Drug Induced Maculopathy
All content on Eyewiki is protected by copyright law and the Terms of Service. This content may not be reproduced, copied, or put into any artificial intelligence program, including large language and generative AI models, without permission from the Academy.
Retinal toxicity or abnormality can be induced by usage of various medications. In the majority of instances, toxicity is reversible following discontinuation of the inciting drug. However, permanent or progressive vision loss may occur in a few instances even despite drug cessation. This page will review the toxicities most likely to be encountered in common clinical practice.
Patterns of retinal toxicity
Retinal drug toxicities can manifest in a variety of patterns. This table presents a brief overview of drug-induced retinal toxicities and the pattern of injury typically associated with each drug.
| Disruption of the retina and retinal pigment epithelium |
Vascular damage | Cystoid Macular Edema | Crystalline retinopathy | Uveitis | Miscellaneous |
|---|---|---|---|---|---|
|
|
|
|
|
|
Adapted from Zhu I, Andreoli MT, Mittra RA, et al. Drug Toxicity of the Posterior Segment. Ryan Retina.
Disruption of retina and pigment epithelium
Drugs can be directly toxic to the cells of the retina or the retinal pigment epithelium (RPE). The most common manifestation of injury to the retina and RPE is a pigmentary maculopathy. Medications that can induce a pigmentary maculopathy include chloroquine derivatives (chloroquine, hydroxychloroquine), phenothiazines (thioridazine, chlorpromazine), pentosan polysulfate sodium, clofazimine, dideoxyinosine, deferoxamine, ritonavir, alkylating agents (cisplatin, BCNU), potassium or sodium iodate, and denileuken diftitiox. Other forms of toxicity can also include serous retinal detachments (MEK inhibitors, FGFR inhibitors) and retinal edema and atrophy (quinine).
Chloroquine derivatives
Chloroquine and its derivative, hydroxychloroquine sulfate, were initially used as malarial prophylaxis. Today, they are more commonly used to treat autoimmune conditions such as rheumatoid arthritis or systemic lupus erythematosus. Prolonged use of the chloroquine derivatives typically results in a pigmentary maculopathy.
The exact mechanism of chloroquine derivative toxicity is unknown, though they are known to have an affinity for binding to melanin and concentrating in the RPE and uvea.[1] Various mechanisms have been proposed including inhibition of retinol recycling,[2] disruption of lysosomal function,[3][4][5] and primary retinal toxicity.[6]
Symptoms
Early toxicity may be asymptomatic. Symptoms that may eventually develop include paracentral scotoma, decreased visual acuity, metamorphopsia, and photophobia.
Signs
The earliest signs of toxicity involve loss of the foveal light reflex with subtle macular stippling. The classic presentation is a bull's eye-maculopathy appearance where the fovea is surrounded by a ring of depigmentation followed by a ring of hyperpigmentation. In the advanced stages of toxicity, the pigment abnormalities can involve the peripheral retina with a clinical picture that resembles primary tapetoretinal degeneration with optic disc pallor, retinal vessel attenuation, and bone spicules. Visual acuity is affected when the pigmentary abnormalities involve the center of the fovea.
Other ocular changes may involve the cornea, lens, and uvea.
Diagnostic testing
No single diagnostic test can reliably detect retinopathy. The imaging changes induced by chloroquine derivatives are nonspecific and often a multimodal imaging approach is utilized with the results interpreted in the appropriate clinical context.
- Fundus autofluorescence (FAF): Highly sensitive; early RPE changes may manifest as a paracentral ring of hyperautofluorescence while more advanced RPE changes appear hypoautofluorescent. Wide-field FAF may be helpful in detecting more peripheral RPE changes, especially in Asian patients who are more likely to have peripheral involvement.
- Spectral-domain optical coherence tomography (SD-OCT): Discontinuity of the inner segment/outer segment junction, thinning of the outer nuclear layer, and hyperscattering of the outer segment may be present. Rapid retinal thinning which can be quantified using sequential macular thickness maps appear to be an even earlier sign of toxicity.[7]
- Fluorescein angiography (FA): Pigment abnormalities may appear hyperfluorescent due to window defects.
- Electroretinogram (ERG)/Electro-oculogram (EOG): The ERG and EOG may be abnormal in early cases of photoreceptor (b-wave) or pigment toxicity. The multifocal ERG (mf-ERG) may be abnormal early when other tests such as visual field, full-field ERG (ff-ERG), and OCT are normal.
- Perimetry: Quantifies degree of visual field loss and can be used for screening and monitoring for progression.

Histopathologic changes
Loss of RPE pigmentation with accumulation of pigment-laden cells in the outer retinal layers and damage and reduction of photoreceptors.[9] Electron microscopic studies have also revealed damage to the retina, especially the ganglion cell layer.[10]
Prevalence and risk factors
Overall prevalence has been calculated around 7.5%, according to one study, though this varies with duration of use and daily dose.[11] Additional risk factors for toxicity include co-existing renal dysfunction and concurrent use of tamoxifen.
Recommendations for screening

The American Academy of Ophthalmology released updated guidelines for screening in 2016.[8] The most notable updates included recommendations for screening the extramacular region of Asian patients and revision of the recommended daily doses based on real body weight.
- Pattern of Retinopathy: Often parafoveal in most eyes, though Asian patients often show an extramacular pattern of damage.
- Dose: Maximum daily dose of 5.0 mg/kg real body weight for HCQ. Dose comparisons of CQ in older literature suggest a maximum daily dose of 2.3 mg/kg real body weight.
- Risk of Toxicity: The risk of toxicity is dependent on daily dose and duration of use. At recommended doses, the risk of toxicity up to 5 years is under 1% and up to 10 years is under 2%, but it rises to almost 20% after 20 years. However, even after 20 years, a patient without toxicity has only a 4% risk of converting in the subsequent year.
 Clinical Examination Techniques. Reused, with permission, from American Academy of Ophthalmology.[13]
Clinical Examination Techniques. Reused, with permission, from American Academy of Ophthalmology.[13] - Major Risk Factors: High dose and long duration of use are the most significant risks. Other major factors are concomitant renal disease or use of tamoxifen.
- Screening Schedule: A baseline fundus examination should be performed to rule out preexisting maculopathy with the frequency of ocular examinations afterwards determined by concomitant eye conditions. After 5 years, patients on acceptable doses and without major risk factors should begin annual screening.
- Screening Tests: The primary screening tests are automated visual fields and SD-OCT. These should look beyond the central macula in Asian patients. mfERG can provide objective corroboration for visual fields and FAF can show damage topographically. Modern screening should detect retinopathy before it is visible in the fundus.
- Toxicity: Retinopathy is not reversible and there is no present therapy. Recognition at an early stage (before any significant RPE loss) is important to prevent central visual loss. Questionable test results should be repeated or validated with additional procedures to avoid unnecessary cessation of valuable medication.
- Counseling: Patients (and prescribing physicians) should be informed about risk of toxicity, proper dose levels, and the importance of regular annual screening.
Phenothiazines
Thioridazine and chlorpromazine are two agents in the phenothiazine drug class used as antipsychotics to treat schizophrenia and other psychiatric conditions. The exact mechanism of toxicity is unknown, though it may involve enyzme disruption resulting in abnormal rhodopsin synthesis.[14][15] Thioridazine has a piperidyl side chain and is much more likely to cause toxicity than chlorpromazine.
Symptoms
Symptoms include blurred vision, dyschromatopsia, nyctalopia, and visual field scotomas depending on disease severity.
Signs
- Early: Normal or mild granular pigment stippling.
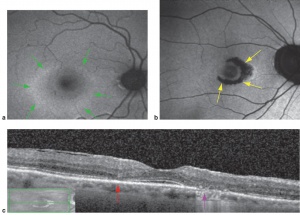
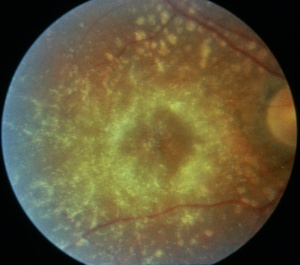
- Intermediate: Characteristic circumscribed nummular areas of RPE loss from the posterior pole to the midperiphery. (See figure.)
- Late: Widespread areas of depigmentation, hyperpigmented plaques, vascular attenuation, and optic atrophy.
The early fundus changes often progress despite discontinuation of the drug. Chlorpromazine binds strongly to melanin and can also cause hyperpigmentation in the skin, conjunctiva, cornea, and lens. Other ocular effects include oculogyric crisis, miosis, and cycloplegia.
Diagnostic testing
- FA: Disruption of choriocapillaris in zones of pigment disruption.
- ERG/EOG: ERG can show decreased oscillatory potentials in early disease. ERG/EOG are both markedly abnormal in advanced disease.
Histopathology
Atrophy and disorganization of photoreceptor outer segments with secondary loss of RPE and choriocapillaris.[16]
Risk factors
The risk of retinopathy associated with use of the phenothiazine drug class is more dependent on total daily dose rather than cumulative amount of drug received. Thioridazine toxicity at dosages less than 800 mg/day is rare, though cases have been reported with lower doses over several years. At higher doses, toxicity can occur rapidly, even within weeks. Regardless of the dose, patients taking thioridazine should be regularly monitored for signs of toxicity.
Chlorpromazine retinal toxicity is rare and typically only occurs when massive doses are given (i.e., >2400 mg/day).
Pentosan polysulfate sodium (PPS)
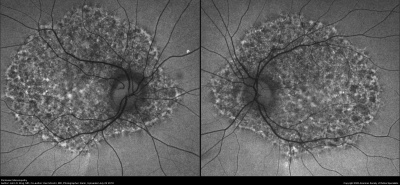
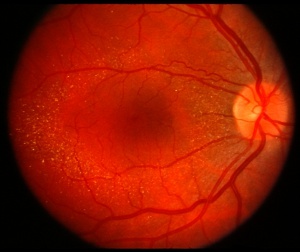
PPS is a medication mainly used for the treatment of interstitial cystitis that was recently found to be associated with a pigmentary maculopathy. Toxicity is typically reported after chronic use of the drug (>15 years).[17] Women are more commonly affected due to the increased prevalence of interstitial cystitis compared to men. Common symptoms include blurred vision, difficulty reading, metamorphopsia, paracentral scotomas, and prolonged dark adaptation. Signs include parafoveal pigmented deposits at the level of the RPE, vitelliform deposits, and patchy paracentral RPE atrophy similar in appearance to pattern dystrophy. The maculopathy appears to progress even after discontinuation of the drug.[18][19][20]
Diagnostic testing
- FAF: Speckled hyperautofluorescence in the posterior pole with peripapillary extension. Hyperfluorescent spots correspond to vitelliform lesions noted on clinical exam.[21]
- Near-infrared reflectance (NIR) imaging: Punctate areas of hyperreflectance corresponding to hyperpigmented areas noted on clinical exam.
- SD-OCT: Focal nodular RPE excrescences and late RPE atrophy with outer retinal thinning.
- ERG: ff-ERG may be normal given the localized maculopathy. mf-ERG demonstrates variable mild to moderate attenuation in cone- and rod-derived responses.
- Perimetry: Can reveal functional deficits ranging from decreased foveal thresholds to large paracentral scotomas.
Risk factors
The risk of toxicity with PPS rises significantly in patients exposed to greater than 1500 g.[22] The average reported duration of therapy at time of presentation ranged from 15 to 20 years and average cumulative dose ranged from 2263 to 3375.4 g. Some experts have recommended screening to include an initial baseline exam with OCT, NIR, FAF imaging, and annual follow-up, though no consensus guidelines have been published.
MEK Inhibitors
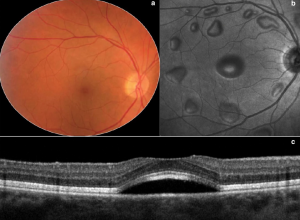
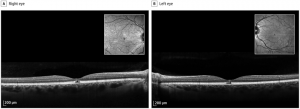
Mitogen-activated protein kinase (MEK) inhibitors are a novel class of chemotherapeutic agents used to treat metastatic melanoma by inhibiting the MEK enzyme. They are often used in conjunction with BRAF inhibitors. Trametinib (Mekinist), cobimetinib (Cotellic), binimetinib (Mektovi), and selumetinib (Koselugo) are currently FDA approved drugs within this class. The most commonly described ocular manifestation associated with MEK inhibitors is bilateral multifocal serous retinal detachment with at least one focus involving the fovea.[23] The mechanism is thought to be due to RPE induced dysfunction with subsequent accumulation of subretinal fluid.
SD-OCT is extremely helpful in detailing the extent and location of the serous detachments. The fluid characteristics seen on SD-OCT can be described as dome, caterpillar, wavy, and splitting. These features can be helpful in distinguishing the MEK inhibitor associated serous detachments from central serous chorioretinopathy (CSR). In addition, MEK inhibitor associated serous detachments do not show any evidence of choroidal abnormalities, focal RPE defects, or leakage on FAF, FA, or SD-OCT as would be expected in CSR.
Onset can occur days to weeks after initiation of therapy. Visual symptoms are typically minimal with the fluid often spontaneously resolving. For persistent cases, discontinuation of the drug usually leads to complete resolution.
FGFR Inhibitors
Fibroblast growth factor receptor (FGFR) inhibitors are another novel class of chemotherapeutic agents used to treat cholangiocarcinoma and urothelial carcinoma. Currently FDA approved drugs within this class include erdafitinib (Balserva), pemigatinib (Pemazyre), and infigratinib (Truseltiq). FGFR inhibitors act upstream of the MEK kinase and, therefore, appear to cause serous retinal detachments, similar to MEK retinopathy.
Few reports are available in the literature of FGFR retinopathy. One case report described an asymptomatic patient on pemigatinib with a multifocal serous retinopathy similar to MEK retinopathy.[24] In this case, the drug was stopped with near complete resolution after 5 days. A second case report described thickening of the central macular interdigitation and ellipsoid zones on OCT a few weeks after starting erdafitinib that evolved into bilateral subfoveal serous fluid 3 months later.[25] The drug was continued as the patient remained asymptomatic with good vision. Another sub-study of patients in a clinical trial of another FGFR inhibitor, AZD4547, reported multilobular subretinal fluid pockets in 5 of 23 (22%) eyes.[26] A case report of pseudovitelliform subfoveal lesions with subretinal fluid in a patient using erdafitinib has also been reported that developed within 1 week of starting the drug.[27] The fluid regressed spontaneously even with continuing the drug. No reports are available on the long-term effects of FGFR retinopathy.
A study of 339 patients receiving mitogen-activated pathway kinase (MAPK) inhibitors compared the effects of direct interference of MAPK with MEK or extracellular signal-regulated kinase (ERK) inhibitors vs. indirect inhibition with FGFR inhibitors.[28] Of these patients, 128 eyes of 65 patients had evidence of retinopathy on OCT (31% on ERK inhibitors, 23% on MEK inhibitors, and 12% on FGFR inhibitors) with >70% of patients on MEK and ERK inhibitors having multifocal foci of subretinal fluid compared to 2.5% on FGFR inhibitors. Intraretinal edema was seen in 45% of eyes on ERK inhibitors, 2% on MEK inhibitors, and none on FGFR inhibitors. In all cases, the findings were self-limited, did not require medical intervention, and did not cause irreverisible vision loss.
Vascular damage
Various agents can cause toxicity by damaging the retinal vasculature with either direct particle clogging of blood vessels, induction of a hypercoagulable state, or both. Drugs that damage the retinal vasculature include alkylating agents (cisplatin, BCNU), talc, oral contraceptives, intravitreal aminoglycosides, intracameral vancomycin, interferon, tacrolimus, gemcitabine, methamphetamine/cocaine, ergot alkaloids, and over-the-counter phenylpropranolamine.
Talc
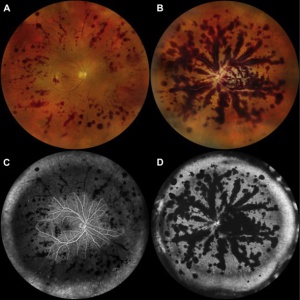
Talc retinopathy typically occurs in intravenous drug abusers who crush oral medications that contain talc as an inert filler and inject it intravenously.[29] The talc particles lodge in the pulmonary vasculature and eventually pass to retinal end arterioles. Glistening crystals located in the end arterioles is the typical appearance. An ischemic retinopathy may develop with capillary nonperfusion, microaneurysm formation, cotton wool spots, venous loops, and eventual neovascularization may develop.
Aminoglycoside antibiotics (intraocular)
Intravitreal injection of aminoglycoside antibiotics is extremely toxic to the posterior segment. Gentamicin is the most toxic aminoglycoside followed by tobramycin and amikacin. The fundus will show intraretinal hemorrhages, retinal edema, cotton wool spots, arteriolar narrowing, venous beading, and severe retinal infarction. Pathology shows granulocyte clogging of retinal vasculature. OCT will acutely show inner retinal edema with subsequent thinning.
Factors that can worsen the toxicity include an intravitreal injection with the needle bevel pointed towards the posterior pole and increased rate of injection.[30][31] Patients who undergo inadvertent injection should undergo immediate pars plana vitrectomy and lavage. After surgery, they should be placed in an upright position to prevent gravity from directing remaining drug molecules to the posterior segment.
Vancomycin (intraocular)
Intracameral vancomycin has been reported to cause a hemorrhagic occlusive retinal vasculitis after use as endophthalmitis prophylaxis at the end of cataract surgery. The mechanism is thought to be related to a rare type IV hypersensitivity reaction causing damage to the vascular endothelium approximately a week after administration.[32] Clinical examination will show intraretinal hemorrhage, vascular occlusion, and retinal ischemia. Prognosis is often poor though early recognition and treatment may lead to better outcomes. Treatment includes high-dose corticosteroids, antiviral medication, anti-VEGF injections, and pars plana vitrectomy.
Interferon
Interferon is used in the treatment of various malignancies. Systemic therapy has been associated with cotton wool spots, intraretinal hemorrhages, venous occlusion, arterial occlusion, CME, and ischemic optic neuropathy.[33] The exact mechanism of toxicity is unknown but thought to involve impaired retinal microcirculation. Visual acuity is usually unaffected if only cotton wool spots and retinal hemorrhages are present. Changes typically present 4-8 weeks after initiation of therapy and usually resolve after treatment cessation. Patients with underlying conditions such as diabetes or hypertension were at higher risk for retinopathy.
Cystoid macular edema (CME)
Various agents can cause cystoid macular edema including topical epinephrine, nicotinic acid, topical prostaglandin analogs (e.g., latanoprost), antimicrotubule agents (paclitaxel, docetaxel), fingolimod, imatinib, glitazones (rosiglitazone, pioglitazone), and trastuzumab. Notably, the CME seen with nicotinic acid and the antimicrotubule agents paclitaxel and doxetaxel do not leak on FA. In most cases, the CME will resolve with discontinuation of the medication. In cases where the CME is refractory or the inciting agent cannot be discontinued, various treatments can be trialed for CME including topical anti-inflammatory agents, sub-Tenon or intravitreal corticosteroids, and topical or oral carbonic anhydrase inhibitors.
Crystalline retinopathy
Talc
See previous section.
Tamoxifen
Tamoxifen is an anti-estrogen agent used to treat certain types of cancer. Retinal toxicity consists of inner retinal crystalline deposits, macular edema, and punctate pigmentary changes. Corneal toxicity may include vortex keratopathy. Visual acuity and color vision may be decreased. Ancillary testing may be helpful in making the diagnosis and distinguishing this entity from macular telangiectasia type 2, which can appear similar to tamoxifen retinopathy in its early stages.
- OCT: hyperreflective punctate lesions in the inner retina with or without CME; in early stages, may show a foveal cyst similar to those seen in macular telangiectasia type 2
- FA: late focal staining in macula consistent with CME
- FAF: normal or macular hypofluorescence with foveal hyperfluorescence
- ERG: decreased photopic and scotopic a- and b-wave amplitudes
- EOG: may show decreased Arden ratio
Toxicity typically manifests after 2-3 years though it depends on the daily dose.[34] High doses (60-100 mg/day) may manifest as early as 1 year after initiation of treatment while lower doses (10-20 mg/day) may take years to manifest. Patients with high BMI and dyslipidemia are at higher risk to develop tamoxifen retinopathy.
Visual function and macular edema typically improve after drug cessation, though the crystalline deposits remain. Persistent macular edema can be treated with intravitreal anti-VEGF therapy, intravitreal corticosteroids, or oral carbonic anhydrase inhibitors.
Canthaxanthine
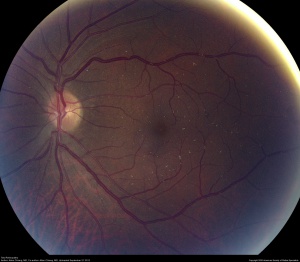
Canthaxanthine is a naturally occurring carotenoid that can be used as a food-coloring agent, treatment for vitiligo/photosensitivity disorders, and tanning agent. At high doses (total dose >19 g over 2 years), a ring-shaped deposit of superficial yellow-orange crystals has been described.[35] While the crystals are only clinically observed in the posterior segment, histopathology has shown the crystals in the entire inner retina and ciliary body. Most patients are typically asymptomatic with a normal FA. With discontinuation of treatment, the crystals may clear slowly over 20 years.
Methoxyflurane
Methoxyflurane is an inhaled anesthetic. If used for extended periods of time, calcium oxalate crystals can deposit in the kidneys and other tissues. These yellow-white punctate crystals are found in the posterior pole and periarterially.[36] Histologically, they are found in the inner retina and RPE.
Uveitis
Checkpoint inhibitors
Checkpoint inhibitors are a targeted form of cancer immunotherapy aimed at activating the immune system against cancer cells.[37] They currently include ipilimumab, pembrolizumab, nivolumab, cemiplimab, atezolizumab, avelumab, durvalumab. The majority of ocular side effects are related to induced autoimmune reactions. The most common ocular side effects include dry eye syndrome and myasthenia gravis-like ocular myopathy. In the posterior segment, the most common manifestation is uveitis, typically anterior with or without CME though posterior uveitis, panuveitis, retinal vasculitis, serous retinal detachments, and others have also been described. Treatment can involve topical, local, or systemic corticosteroid therapy depending on the severity of inflammation. The patient should be managed closely with an oncologist.
BRAF inhibitors
BRAF inhibitors are small molecule inhibitors that target the BRAF protein and are used in the treatment of metastatic cutaneous melanoma. They can be used in conjunction with MEK inhibitors. The most commonly reported side effect is an anterior uveitis, though panuveitis and Vogt-Koyanagi-Harada (VKH)-like uveitis have also been reported.[38] Treatment of inflammation includes topical, local, or systemic corticosteroids. If vision loss is severe and uncontrolled, the drug can be stopped.
Rifabutin
Rifabutin is used to treat and prevent disseminated Mycobacterium avium-complex infection. A minority of patients treated with higher doses (300 to >450 mg/day) can develop uveitis 2 weeks to 14 months after treatment initiation.[39][40] The uveitis is usually bilateral, anterior, and can cause a hypopyon, simulating infectious endophthalmitis. Systemic fluconazole, clarithromycin, and protease inhibitors may potentiate toxicity due to their inhibition of rifabutin metabolism through the CYP450 system. Treatment can include corticosteroids, drug dose adjustment, or drug discontinuation.
Cidofovir
Cidofovir is a nucleotide analogue that inhibits viral DNA polymerase and is used in the treatment of CMV retinitis. Intravenous and intravitreal cidofovir have been associated with anterior uveitis, hypotony, and vision loss.[41][42] Patients with low CD4 counts and higher levels of viremia on antiretroviral therapy are at increased risk for developing cidofovir-associated uveitis. It has also been associated with an increased risk of immune recovery uveitis. Treatment is targeted at controlling inflammation and preventing hypotony, including topical corticosteroids, cycloplegics, ibopamine, and oral probenecid.
Miscellaneous
Sulfa derivatives
Sulfa derivatives can cause an idiosyncratic reaction involving ciliary body swelling, choroidal effusion, or both with subsequent anterior movement of the lens-iris diaphragm and resultant myopia and retinal folds.[43] Retinal folds are typically seen in young patients. Symptoms usually resolve after discontinuation of the inciting medication. Medications in this drug class include sulfa antibiotics, acetazolamide, chlorthalidone, disothiazide, ethoxyzolamide, hydrochlorothiazide, metronidazole, sulphonamide, topiramate, and triamterene.
Alkyl nitrites
Alkyl nitrites are a group of volatile inhaled substances of abuse colloquially known as "poppers." Most identified cases involve the compound isopropyl nitrite.[44] Toxicity is thought to result from massive nitric oxide release that causes photoreceptor damage.
Patients can present with decreased central visual acuity, central scotoma, metamorphopsia, and phosphenes with the clinical examination ranging from normal to yellow foveal lesions. SD-OCT is extremely helpful in diagnosis and can reveal subfoveal damage ranging from IS/OS junction disruption to vitelliform-like lesions to IS/OS microhole defect. The appearance can be similar to that resulting from photic damage. Partial, and sometimes complete, resolution of injury is possible after drug abuse cessation.
ALK inhibitors
Anaplastic lymphoma kinase (ALK) inhibitors are a new form of chemotherapy that inhibit the ALK enzyme found in non-small cell lung carcinoma. They include crizotinib (Xalkori), ceritinib (Zykadia), alectinib (Alecensa), brigatinib (Alunbrig), lorlatinib (Lorbrena), entrectinib (Rozlytrek). Early clinical trial data showed that the most common side effect was impaired light/dark adaptation with visual phenomena that improved with the length of time receiving therapy.[45]
Additional Resources
References
- ↑ Ramsey MS, Fine BS. Chloroquine toxicity in the human eye. Histopathologic observations by electron microscopy. American journal of ophthalmology. 1972;73(2):229-235.
- ↑ Xu C, Zhu L, Chan T, et al. Chloroquine and Hydroxychloroquine Are Novel Inhibitors of Human Organic Anion Transporting Polypeptide 1A2. J Pharm Sci. 2016;105(2):884-890.
- ↑ Ehrenfeld M, Nesher R, Merin S. Delayed-onset chloroquine retinopathy. The British journal of ophthalmology. 1986;70(4):281-283.
- ↑ Mahon GJ, Anderson HR, Gardiner TA. Chloroquine causes lysosomal dysfunction in neural retina and implications for retinopathy. Curr Eye Res. 2003;28:277-284.
- ↑ Ivanina TA, Zueva MV, Lebedeva MN, Bogoslovsky AI, Bunin AJ. Ultrastructural alterations in rat and cat retina and pigment epithelium induced by chloroquine. Graefes Arch Clin Exp Ophthalmol. 1983;220(1):32-38.
- ↑ Rosenthal AR, Kolb H, Bergsma D, Huxsoll D, Hopkins JL. Chloroquine retinopathy in the rhesus monkey. Invest Ophthalmol Vis Sci. 1978;17(12):1158-1175.
- ↑ Melles RB, Marmor MF. Rapid Macular Thinning Is an Early Indicator of Hydroxychloroquine Retinal Toxicity. Ophthalmology. 2022 Sep;129(9):1004-1013. doi: 10.1016/j.ophtha.2022.05.002. Epub 2022 May 11. PMID: 35568277.
- ↑ 8.0 8.1 Recommendations on Screening for Chloroquine and Hydroxychloroquine Retinopathy, Revised 2016. The Ophthalmic News and Education Network, American Academy of Ophthalmology. https://www.aao.org/clinical-statement/revised-recommendations-on-screening-chloroquine-h. Accessed December 17, 2018.
- ↑ Wetterholm DH, Winter FC. Histopathology of chloroquine retinal toxicity. Arch Ophthalmol. 1964;71:82-87.
- ↑ Ramsey MS, Fine BS. Chloroquine toxicity in the human eye. Histopathologic observations by electron microscopy. American journal of ophthalmology. 1972;73(2):229-235.
- ↑ Melles RB, Marmor MF. The risk of toxic retinopathy in patients on long-term hydroxychloroquine therapy. JAMA Ophthalmol. 2014;132(12):1453-1460.
- ↑ Recommendations on Screening for Chloroquine and Hydroxychloroquine Retinopathy, Revised 2016. The Ophthalmic News and Education Network, American Academy of Ophthalmology. https://www.aao.org/clinical-statement/revised-recommendations-on-screening-chloroquine-h. Accessed December 17, 2018.
- ↑ Recommendations on Screening for Chloroquine and Hydroxychloroquine Retinopathy, Revised 2016. The Ophthalmic News and Education Network, American Academy of Ophthalmology. https://www.aao.org/clinical-statement/revised-recommendations-on-screening-chloroquine-h. Accessed December 17, 2018.
- ↑ Bonting SL, Caravaggio LL, Canady MR. Studies on sodium potassium-activated adenosine triphosphatase. X Occurrence in retinal rods and relation to rhodopsin Exp Eye Res. 1964;3 SRC - GoogleScholar:47-56.
- ↑ Cerletti A, Taeschler M, Weidmann H. Pharmacologic studies on the structure-activity relationship of hydroxyindole alkylamines. Advances in pharmacology. 1968;6(Pt B):233-246.
- ↑ Miller FS, Bunt-Milam AH, Kalina RE. Clinical-ultrastructural study of thioridazine retinopathy. Ophthalmology. 1982;89(12):1478-1488.
- ↑ Wang D, Au A, Gunnemann F, et al. Pentosan-associated maculopathy: prevalence, screening guidelines, and spectrum of findings based on prospective multimodal analysis. Can J Ophthalmol. 2020
- ↑ Jung EH, Lindeke-Myers A, Jain N. Two-Year Outcomes After Variable Duration of Drug Cessation in Patients With Maculopathy Associated With Pentosan Polysulfate Use. JAMA Ophthalmol. 2023 Mar 1;141(3):260-266. doi: 10.1001/jamaophthalmol.2022.6093. PMID: 36729449; PMCID: PMC9896370.
- ↑ Somisetty S, Santina A, Au A, Romero-Morales V, Bousquet E, Sarraf D. Progression of Pentosan Polysulfate Sodium Maculopathy in a Prospective Cohort. Am J Ophthalmol. 2023 Nov;255:57-67. doi: 10.1016/j.ajo.2023.05.021. Epub 2023 Jun 15. PMID: 37327961.
- ↑ Hall BP, Shiromani S, Jung EH, Lyons RJ, Tribe J, Jain N. Pentosan Polysulfate Maculopathy: Final Outcomes from a 4-Year Prospective Study of Disease Progression after Drug Cessation. Am J Ophthalmol. 2024 Dec 10:S0002-9394(24)00562-2. doi: 10.1016/j.ajo.2024.12.004. Epub ahead of print. PMID: 39667696.
- ↑ Hanif AM, Armenti ST, Taylor SC, et al. Phenotypic Spectrum of Pentosan Polysulfate Sodium-Associated Maculopathy: A Multicenter Study. JAMA Ophthalmol. 2019.
- ↑ Hadad A, Helmy O, Leeman S, Schaal S. A Novel Multimethod Image Analysis to Quantify Pentosan Polysulfate Sodium Retinal Toxicity. Ophthalmology. 2019.
- ↑ Francis JH, Habib LA, Abramson DH, et al. Clinical and Morphologic Characteristics of MEK Inhibitor-Associated Retinopathy: Differences from Central Serous Chorioretinopathy. Ophthalmology. 2017;124(12):1788-1798.
- ↑ Alekseev O, Ojuok E, Cousins S. Multifocal serous retinopathy with pemigatinib therapy for metastatic colon adenocarcinoma. Int J Retina Vitreous. 2021 Apr 23;7(1):34. doi: 10.1186/s40942-021-00305-9. PMID: 33892812; PMCID: PMC8067404.
- ↑ Parikh D, Eliott D, Kim LA. Fibroblast Growth Factor Receptor Inhibitor-Associated Retinopathy. JAMA Ophthalmol. 2020 Oct 1;138(10):1101-1103. doi: 10.1001/jamaophthalmol.2020.2778. PMID: 32789485.
- ↑ Charng J, Attia MS, Arunachalam S, Lam WS, Creaney J, Muruganandan S, Read C, Millward M, Spiro J, Chakera A, Lee YCG, Nowak AK, Chen FK. Increased interdigitation zone visibility on optical coherence tomography following systemic fibroblast growth factor receptor 1-3 tyrosine kinase inhibitor anticancer therapy. Clin Exp Ophthalmol. 2021 Aug;49(6):579-590. doi: 10.1111/ceo.13940. Epub 2021 May 26. PMID: 33934469.
- ↑ Ramtohul P, Denis D, Comet A. Pseudovitelliform Maculopathy Associated with FGFR Inhibitor Therapy. Ophthalmol Retina. 2021 Feb;5(2):140. doi: 10.1016/j.oret.2020.10.005. PMID: 33549201.
- ↑ Francis JH, Foulsham W, Canestraro J, Harding JJ, Diamond EL, Drilon A, Abramson DH. Mitogen-Activated Pathway Kinase Inhibitor-Associated Retinopathy: Do Features Differ with Upstream versus Downstream Inhibition? Ocul Oncol Pathol. 2023 Aug;9(1-2):25-31. doi: 10.1159/000529127. Epub 2023 Feb 8. PMID: 38376085; PMCID: PMC10821790.
- ↑ AtLee WE. Talc and cornstarch emboli in eyes of drug abusers. JAMA. 1972;219(1):49-51.
- ↑ Peyman GA, Vastine DW, Crouch ER. Clinical use of intravitreal antibiotics to treat bacterial endophthalmitis. Trans Am Acad Ophthalmol Otolaryngol. 1974;78:862-875.
- ↑ Zachary IG, Forster RK. Experimental intravitreal gentamicin. American journal of ophthalmology. 1976;82(4):604-611.
- ↑ Witkin AJ, Chang DF, Jumper JM, et al. Vancomycin-Associated Hemorrhagic Occlusive Retinal Vasculitis: Clinical Characteristics of 36 Eyes. Ophthalmology. 2017;124(5):583-595.
- ↑ Raza A, Mittal S, Sood GK. Interferon-associated retinopathy during the treatment of chronic hepatitis C: a systematic review. J Viral Hepat. 2013;20(9):593-599.
- ↑ Kim HA, Lee S, Eah KS, Yoon YH. Prevalence and Risk Factors of Tamoxifen Retinopathy. Ophthalmology. 2019.
- ↑ Lonn LI. Canthaxanthin retinopathy. Archives of ophthalmology (Chicago, Ill : 1960). 1987;105(11):1590-1591.
- ↑ Novak MA, Roth AS, Levine MR. Calcium oxalate retinopathy associated with methoxyflurane abuse. Retina (Philadelphia, Pa). 1988;8(4):230-236.
- ↑ Dalvin LA, Shields CL, Orloff M, Sato T, Shields JA. CHECKPOINT INHIBITOR IMMUNE THERAPY: Systemic Indications and Ophthalmic Side Effects. Retina. 2018;38(6):1063-1078.
- ↑ Venkat AG, Arepalli S, Sharma S, et al. Local therapy for cancer therapy-associated uveitis: a case series and review of the literature. Br J Ophthalmol. 2019.
- ↑ Saha N, Bansal S, Bishop F, McWhinney P. Bilateral hypopyon and vitritis associated with rifabutin therapy in an immunocompetent patient taking itraconazole. Eye (London, England). 2009;23(6):1481.
- ↑ Lin HC, Lu PL, Chang CH. Uveitis associated with concurrent administration of rifabutin and lopinavir/ritonavir (Kaletra). In: Eye (Lond). Vol 21. England2007:1540-1541.
- ↑ Tacconelli E, Tumbarello M, Rabagliati R, et al. Correlation between cidofovir-associated uveitis and failing immunorestoration during HAART. Eur J Clin Microbiol Infect Dis. 2003;22(2):114-117.
- ↑ Wang L, Damji KF, Chialant D, Hodge WG. Hypotony after intravenous cidofovir therapy for the treatment of cytomegalovirus retinitis. Canadian journal of ophthalmology Journal canadien d'ophtalmologie. 2002;37(7):419-422.
- ↑ Ryan EH, Jr., Jampol LM. Drug-induced acute transient myopia with retinal folds. Retina. 1986;6(4):220-223.
- ↑ Gruener AM, Jeffries MA, El Housseini Z, Whitefield L. Poppers maculopathy. Lancet. 2014;384(9954):1606.
- ↑ Kwak EL, Bang YJ, Camidge DR, et al. Anaplastic lymphoma kinase inhibition in non-small-cell lung cancer. N Engl J Med. 2010;363(18):1693-1703.