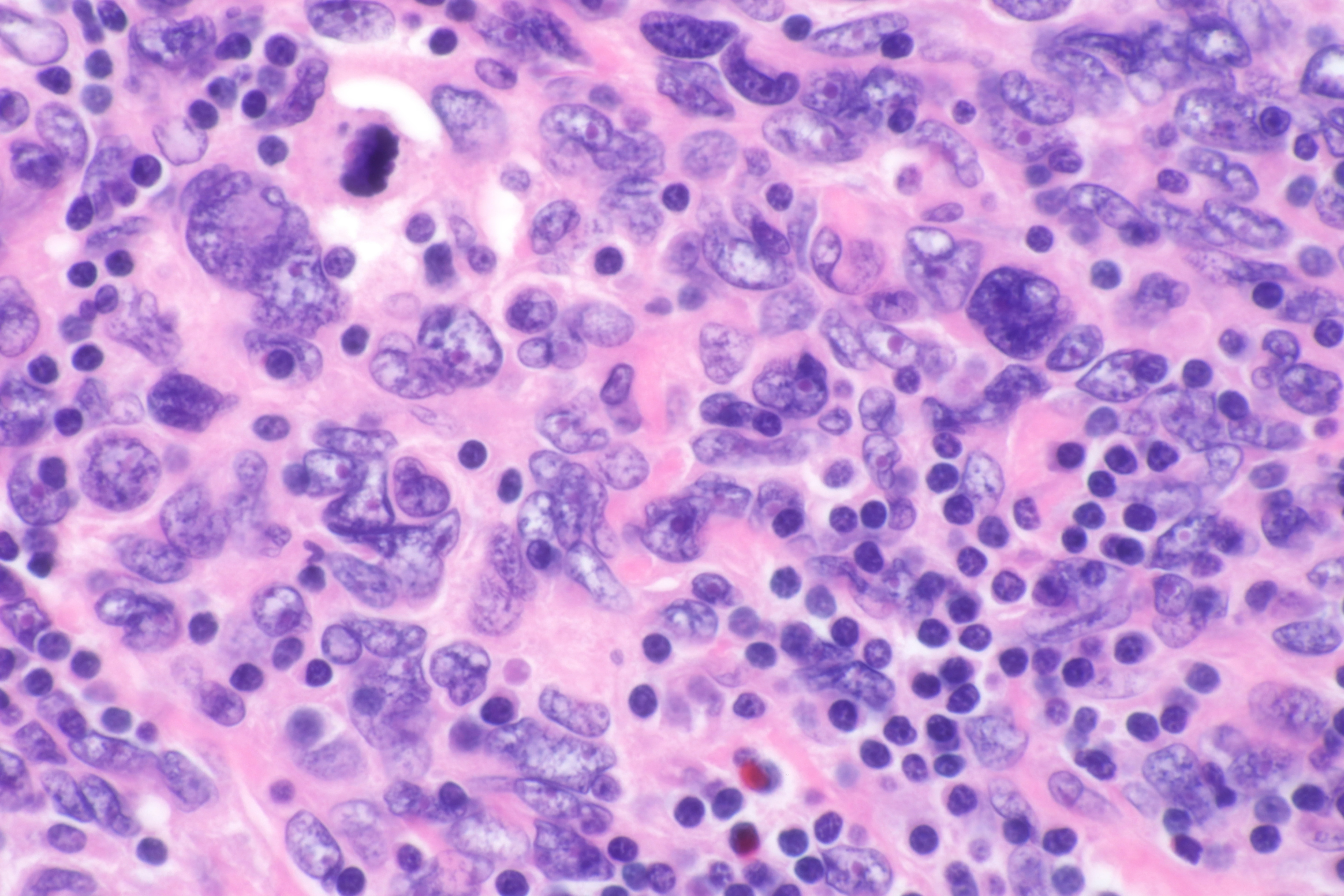
Cranio-Orbital Follicular Dendritic Cell Sarcoma
All content on Eyewiki is protected by copyright law and the Terms of Service. This content may not be reproduced, copied, or put into any artificial intelligence program, including large language and generative AI models, without permission from the Academy.
Disease Entity
Follicular dendritic cell sarcoma (FDCS) is a rare dendritic cell neoplasm representing only 0.4% of all soft tissue sarcomas. Extranodal FDCS of the head and neck region has only been described three times intracranially, and there is only one reported intraorbital case[1][2]. Due to the potential involvement of the cavernous sinus and clivus, complications can include decreased visual acuity, visual field defects, ophthalmoplegia, and papilledema. This EyeWiki will discuss the unique characteristics of FDCS and its relevance to neuro-ophthalmology and strabismus.
Epidemiology
In a pooled analysis of 343 FDCS cases, the median age of diagnosis was 50 years, and there was an equal distribution of males and females. Interestingly, there was a substantial proportion of patients of Asian ancestry[3].
Pathophysiology
Dendritic cells are pivotal in processing and presenting antigens to B and T cells. Follicular dendritic cells, originating from the mesenchyme, exhibit a distinct immunophenotype compared to other histiocytic tumors. The mechanism behind cellular neoplasm by follicular dendritic cells remains uncertain. However, it is believed to involve widespread chromosomal instability, dysregulation of cell cycle progression, activation of nuclear factor kappa beta, mitogen-activated protein kinase, and immune evasion. Mutations in PTEN, TP53, and components of NF-KB have also been documented. One study reported that 67% of FDCS tumors exhibited overexpression of EZH2, and 80% expressed phosphorylated extracellular signal-related protein kinases 1 and 2 (p-ERK1/2)[4]. In addition, 20% of FDCS cases feature the BRAF V600E mutation[5].
The inflammatory variant of FDCS is correlated with Epstein-Barr virus (EBV) infection. Histologically, FDCS is a low-grade sarcoma characterized by spindle-shaped cells with weakly eosinophilic cytoplasm organized in a storiform or whorled pattern. Immunohistochemically, neoplastic FDCS cells express dendritic cell markers CD21, CD23, and CD35, along with vimentin, fascin, human leukocyte antigen, and desmoplakin. Most FDCS cases express clusterin, CXCL13, podoplanin, and gamma synuclein. Notably, the expression of follicular dendritic cell-secreted protein and serglycin exhibits high specificity and sensitivity. Interestingly, despite its classification as a sarcoma, nearly all FDCS cases show overexpression of the epidermal growth factor receptor[6].
Diagnosis
Signs and Symptoms
FDCS typically presents with painless lymphadenopathy but may also develop in extranodal regions (e.g., tonsils, lungs, liver, spleen). Unexplained lymphadenopathy or liver and/or spleen masses accompanied by B symptoms should prompt consideration for FDCS in the differential diagnosis. Intracranial FDCS is exceedingly rare but may produce confusion, memory impairment, and diffuse headaches. The abducens nerve can result in binocular horizontal diplopia when the clivus or sphenoid sinus is involved. Orbital tumors can produce blurred vision, reduced visual acuity, relative afferent pupillary defect (RAPD), ptosis, limited ocular movement, or proptosis[1][2].
Diagnostic Procedure
The diagnosis of FDCS requires histopathologic confirmation with (e.g., CD21, CD23, CD35, epidermal growth factor receptor, KIT, programmed death ligand 1, and BRAF V600E mutation). Analysis of tissue samples demonstrates ovoid or spindle-shaped cells adopting a whorled pattern.[7]
•Imaging studies include computed tomography (CT) or magnetic resonance imaging (MRI) but positron emission tomography (PET) scanning may also be necessary to define the extent of disease.
•Full ophthalmic examination, including visual acuity, visual fields ocular alignment, stereopsis, and fundoscopic examination, may be helpful for patients with possible intracranial or intraorbital disease.
•Clinicians should maintain a high index of suspicion for concurrent Castleman disease (a non-clonal lymphoproliferative disorder)
•Assessment for signs and symptoms indicative of paraneoplastic phenomena (myasthenia gravis)[8].
Differential Diagnosis
•Soft tissue sarcomas (interdigitating dendritic cell sarcomas, GIST).
•Histiocytic neoplasms (Langerhans cell histiocytosis, Rosai-Dorfman Disease).
•Melanoma
Management
The mainstay of treatment for orbital or intracranial FDCS is surgical resection. Patients had more favorable outcomes after complete tumor resection than those managed solely with chemotherapy or radiation therapy. Adjuvant radiation therapy (RT) and chemotherapy may enhance local control, improving progression-free and overall survival rates. Radiation doses of 50 to 55 Gy are commonly administered when suitable. Chemotherapy utilization has yielded a median recurrence-free survival of 2.9 years. Among the effective regimens, a prominent combination involves gemcitabine and docetaxel for treating FDCS.
In cases of diplopia secondary to abducens palsy, prism glasses could be considered to relieve diplopia, and in some cases, strabismus surgery may be indicated.
Although a standardized approach to post-treatment surveillance remains elusive, a collaborative multidisciplinary team involving primary care physicians, oncologists, neuroradiologists, and, if necessary, ophthalmologists is integral. Regular follow-up may be necessary, and repeat neuroimaging may be indicated until stability is attained.
Prognosis
The prognosis for FDCS is variable and ill-defined. Multiple management strategies have been proposed, but only relatively small and retrospective studies on FDCS exist. A pattern of local recurrence after complete resection and a limited occurrence of systemic metastases has been noted. Several factors, particularly lymphoplasmacytic infiltration and tumor size, have been linked to an unfavorable prognosis. Notably, the 2-year survival rate for cases of localized disease stood at approximately 83%, whereas it reduced to approximately 43% for instances of metastatic disease[9].
Summary
FDCS is an uncommon, low-grade sarcoma characterized by a gradual and painless growth pattern and distinct histological features. When the tumor affects the orbit or intracranial regions like the clivus or sphenoid sinus, it can lead to specific ophthalmic manifestations such as diplopia, blurred vision, and visual field impairments. In such instances, collaboration with neuro-ophthalmology and strabismus specialists becomes essential, involving meticulous diagnostic assessment and targeted treatment strategies.
References
- ↑ Jump up to: 1.0 1.1 Haranhalli N, Ammar AE, Weidenheim KM, et al. Hemorrhagic intracranial follicular dendritic cell sarcoma: A case report. Surg Neurol Int 2017;8:248.
- ↑ Jump up to: 2.0 2.1 Grosinger A, Garrity JA, Salomão DR. Follicular dendritic cell sarcoma: a novel orbital tumor. Canadian Journal of Ophthalmology 2021;56:e84–e86.
- ↑ Saygin C, Uzunaslan D, Ozguroglu M, et al. Dendritic cell sarcoma: A pooled analysis including 462 cases with presentation of our case series. Crit Rev Oncol Hematol 2013;88:253–271.
- ↑ Tian X, Xu J, Fletcher C, et al. Expression of zest homolog 2 (EZH2) protein enhancer in histiocytic and dendritic cell neoplasms with evidence for p-ERK1/2-related, but not MYC- or p-STAT3-related cell signaling. Mod Pathol 2018;31:553–561.
- ↑ Go H, Jeon YK, Huh J, et al. Frequent detection of BRAFV600E mutations in histiocytic and dendritic cell neoplasms. Histopathology 2014;65:261–272.
- ↑ Lorenzi L, Döring C, Rausch T, et al. By whole transcriptome sequencing, novel follicular dendritic cell sarcoma markers, FDCSP and SRGN. Oncotarget 2017;8:16463.
- ↑ Lee, Ryung S. et al., Cerebellopontine angle compression with follicular dendritic cell sarcoma of the skull base, Canadian Journal of Ophthalmology, Volume 0, Issue 0
- ↑ Facchetti F, Simbeni M, Lorenzi L. Follicular dendritic cell sarcoma. Pathologica 2021;113:316.
- ↑ Sasaki M, Izumi H, Yokoyama T, et al. Follicular dendritic cell sarcoma is treated with a variety of chemotherapy. Hematol Oncol 2017;35:905.