Cerebral Venous and Dural Sinus Thrombosis
All content on Eyewiki is protected by copyright law and the Terms of Service. This content may not be reproduced, copied, or put into any artificial intelligence program, including large language and generative AI models, without permission from the Academy.
Cerebral Venous and Dural Sinus Thrombosis
Disease Entity
Cerebral venous sinus thrombosis (CVST) is a clot in the venous drainage system of the brain and can present to ophthalmology.
Epidemiology
CVST is an uncommon type of stroke. It accounts for 0.5-3% of all strokes and affects approximately 5 per one million people annually. A patient presenting with CVST-stroke is more likely to be younger (less than 50 years old) and more likely to be female when compared to typical ischemic strokes. [1][2] [3][4] [5]
Risk Factors
The risk of CVST is related to Virchow’s triad (i.e., stasis, changes in blood composition, and changes in the vessel wall). Risk factors for CVST can be separated into acquired and genetic causes. Females are at higher risk for hormone specific risk factors (e.g., oral contraceptives, pregnancy, and hormone replacement therapy). [6]
The more commonly reported etiologies include:
- Prothrombotic conditions:[4]
- Factor V Leiden, Antithrombin III deficiency, Protein C deficiency, Protein S deficiency
- Antiphospholipid and anticardiolipin antibodies
- Pregnancy
- Puerperium
- Oral contraceptive use
- Hypercellular and hyperviscosity conditions (e.g. polycythemia, leukemia, thrombocytosis)
- Certain drugs use (androgen, danazol, lithium, vitamin A, IVIG, ecstasy)
- Hypercoagulative state in cancer (local compression, hypercoagulable state, antineoplastic drugs)
- Infection (parameningeal infections)
- Hematologic disorders (paroxysmal nocturnal hemoglobinuria, iron deficiency anemia, nephrotic syndrome)
- Other systemic diseases/Collagen vascular diseases (e.g. SLE, GPA, Behcet disease, IBD, sarcoidosis)
- Obesity[7]
- Intracranial hypotension[8]
- Inflammatory bowel disease (Crohn’s disease or ulcerative colitis)[9]
In a study, approximatelly 12.5% of cases did not have a known or identifiable cause= Idiopathic[4]
Pathophysiology
The main pathology is a thrombosis of cerebral veins, most common in junctions between cerebral veins and larger sinuses. The dural sinuses not only act as venous channels, but also contain arachnoid granulations that allow the cerebrospinal fluid (CSF) to drain from the subarachnoid space into the systemic venous system. A thrombosis to the dural sinuses causes a back-up in venous pressure, and may impair this CSF drainage resulting in increased intracranial pressure (e.g. headache, nausea, vomiting, papilledema, and visual problems). [10]
The cortical venous system is anatomically variable. Clinical findings of a cortical vein thrombosis depend on the size and extent of the thrombus, location, and nature of collateral supply. If unfavorable conditions exist, a CVST may lead to increased venous and capillary pressure and a breakdown in the blood brain barrier with resultant vasogenic edema, cytotoxic edema, and hemorrhage. [11]
The proposed pathogenesis is as follows: Cortical vein occlusion Increased venous and capillary hydrostatic pressure further blood brain barrier (BBB) disruption Vasogenic edema Intracranial pressure continues to increase Venous/ Capillary rupture and hemorrhage and decreased cerebral perfusion with subsequent ischemia and cytotoxic edema. In most instances, both dural sinus and cortical venous thrombosis occurs simultaneously, with isolation of either being rare due to their effects on one another. [12][13]
The pathophysiology behind the visual impairments often seen in CVST include:
- Raised ICP without infarction
- Compensatory increase in CSF absorption by the arachnoid granulations occurs if ICP is increased. If these arachnoid granulations are disrupted as seen in dural sinus thrombosis, CSF backs up leading to axoplasmic flow stasis with swelling of the optic nerve fiber and optic disc with subsequent venous stasis and extracellular fluid accumulation (i.e., papilledema). Patients can present with signs and symptoms of raised ICP and may be indistinguishable from idiopathic intracranial hypertension (IIH). Hence it is recommended that any patient with suspected IIH (e.g., papilledema) should undergo both a standard magnetic resonance (MR) scan of the head and a venogram (e.g., MRV). Patients with papilledema may complain of transient visual obscurations (lasting seconds at a time) or may develop visual field defects from papilledema. Diplopia may occur due to a non-localizing finding of a sixth nerve palsy due to increased ICP. Headache and pulsatile tinnitus may also occur as non-localizing symptoms of increased ICP and can mimic the presentation of IIH.
- Venous infarcts involving the geniculocalcarine tract, especially the primary visual cortex. These occipital infarcts might produce a homonymous hemianopsia.
- Raised ICP following the development of secondary dural arteriovenous fistula:
- a. Dural AV fistulas can be a late complication of CVST. These can cause increased dural sinus pressure with subsequent decreased CSF absorption and increased ICP.
- Occipital arterial infarcts secondary to mass effect from herniated large venous infarcts. [14]
Diagnosis
History
In 1825, Ribes MF noted the first case of CVST in a 45 year old man presenting with headache, seizures, delirium and autopsy confirmed superior sagittal and lateral sinus thrombosis . The first post-partum autopsy confirmed CVST was in 1828 by Abercrombie of a 25 year old woman with headache and seizures 2 weeks after an uncomplicated delivery. Currently the largest studies exploring CVST are an Italian multicenter study of 706 patients with CVST, and the International Study on Cerebral Vein and Dural Sinus Thrombosis (ISCVDST) which included 624 patients with CVST. [15]
Signs and Symptoms [16]
- Headache: In the ISCVDST, headache was present in 88.8% making it the most common symptom in CVST . Headaches may be the only presenting sign, further complicating the diagnosis. [17] CVST in the absence of a headache is more common in older patients and men when compared to CVST with a headache.[18] There is also a higher incidence of seizures and paresis and a lower incidence of papilledema in CVST without a headache.
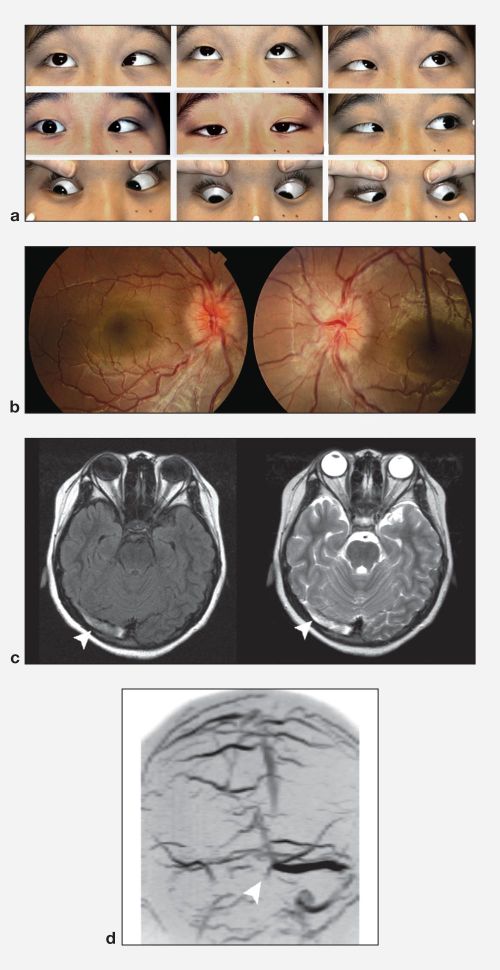
- Visual problems: Visual problems are a common presenting sign/symptom in CVST according to the ISCVST. Visual loss (13.2%), papilledema (28.3%), and diplopia (13.5%) were all noted. Migraine-like visual phenomena (colored photopsia, dark spots, and visual blurring associated with vertical wavy lines), have also been reported. Papilledema is often seen with CVST and there is a direct association of papilledema with elevated ICP. However, in eyes in previous papilledema that have resulted in optic atrophy, the absence of papilledema cannot be used as a marker for raised ICP. Other facial or craniofacial pains may be present as well. If
- Seizures (39.3%) When seizure CVT compared to arterial stroke (40% vs 6%)
- Paresis (37.2%)
- Mental status changes (22%)
- Aphasia (19.1%)
- Stupor/Coma (13.9%)
- Sensory deficits (5.4%)
Clinical diagnosis
The clinical presentation of a CVST can be variable and the diagnosis should be suspected in patients with new onset focal neurological deficits, signs of increased ICP, seizures, or mental status changes. A complete ocular exam including dilated fundus examination, visual field examination, and optic nerve photographs are recommended in patients with CVST.
Diagnostic procedures
MRI/MRV: MRI T2 weighted imaging with MRV is the most sensitive test for identifying CVST . The appearance on MRI is dependent on the age of the thrombus. In the acute setting (days 1-5), the thrombus is typically hypointense on T2 and isointense on T1 weighte MRI. The subacute thrombosis (days 6-15) is usually strongly hyperintense on both T1 and T2 weighted images. After 3 weeks, the signal becomes irregular and either flow was restored or a persistent thrombus was seen. [19]
CT/CTV: Non-contrast head CT is usually the first test ordered with new onset neurological deficits. This test is not very specific for CVST and is abnormal in only approximately 30% of cases. In the roughly 30% of cases where CT reveals a CVST, an empty delta sign may be seen represented as a dense triangle in the posterior portion of the superior sagittal sinus. In areas where MRI/MRV are not as readily available CTV may be added to CT to aid in the suspected diagnosis. [20]
Laboratory test
Routine recommended blood studies include:
- CBC
- CMP
- PT/INR, aPTT
- Hypercoagulable state
Infectious or inflammatory states is recommended in CVST depending on the case presentation and associated symptoms. There is no laboratory study able to help rule out a CVST in the acute state however. [4]
Differential diagnosis
Due to the varying presentation of CVST, the differential diagnosis list is dependent on the presenting symptom and is quite broad. Listed below are some of the top diagnoses to rule out.
- Idiopathic Intracranial Hypertension (IIH)
- Intracranial tumor
- Brain abscess
- Intracerebral hemorrhage
- Ischemic stroke
- Meningoencephalitis
- Autoimmune encephalitis
- Paraneoplastic encephalitis
- Neuromyelitis optica
Management
Medical therapy
If there are no contraindications , anticoagulation in the acute phase with either body-weight adjusted subcutaneous LMWH or dose- adjusted intravenous heparin is the medical therapy of choice. Concomitant intracranial hemorrhage related to the CVST is not an absolute contraindication for heparin therapy. In uncomplicated cases, LMWH is preferred over IV heparin due to less major bleeding problems. There is no evidence available for duration of anticoagulation after the acute phase. [22][23][15]
In cases of intracranial hypertension with secondary papilledema, progressive headache, or third or sixth nerve palsies, management consists of a collection of strategies to reduce the pressure and preserve vision. The first measure is listed above; anticoagulation to reduce thrombotic occlusion of venous outflow. Other measures resemble the treatment of IIH. Serial lumbar punctures to reduce CSF volume can be considered with the caveat of needing to hold anticoagulation while it is performed. Other alternatives include treatment with acetazolamide to decrease CSF production.[19] Because blindness can be the long-term complication of elevated pressures on the optic nerve, close monitoring of visual acuity and visual fields is important in patients with elevated ICP.
All neurologists need to be able to recognise and treat cerebral venous thrombosis (CVT). It is difficult to diagnose, partly due to its relative rarity, its multiple and various clinical manifestations (different from ‘conventional’ stroke, and often mimicking other acute neurological conditions), and because it is often challenging to obtain and interpret optimal and timely brain imaging. Although CVT can result in death or permanent disability, it generally has a favourable prognosis if diagnosed and treated early. Neurologists involved in stroke care therefore also need to be aware of the treatments for CVT (with varying degrees of supporting evidence): the mainstay is prompt anticoagulation but patients who deteriorate despite treatment can be considered for endovascular procedures (endovascular thrombolysis or thrombectomy) or neurosurgery (decompressive craniotomy). This review summarises current knowledge on the risk factors, diagnosis, treatment and prognosis of CVT in adults, and highlights some areas for future research.
Surgery
Optic Nerve Sheath Fenestration (ONSF) can be considered in patients with CVST with elevated ICP when medical management has failed and visual function is failing[24][25]. In patients where intracranial hypertension remains persistent despite adequate medical management and a lumbar drain, a CSF diversion procedure (ventriculoperitoneal or lumboperitoneal shunt) may be considered. [26]
Endovascular thrombolysis and mechanical thrombectomy have not played a prominent role in the treatment of CVST but may be considered in cases of severe neurological deterioration despite use of anticoagulation, venous infarcts causing mass effect, or intracerebral hemorrhage causing treatment resistant intracranial hypertension. [27]
Prognosis
CVT can result in demise or permanent disability if diagnosed and treated late[28].
Early Death: Approximately 3-15% of patients die during the acute phase of CVST. Causes of death were mainly secondary to transtentorial herniation due to local mass effect or diffuse edema and multiple parenchymal lesions.
Late Death: Most are related to underlying diseases; especially malignancy.
Long-term outcomes: The overall death a dependency rate is 15%. Complete recovery occurred in 79% of patients.[29]
Visual prognosis depends on decompression of optic nerve. Therefore if CVT vascular treatment has not improved optic nerve edema, then optic nerve sheath fenestration may be the next step for better visual prognosis[24][25]
Additional Resources
References
- ↑ Coutinho, J. M., Ferro, J. M., Canhão, P., Barinagarrementeria, F., Cantú, C., Bousser, M., & Stam, J.(2009)
- ↑ Behrouzi R, Punter M. Diagnosis and management of cerebral venous thrombosis [published correction appears in Clin Med (Lond). 2018 Mar;18(2):192]. Clin Med (Lond). 2018;18(1):75-79. doi:10.7861/clinmedicine.18-1-75.
- ↑ Cerebral Venous and Sinus Thrombosis in Women. Stroke, 40(7), 2356-2361. doi:10.1161/strokeaha.108.543884
- ↑ 4.0 4.1 4.2 4.3 Saposnik G, Barinagarrementeria F, Brown RD, et al. Diagnosis and Management of Cerebral Venous Thrombosis. Stroke. 2011;42(4):1158-1192. doi:10.1161/str.0b013e31820a8364.
- ↑ Saposnik G, Bushnell C, Coutinho JM, et al. Diagnosis and Management of Cerebral Venous Thrombosis: A Scientific Statement From the American Heart Association. Stroke. 2024;55(3):e77-e90. doi:10.1161/STR.0000000000000456
- ↑ Bousser M-G, Ferro JM. Cerebral venous thrombosis: an update. The Lancet Neurology. 2007;6(2):162-170. doi:10.1016/s1474-4422(07)70029-7.
- ↑ Zuurbier SM, Arnold M, Middeldorp S, et al. Risk of Cerebral Venous Thrombosis in Obese Women. JAMA Neurol. 2016;73(5):579-584. doi:10.1001/jamaneurol.2016.0001
- ↑ Fujii N, Fujii H, Fujita A, Kim Y, Sugimoto H. Spontaneous intracranial hypotension complicated by cerebral venous thrombosis. Radiol Case Rep. 2018;13(4):834-838. Published 2018 Jun 20. doi:10.1016/j.radcr.2018.05.014
- ↑ Taous A, Berri MA, Lamsiah T, Zainoun B, Ziadi T, Rouimi A. Cerebral venous thrombosis revealing an ulcerative colitis. Pan Afr Med J. 2016;23:120. Published 2016 Mar 24. doi:10.11604/pamj.2016.23.120.9186
- ↑ Coutinho, JM. Cerebral venous thrombosis. J Thromb Haemost 2015; 13 (Suppl. 1): S238– S44.
- ↑ Itrat, Ahmed, Shoucat, Sana, Kamal, Ayeesha K. Pathophysiology of Cerebral Venous Thrombosis- An Overview. Journal of the Pakistan Medical Association. 2006; 56 (11).
- ↑ Gotoh, M., Ohmoto, T., & Kuyama, H. (1993). Experimental study of venous circulatory disturbance by dural sinus occlusion. Acta Neurochirurgica., 124(2-4), 120–126.
- ↑ Hayreh SS. Pathogenesis of optic disc edema in raised intracranial pressure. Prog Retin Eye Res. 2016;50:108–144. doi:10.1016/j.preteyeres.2015.10.001
- ↑ Aaron S, Arthur A, Prabakhar AT, et al. Spectrum of Visual Impairment in Cerebral Venous Thrombosis: Importance of Tailoring Therapies Based on Pathophysiology. Ann Indian Acad Neurol. 2017;20(3):294–301. doi:10.4103/aian.AIAN_11_17
- ↑ 15.0 15.1 Luo Y, Tian X, Wang X. Diagnosis and Treatment of Cerebral Venous Thrombosis: A Review. Front Aging Neurosci. 2018;10:2. Published 2018 Jan 30. doi:10.3389/fnagi.2018.00002
- ↑ Ferro José M., Canhão Patrícia, Stam J, Bousser M-G, Barinagarrementeria F. Prognosis of Cerebral Vein and Dural Sinus Thrombosis. Stroke. 2004;35(3):664-670. doi:10.1161/01.str.0000117571.76197.26.
- ↑ Cumurciuc R, Crassard I, Sarov M, Valade D, Bousser MG. Headache as the only neurological sign of cerebral venous thrombosis: a series of 17 cases. J Neurol Neurosurg Psychiatry. 2005;76(8):1084–1087. doi:10.1136/jnnp.2004.056275
- ↑ Coutinho JM, Stam J, Canhão P, Barinagarrementeria F, Bousser M-G, Ferro JM. Cerebral Venous Thrombosis in the Absence of Headache. Stroke. 2015;46(1):245-247. doi:10.1161/strokeaha.114.007584.
- ↑ 19.0 19.1 Isensee C, Reul J, Thron A. Magnetic resonance imaging of thrombosed dural sinuses. Stroke. 1994;25(1):29-34. doi:10.1161/01.str.25.1.29.
- ↑ Selim M, Fink J, Linfante I, Kumar S, Schlaug G, Caplan LR. Diagnosis of Cerebral Venous Thrombosis With Echo-Planar T2*-Weighted Magnetic Resonance Imaging. Arch Neurol. 2002;59(6):1021–1026. doi:10.1001/archneur.59.6.1021
- ↑ American Academy of Ophthalmology. Venous sinus thrombosis. https://www.aao.org/education/image/venous-sinus-thrombosis-2 Accessed August 07, 2022.
- ↑ Einhäupl, K. , Stam, J. , Bousser, M. ‐., De Bruijn, S. F., Ferro, J. M., Martinelli, I. and Masuhr, F. (2010), EFNS guideline on the treatment of cerebral venous and sinus thrombosis in adult patients. European Journal of Neurology, 17: 1229-1235. doi:10.1111/j.1468-1331.2010.03011.x
- ↑ Ferro, J. M., Bousser, M. , Canhão, P. , Coutinho, J. M., Crassard, I. , Dentali, F. , Minno, M. , Maino, A. , Martinelli, I. , Masuhr, F. , Aguiar de Sousa, D. , Stam, J. and , (2017), European Stroke Organization guideline for the diagnosis and treatment of cerebral venous thrombosis – endorsed by the European Academy of Neurology. Eur J Neurol, 24: 1203-1213. doi:10.1111/ene.13381
- ↑ 24.0 24.1 Xue X, Zhou C, Gao Y, Ji X, Zhang X. Optic nerve sheath fenestration for visual impairment in cerebral venous diseases. Front Neurol. 2023;14:1065315. Published 2023 Jan 24. doi:10.3389/fneur.2023.1065315
- ↑ 25.0 25.1 Murdock J, Tzu JH, Schatz NJ, Lee WW. Optic nerve sheath fenestration for the treatment of papilledema secondary to cerebral venous thrombosis. J Neuroophthalmol. 2014;34(1):67-69. doi:10.1097/WNO.0000000000000087
- ↑ Acheson JF. Optic nerve disorders: role of canal and nerve sheath decompression surgery. Eye. 2004;18(11):1169-1174. doi:10.1038/sj.eye.6701559.
- ↑ Siddiqui FM, Dandapat S, Banerjee C, et al. Mechanical Thrombectomy in Cerebral Venous Thrombosis: Systematic Review of 185 Cases. Stroke. 2015;46(5):1263-1268. doi:10.1161/strokeaha.114.007465.
- ↑ Ulivi L, Squitieri M, Cohen H, et al. Cerebral venous thrombosis: a practical guide. Practical Neurology 2020;20:356-367.
- ↑ Canhão Patrícia, Ferro José M., Lindgren AG, Bousser M-G, Stam J, Barinagarrementeria F. Causes and Predictors of Death in Cerebral Venous Thrombosis. Stroke. 2005;36(8):1720-1725. doi:10.1161/01.str.0000173152.84438.1c.

