paulo). 2012;67(11):1327-1334. doi:10.6061/clinics/2012(11)18
- ↑ Ferreira TA, Saraiva P, Genders SW, Buchem MV, Luyten GPM, Beenakker JW. CT and MR imaging of orbital inflammation. Neuroradiology. 2018;60(12):1253-1266. doi:10.1007/s00234-018-2103-4
- ↑ Immunoglobulin G4–Related Disease of the Orbit: Imaging Features in 27 Patients.C.A. Tiegs-Heiden, L.J. Eckel, C.H. Hunt,et al. American Journal of Neuroradiology Jul 2014, 35 (7) 1393-1397; DOI: 10.3174/ajnr.A3865
- ↑ Muller K, Lin JH. Orbital granulomatosis with polyangiitis (Wegener granulomatosis): clinical and pathologic findings. Arch Pathol Lab Med. 2014;138(8):1110-1114. doi:10.5858/arpa.2013-0006-RS
- ↑ Shaikh ZA, Bakshi R, Greenberg SJ, Fine EJ, Shatla A, Lincoff NS. Orbital involvement as the initial manifestation of sarcoidosis: magnetic resonance imaging findings. J Neuroimaging. 2000 Jul;10(3):180-3. doi: 10.1111/jon2000103180. PMID: 10918747.
- ↑ Pujari A, Bajaj MS, Obedulla H, Mutha V. Acquired capillary haemangioma of the eyelid in a 10-year-old boy. BMJ Case Rep. 2017;2017:bcr2017221102. Published 2017 Jul 31. doi:10.1136/bcr-2017-221102
- ↑ Woo YJ, Kim CY, Sgrignoli B, Yoon JS. Orbital Lymphangioma: Characteristics and Treatment Outcomes of 12 Cases. Korean J Ophthalmol. 2017;31(3):194-201. doi:10.3341/kjo.2016.0034
- ↑ Pappas A, Araque JM, Sarup V. Orbital Venous Varices: A Rare Bilateral Asymptomatic Presentation. Cureus. 2018;10(9):e3302. Published 2018 Sep 14. doi:10.7759/cureus.3302
- ↑ Warrier S, Prabhakaran VC, Valenzuela A, Sullivan TJ, Davis G, Selva D. Orbital Arteriovenous Malformations. Arch Ophthalmol. 2008;126(12):1669–1675. doi:10.1001/archophthalmol.2008.501
- ↑ Dos Santos D, Monsignore LM, Nakiri GS, Cruz AA, Colli BO, Abud DG. Imaging diagnosis of dural and direct cavernous carotid fistulae. Radiol Bras. 2014;47(4):251-255. doi:10.1590/0100-3984.2013.1799
- ↑ Mu X, Wang H, Li Y, Hao Y, Wu C, Ma L. Magnetic resonance imaging and DWI features of orbital rhabdomyosarcoma. Eye Sci. 2014 Mar;29(1):6-11. PMID: 26016059.
- ↑ Rao AA, Naheedy JH, Chen JY, Robbins SL, Ramkumar HL. A clinical update and radiologic review of pediatric orbital and ocular tumors. J Oncol. 2013;2013:975908. doi:10.1155/2013/975908
- ↑ Qin W, Chong R, Huang X, Liu M, Yin ZQ. Adenoid cystic carcinoma of the lacrimal gland: CT and MRI findings. Eur J Ophthalmol. 2012 May-Jun;22(3):316-9. doi: 10.5301/ejo.5000015. PMID: 21725943.
- ↑ Priego G, Majos C, Climent F, Muntane A. Orbital lymphoma: imaging features and differential diagnosis. Insights Imaging. 2012;3(4):337-344. doi:10.1007/s13244-012-0156-1
- ↑ Xian J, Zhang Z, Wang Z, et al. Evaluation of MR imaging findings differentiating cavernous haemangiomas from schwannomas in the orbit. Eur Radiol. 2010;20(9):2221-2228. doi:10.1007/s00330-010-1774-y
- ↑ Huang M, Patel J, Patel BC. Optic Nerve Glioma. [Updated 2021 May 1]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2021 Jan-. Available from: https://www.ncbi.nlm.nih.gov/books/NBK557878/
- ↑ Parker RT, Ovens CA, Fraser CL, Samarawickrama C. Optic nerve sheath meningiomas: prevalence, impact, and management strategies. Eye Brain. 2018;10:85-99. Published 2018 Oct 24. doi:10.2147/EB.S144345
- ↑ Gala F. Magnetic resonance imaging of optic nerve. Indian J Radiol Imaging. 2015;25(4):421-438. doi:10.4103/0971-3026.169462
- ↑ Kim KS, Jung JW, Yoon KC, Kwon YJ, Hwang JH, Lee SY. Schwannoma of the Orbit. Arch Craniofac Surg. 2015;16(2):67-72. doi:10.7181/acfs.2015.16.2.67
- ↑ Nugent RA, Lapointe JS, Rootman J, Robertson WD, Graeb DA. Orbital dermoids: features on CT. Radiology. 1987 Nov;165(2):475-8. doi: 10.1148/radiology.165.2.3659368. PMID: 3659368.
- ↑ Shah NB, Chang WY, White VA, Heran MK, Rootman J. Orbital lipoma: 2 cases and review of literature. Ophthalmic Plast Reconstr Surg. 2007 May-Jun;23(3):202-5. doi: 10.1097/IOP.0b013e318050ca52. PMID: 17519657.
- ↑ Betharia SM, Sharma V, Pushker N. Ultrasound findings in orbital hydatid cysts. Am J Ophthalmol. 2003 Apr;135(4):568-70. doi: 10.1016/s0002-9394(02)02083-4. PMID: 12654390.
- ↑ Jump up to: 38.0 38.1 Misra S, Bhandari A, Misra N, Gogri P, Mahajan S. Pleomorphic adenoma of a deep orbital ectopic lacrimal gland. Orbit. 2016 Oct;35(5):295-7. doi: 10.1080/01676830.2016.1193526. Epub 2016 Aug 10. PMID: 27541944.
- ↑ Herwig MC, Wojno T, Zhang Q, Grossniklaus HE. Langerhans cell histiocytosis of the orbit: five clinicopathologic cases and review of the literature. Surv Ophthalmol. 2013 Jul-Aug;58(4):330-40. doi: 10.1016/j.survophthal.2012.09.004. Epub 2012 Dec 13. PMID: 23246282; PMCID: PMC4100607.
- ↑ Westendorff C, Kaminsky J, Ernemann U, Reinert S, Hoffmann J. Image-guided sphenoid wing meningioma resection and simultaneous computer-assisted cranio-orbital reconstruction: technical case report. Neurosurgery. 2007 Feb;60(2 Suppl 1):ONSE173-4; discussion ONSE174. doi: 10.1227/01.NEU.0000249235.97612.52. PMID: 17297354.
- ↑ Kushchayeva YS, Kushchayev SV, Glushko TY, et al. Fibrous dysplasia for radiologists: beyond ground glass bone matrix. Insights Imaging. 2018;9(6):1035-1056. doi:10.1007/s13244-018-0666-6
- ↑ Maeng MM, Godfrey KJ, Kazim M. Multilobular dermoid cyst of the orbit. Orbit. 2019 Aug;38(4):328-330. doi: 10.1080/01676830.2018.1509098. Epub 2018 Sep 14. PMID: 30215530.
- ↑ Kikuchi K, Kowada M, Sageshima M. Orbital hemangiopericytoma: CT, MR, and angiographic findings. Comput Med Imaging Graph. 1994 May-Jun;18(3):217-22. doi: 10.1016/0895-6111(94)90033-7. PMID: 8025890.
- ↑ Razek AA, Elkhamary S. MRI of retinoblastoma. Br J Radiol. 2011;84(1005):775-784. doi:10.1259/bjr/32022497
- ↑ Ferreira TA, Grech Fonk L, Jaarsma-Coes MG, van Haren GGR, Marinkovic M, Beenakker JM. MRI of Uveal Melanoma. Cancers (Basel). 2019;11(3):377. Published 2019 Mar 17. doi:10.3390/cancers11030377
- ↑ Udhay P, Noronha OV, Mohan RE. Helical computed tomographic dacryocystography and its role in the diagnosis and management of lacrimal drainage system blocks and medial canthal masses. Indian J Ophthalmol. 2008;56(1):31-37. doi:10.4103/0301-4738.37593
- ↑ Jump up to: 47.0 47.1 Gokhale SM. Dacryoscintigraphy: A Pictorial Essay. Indian J Nucl Med. 2018;33(3):214-217. doi:10.4103/ijnm.IJNM_18_18
- ↑ Caranci F, Cicala D, Cappabianca S, Briganti F, Brunese L, Fonio P. Orbital fractures: role of imaging. Semin Ultrasound CT MR. 2012 Oct;33(5):385-91. doi: 10.1053/j.sult.2012.06.007. PMID: 22964404.
- ↑ Jump up to: 49.0 49.1 Al-Mujaini A, Al-Senawi R, Ganesh A, Al-Zuhaibi S, Al-Dhuhli H. Intraorbital foreign body: clinical presentation, radiological appearance and management. Sultan Qaboos Univ Med J. 2008;8(1):69-74.
- ↑ Atalla ML, McNab AA, Sullivan TJ, Sloan B. Nontraumatic subperiosteal orbital hemorrhage. Ophthalmology. 2001 Jan;108(1):183-9. doi: 10.1016/s0161-6420(00)00482-6. PMID: 11150286.
- ↑ Reiter MJ, Schwope RB, Kini JA, York GE, Suhr AW. Postoperative imaging of the orbital contents. Radiographics. 2015 Jan-Feb;35(1):221-34. doi: 10.1148/rg.351140008. PMID: 25590399.