Neonatal Conjunctivitis: Difference between revisions
No edit summary |
No edit summary |
||
| (17 intermediate revisions by 5 users not shown) | |||
| Line 1: | Line 1: | ||
{{Article | {{Article | ||
|Authors=Kbowman | |Authors=Kbowman | ||
|Category=Articles, Pediatric Ophthalmology/Strabismus | |Additional contributors=Erin.Yu | ||
|Assigned editor= | |Category=Articles,Pediatric Ophthalmology/Strabismus | ||
|Reviewer= | |Assigned editor=Angeline.Nguyen | ||
|Date reviewed=September | |Reviewer=Angeline.Nguyen | ||
|Date reviewed=September 15, 2024 | |||
|Article status=Up to Date | |||
}} | }} | ||
{{Infobox disease | {{Infobox disease | ||
| Line 15: | Line 17: | ||
}} | }} | ||
Neonatal conjunctivitis is defined as conjunctival inflammation occurring within the first 30 days of life. Numerous etiologies have been implicated including chemical conjunctivitis as well as viral and bacterial infections. Complications range from mild hyperemia and scant discharge to permanent scarring and blindness. | Neonatal conjunctivitis is defined as conjunctival inflammation occurring within the first 30 days of life. Numerous etiologies have been implicated, including chemical conjunctivitis as well as viral and bacterial infections. Complications range from mild hyperemia and scant discharge to permanent corneal scarring and blindness. | ||
= Disease Entity = | = Disease Entity = | ||
Neonatal Conjunctivitis. Also known as neonatorum ophthalmia. ICD-9 771.6 | Neonatal Conjunctivitis. Also known as neonatorum ophthalmia. ICD-9 771.6, ICD-10 P39.1 | ||
== Disease == | == Disease == | ||
| Line 27: | Line 29: | ||
== Prevalence == | == Prevalence == | ||
Prevalence of neonatal conjunctivitis has decreased significantly in developed countries since the abandonment of silver nitrate as topical prophylaxis. Current estimates of prevalence of neonatal conjunctivitis in developed countries are typically < 0.5%. However, a higher incidence of neonatal conjunctivitis is still found certain regions of the world, particularly in developing countries. | Prevalence of neonatal conjunctivitis has decreased significantly in developed countries since the abandonment of silver nitrate as topical prophylaxis for bacterial conjunctivitis. Current estimates of prevalence of neonatal conjunctivitis in developed countries are typically < 0.5%. However, a higher incidence of neonatal conjunctivitis is still found in certain regions of the world, particularly in developing countries where routine prenatal screening and postnatal prophylaxis of the neonate are not commonly practiced, and differences in the prevalence of maternal infection continue to exist. One retrospective study reports an 8.98% prevalence of neonatal conjunctivitis in a rural community in Ghana.<ref>Opoku-Agyeman K, Adu-Gyamfi PKT, Ansah C, Mensah KD. Effects of prenatal antibiotic treatment on early infant health: a retrospective study in a rural health facility in Ghana. African Health Sciences. 2023 April 11. doi: 10.4314/ahs.v23i1.44.</ref> Furthermore, studies from sub-Saharan Africa suggest that Chlamydia trachomatis accounted for up to 33% of neonatal conjunctivitis.<ref>Adachi KN, Nielsen-Saines K, Klausner JD. ''Chlamydia trachomatis'' Screening and Treatment in Pregnancy to Reduce Adverse Pregnancy and Neonatal Outcomes: A Review. Front Public Health. 2021 Jun 10;9:531073. doi: 10.3389/fpubh.2021.531073. PMID: 34178906; PMCID: PMC8222807.</ref> | ||
== Etiology == | == Etiology == | ||
| Line 33: | Line 35: | ||
The three main etiologies of neonatal conjunctivitis include: | The three main etiologies of neonatal conjunctivitis include: | ||
* Chemical | * Chemical | ||
* Bacterial | * Bacterial | ||
* Viral | * Viral | ||
=== Chemical === | === Chemical === | ||
Classically, the most common cause of neonatal conjunctivitis was due to the post-delivery use of ophthalmic silver nitrate for the prophylaxis of ocular gonococcal infections. However, the incidence of chemical conjunctivitis in the United States has significantly decreased since replacement of silver nitrate with erythromycin ointment. | |||
=== Bacterial === | === Bacterial === | ||
Bacterial | Bacterial causes of neonatal conjunctivitis include: | ||
* ''Chlamydia trachomatis'' (most common) | * ''Chlamydia trachomatis'' (most common) | ||
* ''Neisseria gonnorhea'' | * ''Neisseria gonnorhea'' | ||
** '''N. gonorrhea''' is one of the most severe causes of neonatal conjunctivitis, requiring prompt diagnosis and treatment | |||
* S. ''aureus'' | * S. ''aureus'' | ||
* ''Pseudomonas aeruginosa ''* | * ''Pseudomonas aeruginosa '' | ||
* ''Streptococcus spp. ''(including S.''haemolyticus,'' S. ''pneumonia'') | ** '''Pseudomonas''', although rare, may lead to potentially blinding complications such as rapid corneal ulceration and perforation | ||
* ''Streptococcus spp. ''(including S. ''haemolyticus,'' S. ''pneumonia'') | |||
* Other bacteria include Klebsiella, Proteus, Enterobacter, Serratia, and Eikenella corrodens<ref name=":6">Chhabra MS, Motley WW 3rd, Mortensen JE. Eikenella corrodens as a causative agent for neonatal conjunctivitis.''J AAPOS''. 2008 Oct;12(5):524-5. </ref> | * Other bacteria include Klebsiella, Proteus, Enterobacter, Serratia, and Eikenella corrodens<ref name=":6">Chhabra MS, Motley WW 3rd, Mortensen JE. Eikenella corrodens as a causative agent for neonatal conjunctivitis.''J AAPOS''. 2008 Oct;12(5):524-5. </ref> | ||
=== Viral === | === Viral === | ||
Viral etiologies include: | Viral etiologies include: | ||
* Herpes simplex virus (HSV) | * Herpes simplex virus (HSV) | ||
== Risk Factors == | == Risk Factors == | ||
| Line 64: | Line 61: | ||
Risk factors of neonatal conjunctivitis may include: | Risk factors of neonatal conjunctivitis may include: | ||
* Maternal infections harbored in the mother's birth canal | * Maternal infections harbored in the mother's birth canal | ||
* HIV- | * Maternal HIV-infection<ref name="gichuhi3">Gichuhi S et al. Risk factors for neonatal conjunctivitis in babies of HIV-1 infected mothers. ''Ophthalmic Epidemiol''. 2009 Nov-Dec;16(6):337-45. </ref> | ||
* Exposure of the infant to infectious organisms | * Exposure of the infant to infectious organisms | ||
* Increased birth weight<ref name="gichuhi3"/> | * Increased birth weight<ref name="gichuhi3"/> | ||
* Inadequacy of ocular prophylaxis immediately after birth | * Inadequacy of ocular prophylaxis immediately after birth | ||
* PROM<ref name=":5">Wu J et al. Influence of premature rupture of membranes on neonatal health. ''Zhonghua Er Ke Za Zhi''. 2009 Jun;47(6):452-6. </ref> | * Premature rupture of membranes (PROM)<ref name=":5">Wu J et al. Influence of premature rupture of membranes on neonatal health. ''Zhonghua Er Ke Za Zhi''. 2009 Jun;47(6):452-6. </ref> | ||
* Ocular trauma during delivery | * Ocular trauma during delivery | ||
* Mechanical ventilation | * Mechanical ventilation | ||
| Line 78: | Line 75: | ||
== Pathophysiology == | == Pathophysiology == | ||
Neonatal conjunctivitis is an ocular surface disease consisting of inflammation of the conjunctiva, blood vessel dilation, tearing, and purulent or non-purulent drainage within 30 days of age. Newborns are vulnerable to developing conjunctivitis due to reduced tear secretion and drainage, as well as decreased immune function. Furthermore, neonates lack lymphoid tissue in the conjunctiva, immunoglobulin A and adequate lysozyme activity which normally serve as natural barriers against infectious pathogens. As a result, neonatal conjunctivitis often gets transmitted during passage through the vaginal canal of a mother with a sexually transmitted infection, and can result in serious complications such as corneal ulceration, perforation, and permanent blindness. | |||
== Prevention == | == Prevention == | ||
Prevention through good prenatal care and treatment of chlamydial, gonococcal, or herpetic infections during pregnancy remains the best preventative method | Prevention through good prenatal care and treatment of chlamydial, gonococcal, or herpetic infections during pregnancy remains the best preventative method. | ||
*Chlamydial infections occur in 4–10% of pregnant women in the United States | *Chlamydial infections occur in 4–10% of pregnant women in the United States | ||
| Line 93: | Line 86: | ||
=== Topical Prophylaxis === | === Topical Prophylaxis === | ||
Use of topical silver nitrate to prevent neonatal gonococcal conjunctivitis was first introduced by Credé in 1880 and has been classically been cited as the most common cause of neonatal conjunctivitis. However, the incidence of chemical conjunctivitis has declined as the use of silver nitrate as prophylaxis has been abandoned in many modern countries in favor of topical medications with a more favorable side effect profile such as erythromycin. | Use of topical silver nitrate to prevent neonatal gonococcal conjunctivitis was first introduced by Credé in 1880 and has been classically been cited as the most common cause of neonatal conjunctivitis. However, the incidence of chemical conjunctivitis has declined as the use of silver nitrate as prophylaxis has been abandoned in many modern countries in favor of topical medications with a more favorable side effect profile such as erythromycin. However, countries often vary in their accessibility to these more favorable topical antibiotics. One study evaluating the current methods of treatment in maternity hospitals in Croatia showed that 75% of all maternity hospitals in Croatia routinely administered preventive topical treatment, including topical tobramycin (83.3%), povidone-iodine (8.3%), erythromycin (4.2%), and silver nitrate (4.2%),<ref>Kaštelan S, Anić Jurica S, Orešković S, Župić T, Herman M, Gverović Antunica A, Marković I, Bakija I. A Survey of Current Prophylactic Treatment for Ophthalmia Neonatorum in Croatia and a Review of International Preventive Practices. Med Sci Monit. 2018 Nov 10;24:8042-8047. doi: 10.12659/MSM.910705. PMID: 30413681; PMCID: PMC6240167.</ref> partially due to the lack of availability of erythromycin and tetracycline in Croatia. Data supporting the superiority of one topical antibiotic over another seem to remain unequivocal. A recent systematic review including 30 trials found little evidence that any prophylaxis involving tetracycline 1%, erythromycin 0.5%, povidone-iodine 2.5%, or silver nitrate 1% provided better prevention from serious outcomes such as blindness or any adverse visual outcomes.<ref>Kapoor VS, Evans JR, Vedula SS. Interventions for preventing ophthalmia neonatorum. Cochrane Database Syst Rev. 2020 Sep 21;9(9):CD001862. doi: 10.1002/14651858.CD001862.pub4. PMID: 32959365; PMCID: PMC8524318.</ref> | ||
=== Systemic Prophylaxis === | === Systemic Prophylaxis === | ||
Infants with possible infectious exposure in utero or during birth process should receive appropriate prophylaxis following birth in attempt to prevent ocular and systemic complications. | Infants with possible infectious exposure in utero or during birth process should receive appropriate prophylaxis following birth in attempt to prevent ocular and systemic complications. Gonococcal prophylaxis includes single injection of ceftriaxone 50 mg/kg IM or IV in those neonates born to mothers with untreated or suspected gonococcal infection. Other preventative measures include proper hand-washing techniques by peripartum and nursery staff. | ||
=== Maternal Screening === | |||
The United States Preventive Services Task (USPSTF) currently recommends routine screening for chlamydial and gonorrheal infection in all sexually active or pregnant women if they are 24 years or younger, or at least 25 years and at increased risk for infection. These preventative screening measures have significantly decreased perinatal chlamydial and gonorrheal infections in the United States. One study done at a medical center in the United States reports a decrease from 15.6% positivity rate of Chlamydia trachomatis eye cultures before the implementation of universal prenatal screening (1986-1993), to 1.8% during the screening period (1994-2002).<ref>Kohlhoff S, Roblin PM, Clement S, Banniettis N, Hammerschlag MR. Universal Prenatal Screening and Testing and Chlamydia trachomatis Conjunctivitis in Infants. Sex Transm Dis. 2021 Sep 1;48(9):e122-e123. doi: 10.1097/OLQ.0000000000001344. PMID: 33346588.</ref> | |||
= Diagnosis = | = Diagnosis = | ||
Prompt diagnosis is key in establishing proper treatment and minimizing potential serious complications of | Prompt diagnosis is key in establishing proper treatment and minimizing potential serious complications of neonatal conjunctivitis. | ||
== History == | == History == | ||
| Line 110: | Line 102: | ||
Time frame of signs/symptoms following birth play an important role in determining the most likely etiology and subsequent proper diagnosis and treatment: | Time frame of signs/symptoms following birth play an important role in determining the most likely etiology and subsequent proper diagnosis and treatment: | ||
*Chemical conjunctivitis ( | *Chemical conjunctivitis (presents within the first 24 hours following birth) | ||
*Neisseria gonorrhea (3-5 days after birth) | *Neisseria gonorrhea (presents 3-5 days after birth) | ||
* | *Chlamydia trachomatis (presents 5-14 days after birth) | ||
*HSV (1-2 weeks) | *HSV (presents 1-2 weeks after birth) | ||
== Physical examination == | == Physical examination == | ||
| Line 131: | Line 123: | ||
==== Chlamydia trachomatis ==== | ==== Chlamydia trachomatis ==== | ||
*Presentation may range from mild hyperemia with scant mucoid discharge to eyelid swelling, chemosis, and pseudomembrane | *Presentation may range from mild hyperemia with scant mucoid discharge to eyelid swelling, chemosis, and pseudomembrane formation | ||
==== Neisseria gonorrhea ==== | ==== Neisseria gonorrhea ==== | ||
*Typically, | *Typically, patients present with acute conjunctivitis, associated with chemosis, severe lid edema, and mucopurulent discharge | ||
*Corneal involvement is the most serious complication, involving diffuse epithelial edema and ulceration that may progress to perforation of the cornea and endophthalmitis | *Corneal involvement is the most serious complication, involving diffuse epithelial edema and ulceration that may progress to perforation of the cornea and endophthalmitis | ||
*Initially, superficial keratitis gives the corneal surface a lackluster appearance followed by marginal and central infiltrates appear, which then ulcerate | *Initially, superficial keratitis gives the corneal surface a lackluster appearance followed by marginal and central infiltrates appear, which then ulcerate | ||
==== HSV ==== | ==== HSV ==== | ||
*Typically present with unilateral or bilateral lid edema, moderate amount of conjunctival injection, and | *Typically, patients present with unilateral or bilateral lid edema, moderate amount of conjunctival injection, and non-purulent, serosanguinous discharge | ||
*Other signs include vesicles on the skin surrounding the eye and corneal epithelial involvement with microdendrites or geographic ulcers | **Other signs include vesicles on the skin surrounding the eye and corneal epithelial involvement with microdendrites or geographic ulcers (in contrast to typical dendrites as seen in adults) | ||
[[Image:Conjunctivitis.jpg|frame]][[Image:Conjunctivitis2.jpg|frame]] | [[Image:Conjunctivitis.jpg|frame]][[Image:Conjunctivitis2.jpg|frame]] | ||
| Line 150: | Line 142: | ||
The differential diagnosis of neonatal conjunctivitis includes: | The differential diagnosis of neonatal conjunctivitis includes: | ||
* | *Dacyrocysitis | ||
*Congenital glaucoma | *Congenital glaucoma | ||
*Nasolacrimal duct obstruction | *Nasolacrimal duct obstruction | ||
*Preseptal/Orbital cellulitis | *Preseptal/Orbital cellulitis | ||
*Infectious keratitis | *Infectious keratitis | ||
| Line 161: | Line 152: | ||
Laboratory studies for suspected infectious etiology should include the following: | Laboratory studies for suspected infectious etiology should include the following: | ||
*Conjunctival scraping w/ Gram stain and Giemsa stain for chlamydia | *Conjunctival scraping w/ Gram stain and Giemsa stain for chlamydia. | ||
*Culture on chocolate agar for N | **Note: Conjunctival specimens for chlamydia testing must include conjunctival epithelial cells because C. trachomatis is an obligate intracellular organism and exudates are not adequate for testing. | ||
*Culture on chocolate agar for N. gonorrheae. (Thayer-Martin media may also be used) | |||
*Culture on blood agar for other strains of bacteria | *Culture on blood agar for other strains of bacteria | ||
*Culture for HSV if vesicles present or is | *Culture for HSV if vesicles present or is suspicious of viral etiology including evidence of perinatal maternal exposure | ||
*Direct antibody testing or PCR may also be indicated.<ref name=":7">Yip PP et al. The use of polymerase chain reaction assay versus conventional methods in detecting neonatal chlamydial conjunctivitis. ''J Pediatr Ophthalmol Strabismus. ''2008 Jul-Aug;45(4):234-9. </ref> | *Direct antibody testing or PCR may also be indicated.<ref name=":7">Yip PP et al. The use of polymerase chain reaction assay versus conventional methods in detecting neonatal chlamydial conjunctivitis. ''J Pediatr Ophthalmol Strabismus. ''2008 Jul-Aug;45(4):234-9. </ref> | ||
*Cultures may need to be repeated if symptoms worsen or recur following treatment. | *Cultures may need to be repeated if symptoms worsen or recur following treatment. | ||
| Line 174: | Line 165: | ||
== Chemical conjunctivitis == | == Chemical conjunctivitis == | ||
No treatment required | No treatment required, as the condition typically resolves within 2-4 days. Supportive care involves artificial tears q.i.d. | ||
== Chlamydial conjunctivitis == | |||
Treatment should be initiated upon a positive diagnostic test. Oral azithromycin may be used for both chlamydial conjunctivitis and pneumonia, 20 mg/kg once daily for three days. Alternatively, erythromycin can be given orally 50 mg/kg/day divided in 4 doses, for 14 days. Topical therapy for chlamydial conjunctivitis has proven to be relatively ineffective compared to oral therapy, particularly in treating nasopharyngeal infection.<ref>Heggie AD, Jaffe AC, Stuart LA, Thombre PS, Sorensen RU. Topical sulfacetamide vs oral erythromycin for neonatal chlamydial conjunctivitis. Am J Dis Child. 1985 Jun;139(6):564-6. doi: 10.1001/archpedi.1985.02140080034027. PMID: 3890519.</ref> | |||
The use of azithromycin and erythromycin are associated with increased risk of infantile hypertrophic pyloric stenosis (IHPS) in infants within 2 weeks of life. Infants should be closely monitored for potential signs of IHPS when started on azithromycin or erythromycin. | |||
== Gonococcal conjunctivitis == | == Gonococcal conjunctivitis == | ||
Topical irrigation with normal saline to remove mucopurulent discharge. | Topical irrigation with normal saline should be used to remove mucopurulent discharge, as Gonococcal infection may produce more mucopurulent discharge than other pathogens. Ceftriaxone should be administered in a single dose (25-50 mg/kg IM or IV, up to a maximum of 125 mg). If there is systemic disease, treatment is required for 7 to 14 days, depending on the nature of the invasive infection. Bacitracin or erythromycin ointment should be administered every 2 to 4 hours in addition to systemic treatment of ceftriaxone. Hospitalization and evaluation for disseminated N. gonorrhea infection should be initiated. Topical atropine should be considered for pain relief if there is corneal involvement. | ||
Ceftriaxone in a single dose (25-50 mg/kg IM or IV, up to a maximum of 125 mg). If there is systemic disease, treatment is required for 7 to 14 days depending on the nature of the invasive infection. | |||
Bacitracin or erythromycin ointment every 2 to 4 hours. | The use of ceftriaxone in infants may cause hyperbilirubinemia, and therefore should be avoided in neonates receiving calcium-containing intravenous fluids. Cefotaxime or ceftazidime may be used instead. | ||
Hospitalization and evaluation for disseminated N. gonorrhea infection. | |||
Topical atropine if corneal involvement. | |||
Of note, all neonates with gonococcal conjunctivitis should also be treated for chlamydia. The patient's mother and sexual partner should be treated as well. | |||
== HSV keratoconjunctivitis == | == HSV keratoconjunctivitis == | ||
Acyclovir IV 45mg/kg/day plus vidarabine 3% ointment 5x/day for 14-21 days depending on presence | Acyclovir IV 45mg/kg/day plus vidarabine 3% ointment 5x/day should be administered for 14-21 days depending on presence of central nervous system involvement. | ||
== Other bacteria: == | == Other bacteria: == | ||
* Gram(+) | * Gram(+): Bacitracin ointment q.i.d. for 2 weeks | ||
* Gram(-) | * Gram(-): Gentamicin, tobramycin or ciprofloxacin q.i.d. for 2 weeks | ||
== Medical follow up == | == Medical follow up == | ||
*Patients with neonatal conjunctivitis should be followed daily for signs of improvement or worsening, especially acutely due to concerns of rapidly progressing infectious complications | *Patients with neonatal conjunctivitis should be followed daily for signs of improvement or worsening, especially acutely due to concerns of rapidly progressing infectious complications | ||
*Patient should be followed closely by pediatrician for evaluation and treatment of potential systemic infection. | *Patient should be followed closely by pediatrician for evaluation and treatment of potential systemic infection. | ||
== Complications == | == Complications == | ||
Ocular complications of neonatal conjunctivitis include pseudomembrane formation, corneal edema, thickened palpebral | Ocular complications of neonatal conjunctivitis include pseudomembrane formation, corneal edema, thickened palpebral conjunctiva, peripheral pannus formation, corneal opacification, corneal perforation, endophthalmitis, loss of eye, and blindness. | ||
Systemic complications of chlamydia conjunctivitis include pneumonitis, otitis, and pharyngeal and rectal colonization. Pneumonia has been reported in 10-20% of infants with chlamydial conjunctivitis. Complications of gonococcal conjunctivitis and | Systemic complications of chlamydia conjunctivitis include pneumonitis, otitis, and pharyngeal and rectal colonization. Pneumonia has been reported in 10-20% of infants with chlamydial conjunctivitis. Complications of gonococcal conjunctivitis and subsequent systemic involvement include arthritis, meningitis, anorectal infection, septicemia, and death. Furthermore, Gonococci have the added potential of being able to penetrate intact corneal epithelium, leading to corneal epithelial edema, ulceration, perforation, and endophthalmitis if unrecognized.<ref>Mallika P, Asok T, Faisal H, Aziz S, Tan A, Intan G. Neonatal conjunctivitis - a review. Malays Fam Physician. 2008 Aug 31;3(2):77-81. PMID: 25606121; PMCID: PMC4170304.</ref> | ||
Risk of complications can minimized with prompt diagnosis and appropriate antibiotic therapy. | Risk of complications can be minimized with prompt diagnosis and appropriate antibiotic therapy. | ||
== Prognosis == | == Prognosis == | ||
The prognosis of neonatal conjunctivitis is generally considered to be good as long as early diagnosis is made and prompt medical therapy is initiated. Most cases of infectious conjunctivitis respond to appropriate treatment. However, morbidity and mortality increases in cases of systemic involvement requiring hospitalization and intensive monitoring. | |||
= Additional Resources = | = Additional Resources = | ||
| Line 227: | Line 212: | ||
* [http://one.aao.org/asset.axd?id=e6b8d112-bcab-4bfa-b1fb-b78d84a3dff4 AAO Preferred Practice Pattern: Conjunctivitis] | * [http://one.aao.org/asset.axd?id=e6b8d112-bcab-4bfa-b1fb-b78d84a3dff4 AAO Preferred Practice Pattern: Conjunctivitis] | ||
* [http://www.aapos.org/terms/conditions/37 AAPOS FAQ about Conjunctivitis] | * [http://www.aapos.org/terms/conditions/37 AAPOS FAQ about Conjunctivitis] | ||
= References = | = References = | ||
<references /> | <references /> | ||
Revision as of 17:14, September 15, 2024
All content on Eyewiki is protected by copyright law and the Terms of Service. This content may not be reproduced, copied, or put into any artificial intelligence program, including large language and generative AI models, without permission from the Academy.
Neonatal conjunctivitis is defined as conjunctival inflammation occurring within the first 30 days of life. Numerous etiologies have been implicated, including chemical conjunctivitis as well as viral and bacterial infections. Complications range from mild hyperemia and scant discharge to permanent corneal scarring and blindness.
Disease Entity
Neonatal Conjunctivitis. Also known as neonatorum ophthalmia. ICD-9 771.6, ICD-10 P39.1
Disease
Neonatal conjunctivitis is defined as conjunctival inflammation occurring within the first 30 days of life. Numerous etiologies have been implicated including chemical conjunctivitis as well as viral and bacterial infections. Complications range from mild hyperemia and scant discharge to permanent scarring and blindness.
Prevalence
Prevalence of neonatal conjunctivitis has decreased significantly in developed countries since the abandonment of silver nitrate as topical prophylaxis for bacterial conjunctivitis. Current estimates of prevalence of neonatal conjunctivitis in developed countries are typically < 0.5%. However, a higher incidence of neonatal conjunctivitis is still found in certain regions of the world, particularly in developing countries where routine prenatal screening and postnatal prophylaxis of the neonate are not commonly practiced, and differences in the prevalence of maternal infection continue to exist. One retrospective study reports an 8.98% prevalence of neonatal conjunctivitis in a rural community in Ghana.[1] Furthermore, studies from sub-Saharan Africa suggest that Chlamydia trachomatis accounted for up to 33% of neonatal conjunctivitis.[2]
Etiology
The three main etiologies of neonatal conjunctivitis include:
- Chemical
- Bacterial
- Viral
Chemical
Classically, the most common cause of neonatal conjunctivitis was due to the post-delivery use of ophthalmic silver nitrate for the prophylaxis of ocular gonococcal infections. However, the incidence of chemical conjunctivitis in the United States has significantly decreased since replacement of silver nitrate with erythromycin ointment.
Bacterial
Bacterial causes of neonatal conjunctivitis include:
- Chlamydia trachomatis (most common)
- Neisseria gonnorhea
- N. gonorrhea is one of the most severe causes of neonatal conjunctivitis, requiring prompt diagnosis and treatment
- S. aureus
- Pseudomonas aeruginosa
- Pseudomonas, although rare, may lead to potentially blinding complications such as rapid corneal ulceration and perforation
- Streptococcus spp. (including S. haemolyticus, S. pneumonia)
- Other bacteria include Klebsiella, Proteus, Enterobacter, Serratia, and Eikenella corrodens[3]
Viral
Viral etiologies include:
- Herpes simplex virus (HSV)
Risk Factors
Risk factors of neonatal conjunctivitis may include:
- Maternal infections harbored in the mother's birth canal
- Maternal HIV-infection[4]
- Exposure of the infant to infectious organisms
- Increased birth weight[4]
- Inadequacy of ocular prophylaxis immediately after birth
- Premature rupture of membranes (PROM)[5]
- Ocular trauma during delivery
- Mechanical ventilation
- Prematurity
- Poor prenatal care
- Poor hygienic delivery conditions
- Post-delivery infection due to direct contact with health care workers or by aerosolization
- Silver nitrate exposure
Pathophysiology
Neonatal conjunctivitis is an ocular surface disease consisting of inflammation of the conjunctiva, blood vessel dilation, tearing, and purulent or non-purulent drainage within 30 days of age. Newborns are vulnerable to developing conjunctivitis due to reduced tear secretion and drainage, as well as decreased immune function. Furthermore, neonates lack lymphoid tissue in the conjunctiva, immunoglobulin A and adequate lysozyme activity which normally serve as natural barriers against infectious pathogens. As a result, neonatal conjunctivitis often gets transmitted during passage through the vaginal canal of a mother with a sexually transmitted infection, and can result in serious complications such as corneal ulceration, perforation, and permanent blindness.
Prevention
Prevention through good prenatal care and treatment of chlamydial, gonococcal, or herpetic infections during pregnancy remains the best preventative method.
- Chlamydial infections occur in 4–10% of pregnant women in the United States
- Infants whose mothers have untreated chlamydial infections have a 30–40% chance of developing conjunctivitis (incidence of 6.2 per 1000 live births)
Topical Prophylaxis
Use of topical silver nitrate to prevent neonatal gonococcal conjunctivitis was first introduced by Credé in 1880 and has been classically been cited as the most common cause of neonatal conjunctivitis. However, the incidence of chemical conjunctivitis has declined as the use of silver nitrate as prophylaxis has been abandoned in many modern countries in favor of topical medications with a more favorable side effect profile such as erythromycin. However, countries often vary in their accessibility to these more favorable topical antibiotics. One study evaluating the current methods of treatment in maternity hospitals in Croatia showed that 75% of all maternity hospitals in Croatia routinely administered preventive topical treatment, including topical tobramycin (83.3%), povidone-iodine (8.3%), erythromycin (4.2%), and silver nitrate (4.2%),[6] partially due to the lack of availability of erythromycin and tetracycline in Croatia. Data supporting the superiority of one topical antibiotic over another seem to remain unequivocal. A recent systematic review including 30 trials found little evidence that any prophylaxis involving tetracycline 1%, erythromycin 0.5%, povidone-iodine 2.5%, or silver nitrate 1% provided better prevention from serious outcomes such as blindness or any adverse visual outcomes.[7]
Systemic Prophylaxis
Infants with possible infectious exposure in utero or during birth process should receive appropriate prophylaxis following birth in attempt to prevent ocular and systemic complications. Gonococcal prophylaxis includes single injection of ceftriaxone 50 mg/kg IM or IV in those neonates born to mothers with untreated or suspected gonococcal infection. Other preventative measures include proper hand-washing techniques by peripartum and nursery staff.
Maternal Screening
The United States Preventive Services Task (USPSTF) currently recommends routine screening for chlamydial and gonorrheal infection in all sexually active or pregnant women if they are 24 years or younger, or at least 25 years and at increased risk for infection. These preventative screening measures have significantly decreased perinatal chlamydial and gonorrheal infections in the United States. One study done at a medical center in the United States reports a decrease from 15.6% positivity rate of Chlamydia trachomatis eye cultures before the implementation of universal prenatal screening (1986-1993), to 1.8% during the screening period (1994-2002).[8]
Diagnosis
Prompt diagnosis is key in establishing proper treatment and minimizing potential serious complications of neonatal conjunctivitis.
History
Time frame of signs/symptoms following birth play an important role in determining the most likely etiology and subsequent proper diagnosis and treatment:
- Chemical conjunctivitis (presents within the first 24 hours following birth)
- Neisseria gonorrhea (presents 3-5 days after birth)
- Chlamydia trachomatis (presents 5-14 days after birth)
- HSV (presents 1-2 weeks after birth)
Physical examination
A thorough examination of the globe and periocular structures of a neonate suspected to have neonatal conjunctivitis is crucial. Corneal involvement should be investigated closely with and without fluorescein and blue cobalt light. A complete systemic examination should be performed by trained physician familiar with the physical exam of a neonate.
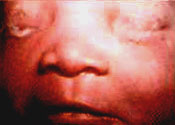
Signs/Symptoms
Non-specific signs of neonatal conjunctivitis include conjunctival injection, tearing, mucopurulent or non-purulent discharge, chemosis, and eyelid swelling.
Signs of specific etiologies include:
Chemical
- Typically results in mild conjunctival injection accompanied by tearing, spontaneously resolving within 2-4 days
Chlamydia trachomatis
- Presentation may range from mild hyperemia with scant mucoid discharge to eyelid swelling, chemosis, and pseudomembrane formation
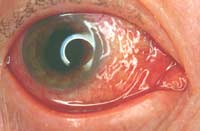
Neisseria gonorrhea
- Typically, patients present with acute conjunctivitis, associated with chemosis, severe lid edema, and mucopurulent discharge
- Corneal involvement is the most serious complication, involving diffuse epithelial edema and ulceration that may progress to perforation of the cornea and endophthalmitis
- Initially, superficial keratitis gives the corneal surface a lackluster appearance followed by marginal and central infiltrates appear, which then ulcerate
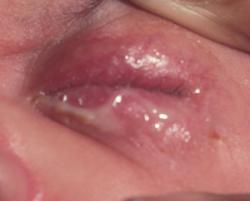
HSV
- Typically, patients present with unilateral or bilateral lid edema, moderate amount of conjunctival injection, and non-purulent, serosanguinous discharge
- Other signs include vesicles on the skin surrounding the eye and corneal epithelial involvement with microdendrites or geographic ulcers (in contrast to typical dendrites as seen in adults)
Differential Diagnosis
The differential diagnosis of neonatal conjunctivitis includes:
- Dacyrocysitis
- Congenital glaucoma
- Nasolacrimal duct obstruction
- Preseptal/Orbital cellulitis
- Infectious keratitis
Laboratory tests
Laboratory studies for suspected infectious etiology should include the following:
- Conjunctival scraping w/ Gram stain and Giemsa stain for chlamydia.
- Note: Conjunctival specimens for chlamydia testing must include conjunctival epithelial cells because C. trachomatis is an obligate intracellular organism and exudates are not adequate for testing.
- Culture on chocolate agar for N. gonorrheae. (Thayer-Martin media may also be used)
- Culture on blood agar for other strains of bacteria
- Culture for HSV if vesicles present or is suspicious of viral etiology including evidence of perinatal maternal exposure
- Direct antibody testing or PCR may also be indicated.[9]
- Cultures may need to be repeated if symptoms worsen or recur following treatment.
Treatment
Chemical conjunctivitis
No treatment required, as the condition typically resolves within 2-4 days. Supportive care involves artificial tears q.i.d.
Chlamydial conjunctivitis
Treatment should be initiated upon a positive diagnostic test. Oral azithromycin may be used for both chlamydial conjunctivitis and pneumonia, 20 mg/kg once daily for three days. Alternatively, erythromycin can be given orally 50 mg/kg/day divided in 4 doses, for 14 days. Topical therapy for chlamydial conjunctivitis has proven to be relatively ineffective compared to oral therapy, particularly in treating nasopharyngeal infection.[10]
The use of azithromycin and erythromycin are associated with increased risk of infantile hypertrophic pyloric stenosis (IHPS) in infants within 2 weeks of life. Infants should be closely monitored for potential signs of IHPS when started on azithromycin or erythromycin.
Gonococcal conjunctivitis
Topical irrigation with normal saline should be used to remove mucopurulent discharge, as Gonococcal infection may produce more mucopurulent discharge than other pathogens. Ceftriaxone should be administered in a single dose (25-50 mg/kg IM or IV, up to a maximum of 125 mg). If there is systemic disease, treatment is required for 7 to 14 days, depending on the nature of the invasive infection. Bacitracin or erythromycin ointment should be administered every 2 to 4 hours in addition to systemic treatment of ceftriaxone. Hospitalization and evaluation for disseminated N. gonorrhea infection should be initiated. Topical atropine should be considered for pain relief if there is corneal involvement.
The use of ceftriaxone in infants may cause hyperbilirubinemia, and therefore should be avoided in neonates receiving calcium-containing intravenous fluids. Cefotaxime or ceftazidime may be used instead.
Of note, all neonates with gonococcal conjunctivitis should also be treated for chlamydia. The patient's mother and sexual partner should be treated as well.
HSV keratoconjunctivitis
Acyclovir IV 45mg/kg/day plus vidarabine 3% ointment 5x/day should be administered for 14-21 days depending on presence of central nervous system involvement.
Other bacteria:
- Gram(+): Bacitracin ointment q.i.d. for 2 weeks
- Gram(-): Gentamicin, tobramycin or ciprofloxacin q.i.d. for 2 weeks
Medical follow up
- Patients with neonatal conjunctivitis should be followed daily for signs of improvement or worsening, especially acutely due to concerns of rapidly progressing infectious complications
- Patient should be followed closely by pediatrician for evaluation and treatment of potential systemic infection.
Complications
Ocular complications of neonatal conjunctivitis include pseudomembrane formation, corneal edema, thickened palpebral conjunctiva, peripheral pannus formation, corneal opacification, corneal perforation, endophthalmitis, loss of eye, and blindness.
Systemic complications of chlamydia conjunctivitis include pneumonitis, otitis, and pharyngeal and rectal colonization. Pneumonia has been reported in 10-20% of infants with chlamydial conjunctivitis. Complications of gonococcal conjunctivitis and subsequent systemic involvement include arthritis, meningitis, anorectal infection, septicemia, and death. Furthermore, Gonococci have the added potential of being able to penetrate intact corneal epithelium, leading to corneal epithelial edema, ulceration, perforation, and endophthalmitis if unrecognized.[11]
Risk of complications can be minimized with prompt diagnosis and appropriate antibiotic therapy.
Prognosis
The prognosis of neonatal conjunctivitis is generally considered to be good as long as early diagnosis is made and prompt medical therapy is initiated. Most cases of infectious conjunctivitis respond to appropriate treatment. However, morbidity and mortality increases in cases of systemic involvement requiring hospitalization and intensive monitoring.
Additional Resources
- http://www.cdc.gov/conjunctivitis/newborns.html
- AAO Preferred Practice Pattern: Conjunctivitis
- AAPOS FAQ about Conjunctivitis
References
- ↑ Opoku-Agyeman K, Adu-Gyamfi PKT, Ansah C, Mensah KD. Effects of prenatal antibiotic treatment on early infant health: a retrospective study in a rural health facility in Ghana. African Health Sciences. 2023 April 11. doi: 10.4314/ahs.v23i1.44.
- ↑ Adachi KN, Nielsen-Saines K, Klausner JD. Chlamydia trachomatis Screening and Treatment in Pregnancy to Reduce Adverse Pregnancy and Neonatal Outcomes: A Review. Front Public Health. 2021 Jun 10;9:531073. doi: 10.3389/fpubh.2021.531073. PMID: 34178906; PMCID: PMC8222807.
- ↑ Chhabra MS, Motley WW 3rd, Mortensen JE. Eikenella corrodens as a causative agent for neonatal conjunctivitis.J AAPOS. 2008 Oct;12(5):524-5.
- ↑ Jump up to: 4.0 4.1 Gichuhi S et al. Risk factors for neonatal conjunctivitis in babies of HIV-1 infected mothers. Ophthalmic Epidemiol. 2009 Nov-Dec;16(6):337-45.
- ↑ Wu J et al. Influence of premature rupture of membranes on neonatal health. Zhonghua Er Ke Za Zhi. 2009 Jun;47(6):452-6.
- ↑ Kaštelan S, Anić Jurica S, Orešković S, Župić T, Herman M, Gverović Antunica A, Marković I, Bakija I. A Survey of Current Prophylactic Treatment for Ophthalmia Neonatorum in Croatia and a Review of International Preventive Practices. Med Sci Monit. 2018 Nov 10;24:8042-8047. doi: 10.12659/MSM.910705. PMID: 30413681; PMCID: PMC6240167.
- ↑ Kapoor VS, Evans JR, Vedula SS. Interventions for preventing ophthalmia neonatorum. Cochrane Database Syst Rev. 2020 Sep 21;9(9):CD001862. doi: 10.1002/14651858.CD001862.pub4. PMID: 32959365; PMCID: PMC8524318.
- ↑ Kohlhoff S, Roblin PM, Clement S, Banniettis N, Hammerschlag MR. Universal Prenatal Screening and Testing and Chlamydia trachomatis Conjunctivitis in Infants. Sex Transm Dis. 2021 Sep 1;48(9):e122-e123. doi: 10.1097/OLQ.0000000000001344. PMID: 33346588.
- ↑ Yip PP et al. The use of polymerase chain reaction assay versus conventional methods in detecting neonatal chlamydial conjunctivitis. J Pediatr Ophthalmol Strabismus. 2008 Jul-Aug;45(4):234-9.
- ↑ Heggie AD, Jaffe AC, Stuart LA, Thombre PS, Sorensen RU. Topical sulfacetamide vs oral erythromycin for neonatal chlamydial conjunctivitis. Am J Dis Child. 1985 Jun;139(6):564-6. doi: 10.1001/archpedi.1985.02140080034027. PMID: 3890519.
- ↑ Mallika P, Asok T, Faisal H, Aziz S, Tan A, Intan G. Neonatal conjunctivitis - a review. Malays Fam Physician. 2008 Aug 31;3(2):77-81. PMID: 25606121; PMCID: PMC4170304.